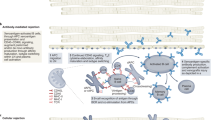
Abstract
Cardiac xenotransplantation (CXTx) might be a promising approach to bridge the gap between the supply and demand of a donor heart. The survival of cardiac xenograft has been significantly extended in pig-to-nonhuman primate (NHP) CXTx, with records of 195 days and 945 days for orthotropic and heterotopic CXTx, respectively. To present the history of CXTx, we list the reported clinical CXTx, compare pigs and NHPs as sources of hearts, and compare three different kinds of preclinical CXTx models. The application of genetically modified pigs and novel immunosuppressive drugs accelerates the development of CXTx, and we summarize the reported pig-to-NHP CXTx with detailed information. Besides, we discuss the underlining mechanisms and potential preventive strategies of immunological barriers, including hyperacute rejection, acute humoral xenograft rejection, acute cellular xenograft rejection, chronic rejection, coagulation dysfunction, and systemic inflammation. Though intense cellular infiltration in cardiac xenograft has only been documented in a small number of studies, we especially stress the importance of cellular rejection in CXTx, because we believe it is often masked by the rapid and strong humoral response and it may eventually become a more important and common type of xenograft rejection. In addition, we conclude other obstacles as well as possible solutions in CXTx, such as perioperative cardiac xenograft dysfunction, detrimental xenograft overgrowth, and porcine endogenous retroviruses. Finally, we briefly introduce several other approaches that have been proposed to deal with the organ heart shortage crisis, and we firmly believe that CXTx provides the best near-term solution.



Similar content being viewed by others
Change history
15 June 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10741-021-10127-4
Abbreviations
- aCD154mAb:
-
Anti-CD154 monoclonal antibody
- aCD20mAb:
-
Anti-CD20 monoclonal antibody
- aCD40mAb:
-
Anti-CD40 monoclonal antibody
- apC:
-
Activated protein C
- ACXR:
-
Acute cellular xenograft rejection
- ADCC:
-
Antibody-dependent cytotoxicity
- AHXR:
-
Acute humoral xenograft rejection
- APC:
-
Antigen-presenting cell
- AT:
-
Antithrombin
- β4GALNT2:
-
β-1,4-N-Acetyl-galactosa-minyl transferase 2
- CC:
-
Consumptive coagulation
- CCS:
-
Cyclophosphamide, cyclosporine A, and steroids/corticosteroids
- CMAH:
-
Cytidine monophosphate-N-acetylneuraminic acid hydroxylase
- CR1:
-
Complement receptor type 1
- C-RP:
-
C-reactive protein
- CVF:
-
Cobra venom factor
- CXTx:
-
Cardiac xenotransplantation
- DXR:
-
Delayed xenograft rejection
- FcR:
-
Fc receptor
- FDA:
-
Food and Drug Administration
- GP1b:
-
Glycoprotein 1b
- GT:
-
Galactosyltransferase
- hCRP:
-
Human complement regulatory proteins
- hEPCR:
-
Human endothelial protein C-receptor
- hTBM:
-
Human thrombomodulin
- hTFPI:
-
Human tissue factor pathway inhibitor
- HAR:
-
Hyperacute rejection
- HLA-I:
-
Human leukocyte antigen class I
- Ig:
-
Immune globulin
- IL:
-
Interleukin
- I/R:
-
Ischemic/reperfusion
- ISHLT:
-
International Society for Heart and Lung Transplantation
- MAC:
-
Membrane attack complex
- MHC-I:
-
Major histocompatibility antigens class I
- MMF:
-
Mycophenolate mofetil
- NHP:
-
Nonhuman primate
- NK:
-
Natural killer
- pC:
-
Protein C
- PCXD:
-
Perioperative cardiac xenograft dysfunction
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
Programmed cell death ligand 1
- pEC:
-
Porcine endothelial cell
- PERVs:
-
Porcine endogenous retrovirus
- SIRP-α:
-
Signal regulatory protein α
- SLA-I:
-
Swine leukocyte antigen class I
- SIXR:
-
Systemic inflammation in xenograft recipients
- TCRs:
-
T cell receptors
- TF:
-
Tissue factor
- TGF-β:
-
Transforming growth factor β
- TMA:
-
Thrombotic microangiopathy
- TNF-α:
-
Tumor necrosis factor-α
- vWF:
-
von Willebrand Factor
References
Tanai E, Frantz S (2015) Pathophysiology of heart failure. Comprehensive Physiology 6(1):187–214. https://doi.org/10.1002/cphy.c140055
Stehlik J, Kobashigawa J, Hunt SA, Reichenspurner H, Kirklin JK (2018) Honoring 50 years of clinical heart transplantation in circulation: in-depth state-of-the-art review. Circulation 137(1):71–87. https://doi.org/10.1161/circulationaha.117.029753
Cooper DK, Ekser B, Ramsoondar J, Phelps C, Ayares D (2016) The role of genetically engineered pigs in xenotransplantation research. J Pathol 238(2):288–299. https://doi.org/10.1002/path.4635
Wang Y, Lei T, Wei L, Du S, Girani L, Deng S (2019) Xenotransplantation in China: present status. Xenotransplantation 26(1):e12490. https://doi.org/10.1111/xen.12490
Smith M, Dominguez-Gil B, Greer DM, Manara AR, Souter MJ (2019) Organ donation after circulatory death: current status and future potential. Intensive Care Med 45(3):310–321. https://doi.org/10.1007/s00134-019-05533-0
Matai I, Kaur G, Seyedsalehi A, McClinton A, Laurencin CT (2020) Progress in 3D bioprinting technology for tissue/organ regenerative engineering. Biomaterials 226:119536. https://doi.org/10.1016/j.biomaterials.2019.119536
Girani L, Xie X, Lei T, Wei L, Wang Y, Deng S (2019) Xenotransplantation in Asia Xenotransplantation 26 (1):e12493. doi:https://doi.org/10.1111/xen.12493
Giwa S, Lewis JK, Alvarez L, Langer R, Roth AE, Church GM, Markmann JF, Sachs DH, Chandraker A, Wertheim JA, Rothblatt M, Boyden ES, Eidbo E, Lee WPA, Pomahac B, Brandacher G, Weinstock DM, Elliott G, Nelson D, Acker JP, Uygun K, Schmalz B, Weegman BP, Tocchio A, Fahy GM, Storey KB, Rubinsky B, Bischof J, Elliott JAW, Woodruff TK, Morris GJ, Demirci U, Brockbank KGM, Woods EJ, Ben RN, Baust JG, Gao D, Fuller B, Rabin Y, Kravitz DC, Taylor MJ, Toner M (2017) The promise of organ and tissue preservation to transform medicine. Nat Biotechnol 35(6):530–542. https://doi.org/10.1038/nbt.3889
Längin M, Mayr T, Reichart B, Michel S, Buchholz S, Guethoff S, Dashkevich A, Baehr A, Egerer S, Bauer A, Mihalj M, Panelli A, Issl L, Ying J, Fresch AK, Buttgereit I, Mokelke M, Radan J, Werner F, Lutzmann I, Steen S, Sjöberg T, Paskevicius A, Qiuming L, Sfriso R, Rieben R, Dahlhoff M, Kessler B, Kemter E, Kurome M, Zakhartchenko V, Klett K, Hinkel R, Kupatt C, Falkenau A, Reu S, Ellgass R, Herzog R, Binder U, Wich G, Skerra A, Ayares D, Kind A, Schönmann U, Kaup F-J, Hagl C, Wolf E, Klymiuk N, Brenner P, Abicht J-M (2018) Consistent success in life-supporting porcine cardiac xenotransplantation. Nature 564(7736):430–433. https://doi.org/10.1038/s41586-018-0765-z
Mohiuddin MM, Singh AK, Corcoran PC, Thomas Iii ML, Clark T, Lewis BG, Hoyt RF, Eckhaus M, Pierson Iii RN, Belli AJ, Wolf E, Klymiuk N, Phelps C, Reimann KA, Ayares D, Horvath KA (2016) Chimeric 2C10R4 anti-CD40 antibody therapy is critical for long-term survival of GTKO.hCD46.hTBM pig-to-primate cardiac xenograft. Nature communications 7:11138. https://doi.org/10.1038/ncomms11138
Lower RR, Shumway NE (1960) Studies on orthotopic homotransplantation of the canine heart. Surg Forum 11:18–19
Hardy JD, Chavez CM (1969) The first heart transplant in man: historical reexamination of the 1964 case in the light of current clinical experience. Transplant Proc 1(2):717–725
Adams DH, Chen RH, Kadner A (2000) Cardiac xenotransplantation: clinical experience and future direction. Ann Thorac Surg 70(1):320–326. https://doi.org/10.1016/s0003-4975(00)01281-9
Murthy R, Bajona P, Bhama JK, Cooper DK (2016) Heart xenotransplantation: historical background, experimental progress, and clinical prospects. Ann Thorac Surg 101(4):1605–1613. https://doi.org/10.1016/j.athoracsur.2015.10.017
Cooley DA, Hallman GL, Bloodwell RD, Nora JJ, Leachman RD (1968) Human heart transplantation. Experience with twelve cases. Am J Cardiol 22(6):804–810. https://doi.org/10.1016/0002-9149(68)90175-6
Barnard CN, Wolpowitz A, Losman JG (1977) Heterotopic cardiac transplantation with a xenograft for assistance of the left heart in cardiogenic shock after cardiopulmonary bypass. South African medical journal = Suid-Afrikaanse tydskrif vir geneeskunde 52(26):1035–1038
Bailey LL, Nehlsen-Cannarella SL, Concepcion W, Jolley WB (1985) Baboon-to-human cardiac xenotransplantation in a neonate. Jama 254(23):3321–3329
Czaplicki J, Blonska B, Religa Z (1992) The lack of hyperacute xenogeneic heart transplant rejection in a human. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 11(2 Pt 1):393–397
Appel JZ 3rd, Buhler L, Cooper DK (2001) The pig as a source of cardiac xenografts. J Card Surg 16(5):345–356. https://doi.org/10.1111/j.1540-8191.2001.tb00534.x
Niemann H, Rath D (2001) Progress in reproductive biotechnology in swine. Theriogenology 56(8):1291–1304. https://doi.org/10.1016/s0093-691x(01)00630-6
Sykes M, Sachs DH (2019) Transplanting organs from pigs to humans. 4 (41):eaau6298. doi:https://doi.org/10.1126/sciimmunol.aau6298%J Science Immunology
Allan JS (1998) The risk of using baboons as transplant donors. Exogenous and endogenous viruses. Annals of the New York Academy of Sciences 862:87–99. https://doi.org/10.1111/j.1749-6632.1998.tb09120.x
Butler D (1999) FDA warns on primate xenotransplants. Nature 398(6728):549. https://doi.org/10.1038/19144
Postrach J, Bauer A, Schmoeckel M, Reichart B, Brenner P (2012) Heart xenotransplantation in primate models. Methods in molecular biology (Clifton, NJ) 885:155–168. https://doi.org/10.1007/978-1-61779-845-0_10
Bauer A, Postrach J, Thormann M, Blanck S, Faber C, Wintersperger B, Michel S, Abicht JM, Christ F, Schmitz C, Schmoeckel M, Reichart B, Brenner P (2010) First experience with heterotopic thoracic pig-to-baboon cardiac xenotransplantation. Xenotransplantation 17(3):243–249. https://doi.org/10.1111/j.1399-3089.2010.00587.x
Abicht JM, Mayr T, Reichart B, Buchholz S, Werner F, Lutzmann I, Schmoeckel M, Bauer A, Thormann M, Langenmayer M, Herbach N, Pohla H, Herzog R, McGregor CG, Ayares D, Wolf E, Klymiuk N, Baehr A, Kind A, Hagl C, Ganswindt U, Belka C, Guethoff S, Brenner P (2015) Pre-clinical heterotopic intrathoracic heart xenotransplantation: a possibly useful clinical technique. Xenotransplantation 22(6):427–442. https://doi.org/10.1111/xen.12213
Mohiuddin MM, Reichart B, Byrne GW, McGregor CGA (2015) Current status of pig heart xenotransplantation. International journal of surgery (London, England) 23 (Pt B):234-239. doi:https://doi.org/10.1016/j.ijsu.2015.08.038
Cooper DK, Keogh AM, Brink J, Corris PA, Klepetko W, Pierson RN, Schmoeckel M, Shirakura R, Warner Stevenson L (2000) Report of the Xenotransplantation Advisory Committee of the International Society for Heart and Lung Transplantation: the present status of xenotransplantation and its potential role in the treatment of end-stage cardiac and pulmonary diseases. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 19(12):1125–1165. https://doi.org/10.1016/s1053-2498(00)00224-2
Griesemer A, Yamada K, Sykes M (2014) Xenotransplantation: immunological hurdles and progress toward tolerance. Immunol Rev 258(1):241–258. https://doi.org/10.1111/imr.12152
Good AH, Cooper DK, Malcolm AJ, Ippolito RM, Koren E, Neethling FA, Ye Y, Zuhdi N, Lamontagne LR (1992) Identification of carbohydrate structures that bind human antiporcine antibodies: implications for discordant xenografting in humans. Transplant Proc 24(2):559–562
Cooper DK, Good AH, Koren E, Oriol R, Malcolm AJ, Ippolito RM, Neethling FA, Ye Y, Romano E, Zuhdi N (1993) Identification of alpha-galactosyl and other carbohydrate epitopes that are bound by human anti-pig antibodies: relevance to discordant xenografting in man. Transpl Immunol 1(3):198–205. https://doi.org/10.1016/0966-3274(93)90047-c
Kuwaki K, Tseng YL, Dor FJ, Shimizu A, Houser SL, Sanderson TM, Lancos CJ, Prabharasuth DD, Cheng J, Moran K, Hisashi Y, Mueller N, Yamada K, Greenstein JL, Hawley RJ, Patience C, Awwad M, Fishman JA, Robson SC, Schuurman HJ, Sachs DH, Cooper DK (2005) Heart transplantation in baboons using alpha1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nat Med 11(1):29–31. https://doi.org/10.1038/nm1171
Bouhours D, Pourcel C, Bouhours JE (1996) Simultaneous expression by porcine aorta endothelial cells of glycosphingolipids bearing the major epitope for human xenoreactive antibodies (Gal alpha 1-3Gal), blood group H determinant and N-glycolylneuraminic acid. Glycoconj J 13(6):947–953. https://doi.org/10.1007/bf01053190
Padler-Karavani V, Varki A (2011) Potential impact of the non-human sialic acid N-glycolylneuraminic acid on transplant rejection risk. Xenotransplantation 18(1):1–5. https://doi.org/10.1111/j.1399-3089.2011.00622.x
Byrne GW, Du Z, Stalboerger P, Kogelberg H, McGregor CG (2014) Cloning and expression of porcine beta1,4 N-acetylgalactosaminyl transferase encoding a new xenoreactive antigen. Xenotransplantation 21(6):543–554. https://doi.org/10.1111/xen.12124
Byrne GW, Stalboerger PG, Du Z, Davis TR, McGregor CG (2011) Identification of new carbohydrate and membrane protein antigens in cardiac xenotransplantation. Transplantation 91(3):287–292. https://doi.org/10.1097/TP.0b013e318203c27d
Lutz AJ, Li P, Estrada JL, Sidner RA, Chihara RK, Downey SM, Burlak C, Wang ZY, Reyes LM, Ivary B, Yin F, Blankenship RL, Paris LL, Tector AJ (2013) Double knockout pigs deficient in N-glycolylneuraminic acid and galactose alpha-1,3-galactose reduce the humoral barrier to xenotransplantation. Xenotransplantation 20(1):27–35. https://doi.org/10.1111/xen.12019
Estrada JL, Martens G, Li P, Adams A, Newell KA, Ford ML, Butler JR, Sidner R, Tector M, Tector J (2015) Evaluation of human and non-human primate antibody binding to pig cells lacking GGTA1/CMAH/beta4GalNT2 genes. Xenotransplantation 22(3):194–202. https://doi.org/10.1111/xen.12161
Waterworth PD, Dunning J, Tolan M, Cozzi E, Langford G, Chavez G, White D, Wallwork J (1998) Life-supporting pig-to-baboon heart xenotransplantation. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 17(12):1201–1207
Schmoeckel M, Bhatti FN, Zaidi A, Cozzi E, Waterworth PD, Tolan MJ, Pino-Chavez G, Goddard M, Warner RG, Langford GA, Dunning JJ, Wallwork J, White DJ (1998) Orthotopic heart transplantation in a transgenic pig-to-primate model. Transplantation 65(12):1570–1577. https://doi.org/10.1097/00007890-199806270-00006
Chen RH, Naficy S, Logan JS, Diamond LE, Adams DH (1999) Hearts from transgenic pigs constructed with CD59/DAF genomic clones demonstrate improved survival in primates. Xenotransplantation 6(3):194–200. https://doi.org/10.1034/j.1399-3089.1999.00017.x
Vial CM, Ostlie DJ, Bhatti FN, Cozzi E, Goddard M, Chavez GP, Wallwork J, White DJ, Dunning JJ (2000) Life supporting function for over one month of a transgenic porcine heart in a baboon. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 19(2):224–229. https://doi.org/10.1016/s1053-2498(99)00099-6
Chan JL, Singh AK, Corcoran PC, Thomas ML, Lewis BG, Ayares DL, Vaught T, Horvath KA, Mohiuddin MM (2017) Encouraging experience using multi-transgenic xenografts in a pig-to-baboon cardiac xenotransplantation model. Xenotransplantation 24(6). https://doi.org/10.1111/xen.12330
Bhatti FN, Schmoeckel M, Zaidi A, Cozzi E, Chavez G, Goddard M, Dunning JJ, Wallwork J, White DJ (1999) Three-month survival of HDAFF transgenic pig hearts transplanted into primates. Transplant Proc 31(1–2):958. https://doi.org/10.1016/s0041-1345(98)01855-7
Lam TT, Paniagua R, Shivaram G, Schuurman HJ, Borie DC, Morris RE (2004) Anti-non-Gal porcine endothelial cell antibodies in acute humoral xenograft rejection of hDAF-transgenic porcine hearts in cynomolgus monkeys. Xenotransplantation 11(6):531–535. https://doi.org/10.1111/j.1399-3089.2004.00175.x
Kuwaki K, Knosalla C, Dor FJ, Gollackner B, Tseng YL, Houser S, Mueller N, Prabharasuth D, Alt A, Moran K, Cheng J, Behdad A, Sachs DH, Fishman JA, Schuurman HJ, Awwad M, Cooper DK (2004) Suppression of natural and elicited antibodies in pig-to-baboon heart transplantation using a human anti-human CD154 mAb-based regimen. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 4(3):363–372. https://doi.org/10.1111/j.1600-6143.2004.00353.x
Singh AK, Chan JL, DiChiacchio L, Hardy NL, Corcoran PC, Lewis BGT, Thomas ML, Burke AP, Ayares D, Horvath KA, Mohiuddin MM (2019) Cardiac xenografts show reduced survival in the absence of transgenic human thrombomodulin expression in donor pigs. Xenotransplantation 26(2):e12465. https://doi.org/10.1111/xen.12465
Byrne GW, Davies WR, Oi K, Rao VP, Teotia SS, Ricci D, Tazelaar HD, Walker RC, Logan JS, McGregor CG (2006) Increased immunosuppression, not anticoagulation, extends cardiac xenograft survival. Transplantation 82(12):1787–1791. https://doi.org/10.1097/01.tp.0000251387.40499.0f
Byrne GW, Du Z, Sun Z, Asmann YW, McGregor CG (2011) Changes in cardiac gene expression after pig-to-primate orthotopic xenotransplantation. Xenotransplantation 18(1):14–27. https://doi.org/10.1111/j.1399-3089.2010.00620.x
Mohiuddin MM, Corcoran PC, Singh AK, Azimzadeh A, Hoyt RF Jr, Thomas ML, Eckhaus MA, Seavey C, Ayares D, Pierson RN 3rd, Horvath KA (2012) B-cell depletion extends the survival of GTKO.hCD46Tg pig heart xenografts in baboons for up to 8 months. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 12(3):763–771. https://doi.org/10.1111/j.1600-6143.2011.03846.x
Mohiuddin MM, Singh AK, Corcoran PC, Hoyt RF, Thomas ML 3rd, Lewis BG, Eckhaus M, Dabkowski NL, Belli AJ, Reimann KA, Ayares D, Horvath KA (2014) Role of anti-CD40 antibody-mediated costimulation blockade on non-Gal antibody production and heterotopic cardiac xenograft survival in a GTKO.hCD46Tg pig-to-baboon model. Xenotransplantation 21(1):35–45. https://doi.org/10.1111/xen.12066
Mohiuddin MM, Singh AK, Corcoran PC, Hoyt RF, Thomas ML 3rd, Lewis BG, Eckhaus M, Reimann KA, Klymiuk N, Wolf E, Ayares D, Horvath KA (2014) One-year heterotopic cardiac xenograft survival in a pig to baboon model. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 14(2):488–489. https://doi.org/10.1111/ajt.12562
Kim H, Hawthorne WJ, Kang HJ, Lee YJ, Hwang JI, Hurh S, Ro H, Jeong JC, Cho B, Yang J, Ahn C (2015) Human thrombomodulin regulates complement activation as well as the coagulation cascade in xeno-immune response. Xenotransplantation 22(4):260–272. https://doi.org/10.1111/xen.12173
Jung SH, Hwang JH, Kim SE, Young Kyu K, Park HC, Lee HT (2017) The potentiating effect of hTFPI in the presence of hCD47 reduces the cytotoxicity of human macrophages. Xenotransplantation 24(3). https://doi.org/10.1111/xen.12301
Choi K, Shim J, Ko N, Eom H, Kim J, Lee JW, Jin DI, Kim H (2017) Production of heterozygous alpha 1,3-galactosyltransferase (GGTA1) knock-out transgenic miniature pigs expressing human CD39. Transgenic Res 26(2):209–224. https://doi.org/10.1007/s11248-016-9996-7
Abicht JM, Sfriso R, Reichart B, Langin M, Gahle K, Puga Yung GL, Seebach JD, Rieben R, Ayares D, Wolf E, Klymiuk N, Baehr A, Kind A, Mayr T, Bauer A (2018) Multiple genetically modified GTKO/hCD46/HLA-E/hbeta2-mg porcine hearts are protected from complement activation and natural killer cell infiltration during ex vivo perfusion with human blood. Xenotransplantation 25(5):e12390. https://doi.org/10.1111/xen.12390
Buermann A, Petkov S, Petersen B, Hein R, Lucas-Hahn A, Baars W, Brinkmann A, Niemann H, Schwinzer R (2018) Pigs expressing the human inhibitory ligand PD-L1 (CD 274) provide a new source of xenogeneic cells and tissues with low immunogenic properties. Xenotransplantation 25(5):e12387. https://doi.org/10.1111/xen.12387
Davis EA, Pruitt SK, Greene PS, Ibrahim S, Lam TT, Levin JL, Baldwin WM 3rd, Sanfilippo F (1996) Inhibition of complement, evoked antibody, and cellular response prevents rejection of pig-to-primate cardiac xenografts. Transplantation 62(7):1018–1023. https://doi.org/10.1097/00007890-199610150-00022
Kobayashi T, Taniguchi S, Neethling FA, Rose AG, Hancock WW, Ye Y, Niekrasz M, Kosanke S, Wright LJ, White DJ, Cooper DK (1997) Delayed xenograft rejection of pig-to-baboon cardiac transplants after cobra venom factor therapy. Transplantation 64(9):1255–1261. https://doi.org/10.1097/00007890-199711150-00005
McGregor CG, Ricci D, Miyagi N, Stalboerger PG, Du Z, Oehler EA, Tazelaar HD, Byrne GW (2012) Human CD55 expression blocks hyperacute rejection and restricts complement activation in Gal knockout cardiac xenografts. Transplantation 93(7):686–692. https://doi.org/10.1097/TP.0b013e3182472850
Niu D, Wei HJ, Lin L, George H, Wang T, Lee IH, Zhao HY, Wang Y, Kan Y, Shrock E, Lesha E, Wang G, Luo Y, Qing Y, Jiao D, Zhao H, Zhou X, Wang S, Wei H, Guell M, Church GM, Yang L (2017) Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9. Science (New York, NY) 357(6357):1303–1307. https://doi.org/10.1126/science.aan4187
Cooper DKC, Hara H, Iwase H, Yamamoto T, Li Q, Ezzelarab M, Federzoni E, Dandro A, Ayares D (2019) Justification of specific genetic modifications in pigs for clinical organ xenotransplantation. Xenotransplantation 26(4):e12516. https://doi.org/10.1111/xen.12516
Cooper DKC, Ezzelarab M, Iwase H, Hara H (2018) Perspectives on the optimal genetically engineered pig in 2018 for initial clinical trials of kidney or heart xenotransplantation. Transplantation 102(12):1974–1982. https://doi.org/10.1097/TP.0000000000002443
Yamada K, Sykes M, Sachs DH (2017) Tolerance in xenotransplantation. Current opinion in organ transplantation 22(6):522–528. https://doi.org/10.1097/MOT.0000000000000466
Liu L, He C, Liu J, Lv Z, Wang G, Gao H, Dai Y, Cooper DKC, Cai Z, Mou L (2018) Transplant tolerance: current insights and strategies for long-term survival of xenografts. Arch Immunol Ther Exp 66(5):355–364. https://doi.org/10.1007/s00005-018-0517-7
Llore NP, Bruestle KA, Griesemer A (2018) Xenotransplantation tolerance: applications for recent advances in modified swine. Current opinion in organ transplantation 23(6):642–648. https://doi.org/10.1097/MOT.0000000000000585
Cooper DK, Ezzelarab MB, Hara H, Iwase H, Lee W, Wijkstrom M, Bottino R (2016) The pathobiology of pig-to-primate xenotransplantation: a historical review. Xenotransplantation 23(2):83–105. https://doi.org/10.1111/xen.12219
Kobayashi T, Cooper DK (1999) Anti-Gal, alpha-Gal epitopes, and xenotransplantation. Subcell Biochem 32:229–257. https://doi.org/10.1007/978-1-4615-4771-6_10
Muller-Eberhard HJ (1988) Molecular organization and function of the complement system. Annu Rev Biochem 57:321–347. https://doi.org/10.1146/annurev.bi.57.070188.001541
Morgan BP (1995) Physiology and pathophysiology of complement: progress and trends. Crit Rev Clin Lab Sci 32(3):265–298. https://doi.org/10.3109/10408369509084686
Sandrin MS, Fodor WL, Mouhtouris E, Osman N, Cohney S, Rollins SA, Guilmette ER, Setter E, Squinto SP, McKenzie IF (1995) Enzymatic remodelling of the carbohydrate surface of a xenogenic cell substantially reduces human antibody binding and complement-mediated cytolysis. Nat Med 1(12):1261–1267. https://doi.org/10.1038/nm1295-1261
Cabezas-Cruz A, Mateos-Hernandez L, Alberdi P, Villar M, Riveau G, Hermann E, Schacht AM, Khalife J, Correia-Neves M, Gortazar C, de la Fuente J (2017) Effect of blood type on anti-alpha-Gal immunity and the incidence of infectious diseases. Exp Mol Med 49(3):e301. https://doi.org/10.1038/emm.2016.164
Galili U, Mandrell RE, Hamadeh RM, Shohet SB, Griffiss JM (1988) Interaction between human natural anti-alpha-galactosyl immunoglobulin G and bacteria of the human flora. Infect Immun 56(7):1730–1737
Cooper DK, Human PA, Lexer G, Rose AG, Rees J, Keraan M, Du Toit E (1988) Effects of cyclosporine and antibody adsorption on pig cardiac xenograft survival in the baboon. The Journal of heart transplantation 7(3):238–246
Fischel RJ, Matas AJ, Perry E, Dalmasso A, Noreen H, Bolman RM 3rd (1992) Plasma exchange, organ perfusion, and immunosuppression reduce “natural” antibody levels as measured by binding to xenogeneic endothelial cells and prolong discordant xenograft survival. Transplant Proc 24(2):574–575
Besse T, Duck L, Latinne D, Gianello P, Squifflet JP, Lievens M, Tomasi JP, Lavenne-Pardonge EM, Lambotte L, Alexandre GP et al (1994) Effect of plasmapheresis and splenectomy on parameters involved in vascular rejection of discordant xenografts in the swine to baboon model. Transplant Proc 26(2):1042–1044
Taniguchi S, Neethling FA, Korchagina EY, Bovin N, Ye Y, Kobayashi T, Niekrasz M, Li S, Koren E, Oriol R, Cooper DK (1996) In vivo immunoadsorption of antipig antibodies in baboons using a specific Gal (alpha)1-3Gal column. Transplantation 62(10):1379–1384. https://doi.org/10.1097/00007890-199611270-00001
Kobayashi T, Taniguchi S, Ye Y, Niekrasz M, Kosanke S, Neethling FA, Wright LJ, Rose AG, White DJ, Cooper DK (1996) Delayed xenograft rejection in C3-depleted discordant (pig-to-baboon) cardiac xenografts treated with cobra venom factor. Transplant Proc 28(2):560
Byrne GW, Azimzadeh AM, Ezzelarab M, Tazelaar HD, Ekser B, Pierson RN, Robson SC, Cooper DK, McGregor CG (2013) Histopathologic insights into the mechanism of anti-non-Gal antibody-mediated pig cardiac xenograft rejection. Xenotransplantation 20(5):292–307. https://doi.org/10.1111/xen.12050
Platt JL (2000) Acute vascular rejection. Transplant Proc 32(5):839–840. https://doi.org/10.1016/s0041-1345(00)01000-9
Hisashi Y, Yamada K, Kuwaki K, Tseng YL, Dor FJ, Houser SL, Robson SC, Schuurman HJ, Cooper DK, Sachs DH, Colvin RB, Shimizu A (2008) Rejection of cardiac xenografts transplanted from alpha1,3-galactosyltransferase gene-knockout (GalT-KO) pigs to baboons. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 8(12):2516–2526. https://doi.org/10.1111/j.1600-6143.2008.02444.x
Xu H, Gundry SR, Hancock WW, Matsumiya G, Zuppan CW, Morimoto T, Slater J, Bailey LL (1998) Prolonged discordant xenograft survival and delayed xenograft rejection in a pig-to-baboon orthotopic cardiac xenograft model. J Thorac Cardiovasc Surg 115(6):1342–1349. https://doi.org/10.1016/s0022-5223(98)70218-1
Itescu S, Kwiatkowski P, Artrip JH, Wang SF, Ankersmit J, Minanov OP, Michler RE (1998) Role of natural killer cells, macrophages, and accessory molecule interactions in the rejection of pig-to-primate xenografts beyond the hyperacute period. Hum Immunol 59(5):275–286. https://doi.org/10.1016/s0198-8859(98)00026-3
Lanier LL (2005) NK cell recognition. Annu Rev Immunol 23:225–274. https://doi.org/10.1146/annurev.immunol.23.021704.115526
Watier H, Guillaumin JM, Vallee I, Thibault G, Gruel Y, Lebranchu Y, Bardos P (1996) Human NK cell-mediated direct and IgG-dependent cytotoxicity against xenogeneic porcine endothelial cells. Transpl Immunol 4(4):293–299. https://doi.org/10.1016/s0966-3274(96)80050-5
Watier H, Guillaumin JM, Piller F, Lacord M, Thibault G, Lebranchu Y, Monsigny M, Bardos P (1996) Removal of terminal alpha-galactosyl residues from xenogeneic porcine endothelial cells. Decrease in complement-mediated cytotoxicity but persistence of IgG1-mediated antibody-dependent cell-mediated cytotoxicity. Transplantation 62(1):105–113. https://doi.org/10.1097/00007890-199607150-00020
Long EO, Kim HS, Liu D, Peterson ME, Rajagopalan S (2013) Controlling natural killer cell responses: integration of signals for activation and inhibition. Annu Rev Immunol 31:227–258. https://doi.org/10.1146/annurev-immunol-020711-075005
Sullivan JA, Oettinger HF, Sachs DH, Edge AS (1997) Analysis of polymorphism in porcine MHC class I genes: alterations in signals recognized by human cytotoxic lymphocytes. Journal of immunology (Baltimore, Md : 1950) 159(5):2318–2326
Puga Yung G, Schneider MKJ, Seebach JD (2017) The role of NK cells in pig-to-human xenotransplantation. J Immunol Res 2017:4627384. https://doi.org/10.1155/2017/4627384
Lin Y, Vandeputte M, Waer M (1997) Natural killer cell- and macrophage-mediated rejection of concordant xenografts in the absence of T and B cell responses. Journal of immunology (Baltimore, Md : 1950) 158(12):5658–5667
Ide K, Wang H, Tahara H, Liu J, Wang X, Asahara T, Sykes M, Yang YG, Ohdan H (2007) Role for CD47-SIRPalpha signaling in xenograft rejection by macrophages. Proc Natl Acad Sci U S A 104(12):5062–5066. https://doi.org/10.1073/pnas.0609661104
Greenwald AG, Jin R, Waddell TK (2009) Galectin-3-mediated xenoactivation of human monocytes. Transplantation 87(1):44–51. https://doi.org/10.1097/TP.0b013e318191e6b4
Basker M, Alwayn IP, Buhler L, Harper D, Abraham S, Kruger Gray H, DeAngelis H, Awwad M, Down J, Rieben R, White-Scharf ME, Sachs DH, Thall A, Cooper DK (2001) Clearance of mobilized porcine peripheral blood progenitor cells is delayed by depletion of the phagocytic reticuloendothelial system in baboons. Transplantation 72(7):1278–1285. https://doi.org/10.1097/00007890-200110150-00017
Peterson MD, Jin R, Hyduk S, Duchesneau P, Cybulsky MI, Waddell TK (2005) Monocyte adhesion to xenogeneic endothelium during laminar flow is dependent on alpha-Gal-mediated monocyte activation. Journal of immunology (Baltimore, Md : 1950) 174(12):8072–8081. https://doi.org/10.4049/jimmunol.174.12.8072
El-Ouaghlidi A, Jahr H, Pfeiffer G, Hering BJ, Brandhorst D, Brandhorst H, Federlin K, Bretzel RG (1999) Cytokine mRNA expression in peripheral blood cells of immunosuppressed human islet transplant recipients. Journal of molecular medicine (Berlin, Germany) 77(1):115–117. https://doi.org/10.1007/s001090050315
Cadili A, Kneteman N (2008) The role of macrophages in xenograft rejection. Transplant Proc 40(10):3289–3293. https://doi.org/10.1016/j.transproceed.2008.08.125
Maeda A, Lo PC, Sakai R, Noguchi Y, Kodama T, Yoneyama T, Toyama C, Wang HT, Esquivel E, Jiaravuthisan P, Choi TV, Takakura C, Eguchi H, Tazuke Y, Watanabe M, Nagashima H, Okuyama H, Miyagawa S (2020) A strategy for suppressing macrophage-mediated rejection in xenotransplantation. Transplantation 104(4):675–681. https://doi.org/10.1097/tp.0000000000003024
Yang YG (2010) CD47 in xenograft rejection and tolerance induction. Xenotransplantation 17(4):267–273. https://doi.org/10.1111/j.1399-3089.2010.00601.x
Sykes M (2018) IXA Honorary Member Lecture, 2017: the long and winding road to tolerance. Xenotransplantation 25(3):e12419. https://doi.org/10.1111/xen.12419
Rijkers ES, de Ruiter T, Baridi A, Veninga H, Hoek RM, Meyaard L (2008) The inhibitory CD200R is differentially expressed on human and mouse T and B lymphocytes. Mol Immunol 45(4):1126–1135. https://doi.org/10.1016/j.molimm.2007.07.013
Yan JJ, Koo TY, Lee HS, Lee WB, Kang B, Lee JG, Jang JY, Fang T, Ryu JH, Ahn C, Kim SJ, Yang J (2018) Role of human CD200 overexpression in pig-to-human xenogeneic immune response compared with human CD47 overexpression. Transplantation 102(3):406–416. https://doi.org/10.1097/tp.0000000000001966
Cella M, Dohring C, Samaridis J, Dessing M, Brockhaus M, Lanzavecchia A, Colonna M (1997) A novel inhibitory receptor (ILT3) expressed on monocytes, macrophages, and dendritic cells involved in antigen processing. J Exp Med 185(10):1743–1751. https://doi.org/10.1084/jem.185.10.1743
Jiaravuthisan P, Maeda A, Takakura C, Wang HT, Sakai R, Shabri AM, Lo PC, Matsuura R, Kodama T, Eguchi H, Okuyama H, Miyagawa S (2018) A membrane-type surfactant protein D (SP-D) suppresses macrophage-mediated cytotoxicity in swine endothelial cells. Transpl Immunol 47:44–48. https://doi.org/10.1016/j.trim.2018.02.003
Maeda A, Kawamura T, Nakahata K, Ueno T, Usui N, Eguchi H, Miyagawa S (2014) Monocytic suppressor cells derived from peripheral blood suppress xenogenic natural killer cell lysis. Transplant Proc 46(4):1254–1255. https://doi.org/10.1016/j.transproceed.2013.11.025
Davila E, Byrne GW, LaBreche PT, McGregor HC, Schwab AK, Davies WR, Rao VP, Oi K, Tazelaar HD, Logan JS, McGregor CG (2006) T-cell responses during pig-to-primate xenotransplantation. Xenotransplantation 13(1):31–40. https://doi.org/10.1111/j.1399-3089.2005.00258.x
Hosiawa KA, Wang H, DeVries ME, Garcia B, Jiang J, Zhou D, Cameron MJ, Zhong R, Kelvin DJ (2006) Regulation of B- and T-cell mediated xenogeneic transplant rejection by interleukin 12. Transplantation 81(2):265–272. https://doi.org/10.1097/01.tp.0000196725.53277.60
Sebille F, Guillet M, Brouard S, Gagne K, Petzold T, Blancho G, Vanhove B, Soulillou JP (2001) T-cell-mediated rejection of vascularized xenografts in the absence of induced anti-donor antibody response. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 1(1):21–28. https://doi.org/10.1034/j.1600-6143.2001.010106.x
Siu JHY, Surendrakumar V, Richards JA, Pettigrew GJ (2018) T cell allorecognition pathways in solid organ transplantation. Front Immunol 9:2548. https://doi.org/10.3389/fimmu.2018.02548
Higginbotham L, Ford ML, Newell KA, Adams AB (2015) Preventing T cell rejection of pig xenografts. International journal of surgery (London, England) 23 (Pt B):285-290. doi:https://doi.org/10.1016/j.ijsu.2015.07.722
Dorling A, Lombardi G, Binns R, Lechler RI (1996) Detection of primary direct and indirect human anti-porcine T cell responses using a porcine dendritic cell population. Eur J Immunol 26(6):1378–1387. https://doi.org/10.1002/eji.1830260630
Samy KP, Butler JR, Li P, Cooper DKC, Ekser B (2017) The role of costimulation blockade in solid organ and islet xenotransplantation. J Immunol Res 2017:8415205. https://doi.org/10.1155/2017/8415205
Mardomi A, Mohammadi N, Khosroshahi HT, Abediankenari S (2020) An update on potentials and promises of T cell co-signaling molecules in transplantation. J Cell Physiol 235(5):4183–4197. https://doi.org/10.1002/jcp.29369
Iwase H, Ekser B, Satyananda V, Bhama J, Hara H, Ezzelarab M, Klein E, Wagner R, Long C, Thacker J, Li J, Zhou H, Jiang M, Nagaraju S, Zhou H, Veroux M, Bajona P, Wijkstrom M, Wang Y, Phelps C, Klymiuk N, Wolf E, Ayares D, Cooper DK (2015) Pig-to-baboon heterotopic heart transplantation--exploratory preliminary experience with pigs transgenic for human thrombomodulin and comparison of three costimulation blockade-based regimens. Xenotransplantation 22(3):211–220. https://doi.org/10.1111/xen.12167
Koshika T, Phelps C, Fang J, Lee SE, Fujita M, Ayares D, Cooper DK, Hara H (2011) Relative efficiency of porcine and human cytotoxic T-lymphocyte antigen 4 immunoglobulin in inhibiting human CD4+ T-cell responses co-stimulated by porcine and human B7 molecules. Immunology 134(4):386–397. https://doi.org/10.1111/j.1365-2567.2011.03496.x
Houser SL, Kuwaki K, Knosalla C, Dor FJ, Gollackner B, Cheng J, Shimizu A, Schuurman HJ, Cooper DK (2004) Thrombotic microangiopathy and graft arteriopathy in pig hearts following transplantation into baboons. Xenotransplantation 11(5):416–425. https://doi.org/10.1111/j.1399-3089.2004.00155.x
McGregor CG, Teotia SS, Byrne GW, Michaels MG, Risdahl JM, Schirmer JM, Tazelaar HD, Walker RC, Logan JS (2004) Cardiac xenotransplantation: progress toward the clinic. Transplantation 78(11):1569–1575. https://doi.org/10.1097/01.tp.0000147302.64947.43
De Salvatore S, Segreto A, Chiusaroli A, Congiu S, Bizzarri F (2015) Role of xenotransplantation in cardiac transplantation. J Card Surg 30(1):111–116. https://doi.org/10.1111/jocs.12454
Briffa NP, Shorthouse R, Chan J, Silva H, Billingham M, Brazelton T, Morris RE (2004) Histological and immunological characteristics of, and the effect of immunosuppressive treatment on, xenograft vasculopathy. Xenotransplantation 11(2):149–159. https://doi.org/10.1046/j.1399-3089.2003.00099.x
Wang L, Cooper DKC, Burdorf L, Wang Y, Iwase H (2018) Overcoming coagulation dysregulation in pig solid organ transplantation in nonhuman primates: recent progress. Transplantation 102(7):1050–1058. https://doi.org/10.1097/tp.0000000000002171
Lin CC, Cooper DK, Dorling A (2009) Coagulation dysregulation as a barrier to xenotransplantation in the primate. Transpl Immunol 21(2):75–80. https://doi.org/10.1016/j.trim.2008.10.008
Cowan PJ, Robson SC, d’Apice AJ (2011) Controlling coagulation dysregulation in xenotransplantation. Current opinion in organ transplantation 16(2):214–221. https://doi.org/10.1097/MOT.0b013e3283446c65
Knosalla C, Yazawa K, Behdad A, Bodyak N, Shang H, Buhler L, Houser S, Gollackner B, Griesemer A, Schmitt-Knosalla I, Schuurman HJ, Awwad M, Sachs DH, Cooper DK, Yamada K, Usheva A, Robson SC (2009) Renal and cardiac endothelial heterogeneity impact acute vascular rejection in pig-to-baboon xenotransplantation. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 9(5):1006–1016. https://doi.org/10.1111/j.1600-6143.2009.02602.x
Furie B, Furie BC (2008) Mechanisms of thrombus formation. N Engl J Med 359(9):938–949. https://doi.org/10.1056/NEJMra0801082
Mackman N (2008) Triggers, targets and treatments for thrombosis. Nature 451(7181):914–918. https://doi.org/10.1038/nature06797
Denorme F, Vanhoorelbeke K, De Meyer SF (2019) von Willebrand factor and platelet glycoprotein Ib: a thromboinflammatory axis in stroke. Frontiers in immunology 10:2884. https://doi.org/10.3389/fimmu.2019.02884
Roussel JC, Moran CJ, Salvaris EJ, Nandurkar HH, d’Apice AJ, Cowan PJ (2008) Pig thrombomodulin binds human thrombin but is a poor cofactor for activation of human protein C and TAFI. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 8(6):1101–1112. https://doi.org/10.1111/j.1600-6143.2008.02210.x
Schulte Am Esch J 2nd, Robson SC, Knoefel WT, Hosch SB, Rogiers X (2005) O-linked glycosylation and functional incompatibility of porcine von Willebrand factor for human platelet GPIb receptors. Xenotransplantation 12(1):30–37. https://doi.org/10.1111/j.1399-3089.2004.00187.x
Kopp CW, Robson SC, Siegel JB, Anrather J, Winkler H, Grey S, Kaczmarek E, Bach FH, Geczy CL (1998) Regulation of monocyte tissue factor activity by allogeneic and xenogeneic endothelial cells. Thromb Haemost 79(3):529–538
Rataj D, Werwitzke S, Haarmeijer B, Winkler M, Ramackers W, Petersen B, Niemann H, Wunsch A, Bahr A, Klymiuk N, Wolf E, Abicht JM, Ayares D, Tiede A (2016) Inhibition of complement component C5 prevents clotting in an ex vivo model of xenogeneic activation of coagulation. Xenotransplantation 23(2):117–127. https://doi.org/10.1111/xen.12218
Chen D, Weber M, McVey JH, Kemball-Cook G, Tuddenham EG, Lechler RI, Dorling A (2004) Complete inhibition of acute humoral rejection using regulated expression of membrane-tethered anticoagulants on xenograft endothelium. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 4(12):1958–1963. https://doi.org/10.1111/j.1600-6143.2004.00625.x
Ezzelarab MB, Ekser B, Azimzadeh A, Lin CC, Zhao Y, Rodriguez R, Echeverri GJ, Iwase H, Long C, Hara H, Ayares D, Pierson RN, Thomson AW, Cooper DK (2015) Systemic inflammation in xenograft recipients precedes activation of coagulation. Xenotransplantation 22(1):32–47. https://doi.org/10.1111/xen.12133
Iwase H, Liu H, Li T, Zhang Z, Gao B, Hara H, Wijkstrom M, Long C, Saari R, Ayares D, Cooper DKC, Ezzelarab MB (2017) Therapeutic regulation of systemic inflammation in xenograft recipients. Xenotransplantation 24(2). https://doi.org/10.1111/xen.12296
Iwase H, Ekser B, Zhou H, Liu H, Satyananda V, Humar R, Humar P, Hara H, Long C, Bhama JK, Bajona P, Wang Y, Wijkstrom M, Ayares D, Ezzelarab MB, Cooper DK (2015) Further evidence for sustained systemic inflammation in xenograft recipients (SIXR). Xenotransplantation 22(5):399–405. https://doi.org/10.1111/xen.12182
Christenson RH, deFilippi CP, Kreutzer D (2005) Biomarkers of ischemia in patients with known coronary artery disease: do interleukin-6 and tissue factor measurements during dobutamine stress echocardiography give additional insight? Circulation 112(21):3215–3217. https://doi.org/10.1161/circulationaha.105.581918
Ikonomidis I, Athanassopoulos G, Lekakis J, Venetsanou K, Marinou M, Stamatelopoulos K, Cokkinos DV, Nihoyannopoulos P (2005) Myocardial ischemia induces interleukin-6 and tissue factor production in patients with coronary artery disease: a dobutamine stress echocardiography study. Circulation 112(21):3272–3279. https://doi.org/10.1161/circulationaha.104.532259
Strukova S (2006) Blood coagulation-dependent inflammation. Coagulation-dependent inflammation and inflammation-dependent thrombosis. Frontiers in bioscience : a journal and virtual library 11:59–80. https://doi.org/10.2741/1780
Petersen B, Ramackers W, Lucas-Hahn A, Lemme E, Hassel P, Queisser A-L, Herrmann D, Barg-Kues B, Carnwath JW, Klose J, Tiede A, Friedrich L, Baars W, Schwinzer R, Winkler M, Niemann H (2011) Transgenic expression of human heme oxygenase-1 in pigs confers resistance against xenograft rejection during ex vivo perfusion of porcine kidneys. Xenotransplantation 18(6):355–368. https://doi.org/10.1111/j.1399-3089.2011.00674.x
Oropeza M, Petersen B, Carnwath JW, Lucas-Hahn A, Lemme E, Hassel P, Herrmann D, Barg-Kues B, Holler S, Queisser A-L, Schwinzer R, Hinkel R, Kupatt C, Niemann H (2009) Transgenic expression of the human A20 gene in cloned pigs provides protection against apoptotic and inflammatory stimuli. Xenotransplantation 16(6):522–534. https://doi.org/10.1111/j.1399-3089.2009.00556.x
Rieblinger B, Fischer K, Kind A, Saller BS, Baars W, Schuster M, Wolf-van Buerck L, Schäffler A, Flisikowska T, Kurome M, Zakhartchenko V, Kessler B, Flisikowski K, Wolf E, Seissler J, Schwinzer R, Schnieke A (2018) Strong xenoprotective function by single-copy transgenes placed sequentially at a permissive locus. Xenotransplantation 25(2):e12382. https://doi.org/10.1111/xen.12382
Byrne GW, McGregor CG (2012) Cardiac xenotransplantation: progress and challenges. Current opinion in organ transplantation 17(2):148–154. https://doi.org/10.1097/MOT.0b013e3283509120
Steen S, Paskevicius A, Liao Q, Sjöberg T (2016) Safe orthotopic transplantation of hearts harvested 24 hours after brain death and preserved for 24 hours. Scand Cardiovasc J 50(3):193–200. https://doi.org/10.3109/14017431.2016.1154598
Wheeler DG, Joseph ME, Mahamud SD, Aurand WL, Mohler PJ, Pompili VJ, Dwyer KM, Nottle MB, Harrison SJ, d’Apice AJ, Robson SC, Cowan PJ, Gumina RJ (2012) Transgenic swine: expression of human CD39 protects against myocardial injury. J Mol Cell Cardiol 52(5):958–961. https://doi.org/10.1016/j.yjmcc.2012.01.002
Novitzky D, Cooper DK (2014) Thyroid hormone and the stunned myocardium. J Endocrinol 223(1):R1–R8. https://doi.org/10.1530/joe-14-0389
Iwase H, Ekser B, Hara H, Ezzelarab MB, Long C, Thomson AW, Ayares D, Cooper DK (2016) Thyroid hormone: relevance to xenotransplantation. Xenotransplantation 23(4):293–299. https://doi.org/10.1111/xen.12243
Tanabe T, Watanabe H, Shah JA, Sahara H, Shimizu A, Nomura S, Asfour A, Danton M, Boyd L, Dardenne Meyers A, Ekanayake-Alper DK, Sachs DH, Yamada K (2017) Role of intrinsic (graft) versus extrinsic (host) factors in the growth of transplanted organs following allogeneic and xenogeneic transplantation. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 17(7):1778–1790. https://doi.org/10.1111/ajt.14210
Denner J (2016) How active are porcine endogenous retroviruses (PERVs)? Viruses 8(8). https://doi.org/10.3390/v8080215
Denner J, Tonjes RR (2012) Infection barriers to successful xenotransplantation focusing on porcine endogenous retroviruses. Clin Microbiol Rev 25(2):318–343. https://doi.org/10.1128/cmr.05011-11
Harrison I, Takeuchi Y, Bartosch B, Stoye JP (2004) Determinants of high titer in recombinant porcine endogenous retroviruses. J Virol 78(24):13871–13879. https://doi.org/10.1128/jvi.78.24.13871-13879.2004
Scobie L, Padler-Karavani V, Le Bas-Bernardet S, Crossan C, Blaha J, Matouskova M, Hector RD, Cozzi E, Vanhove B, Charreau B, Blancho G, Bourdais L, Tallacchini M, Ribes JM, Yu H, Chen X, Kracikova J, Broz L, Hejnar J, Vesely P, Takeuchi Y, Varki A, Soulillou J-P (2013) Long-term IgG response to porcine Neu5Gc antigens without transmission of PERV in burn patients treated with porcine skin xenografts. Journal of immunology (Baltimore, Md : 1950) 191(6):2907–2915. https://doi.org/10.4049/jimmunol.1301195
Wynyard S, Nathu D, Garkavenko O, Denner J, Elliott R (2014) Microbiological safety of the first clinical pig islet xenotransplantation trial in New Zealand. Xenotransplantation 21(4):309–323. https://doi.org/10.1111/xen.12102
Patience C, Takeuchi Y, Weiss RA (1997) Infection of human cells by an endogenous retrovirus of pigs. Nat Med 3(3):282–286. https://doi.org/10.1038/nm0397-282
Moalic Y, Blanchard Y, Felix H, Jestin A (2006) Porcine endogenous retrovirus integration sites in the human genome: features in common with those of murine leukemia virus. J Virol 80(22):10980–10988. https://doi.org/10.1128/jvi.00904-06
Wilson CA, Wong S, VanBrocklin M, Federspiel MJ (2000) Extended analysis of the in vitro tropism of porcine endogenous retrovirus. J Virol 74(1):49–56. https://doi.org/10.1128/jvi.74.1.49-56.2000
Denner J, Specke V, Thiesen U, Karlas A, Kurth R (2003) Genetic alterations of the long terminal repeat of an ecotropic porcine endogenous retrovirus during passage in human cells. Virology 314(1):125–133. https://doi.org/10.1016/s0042-6822(03)00428-8
Yang L, Guell M, Niu D, George H, Lesha E, Grishin D, Aach J, Shrock E, Xu W, Poci J, Cortazio R, Wilkinson RA, Fishman JA, Church G (2015) Genome-wide inactivation of porcine endogenous retroviruses (PERVs). Science (New York, NY) 350(6264):1101–1104. https://doi.org/10.1126/science.aad1191
Ross MJ, Coates PT (2018) Using CRISPR to inactivate endogenous retroviruses in pigs: an important step toward safe xenotransplantation? Kidney Int 93(1):4–6. https://doi.org/10.1016/j.kint.2017.11.004
Denner J (2017) Advances in organ transplant from pigs. Science (New York, NY) 357(6357):1238–1239. https://doi.org/10.1126/science.aao6334
Kosicki M, Tomberg K, Bradley A (2018) Repair of double-strand breaks induced by CRISPR-Cas9 leads to large deletions and complex rearrangements. Nat Biotechnol 36(8):765–771. https://doi.org/10.1038/nbt.4192
Denner J (2017) Paving the path toward porcine organs for transplantation. N Engl J Med 377(19):1891–1893. https://doi.org/10.1056/NEJMcibr1710853
Waechter A, Denner J (2014) Novel neutralising antibodies targeting the N-terminal helical region of the transmembrane envelope protein p15E of the porcine endogenous retrovirus (PERV). Immunol Res 58(1):9–19. https://doi.org/10.1007/s12026-013-8430-y
Denner J (2017) Can antiretroviral drugs be used to treat porcine endogenous retrovirus (PERV) infection after xenotransplantation? Viruses 9(8). https://doi.org/10.3390/v9080213
Niu D, Ma X, Yuan T, Niu Y, Xu Y, Sun Z, Ping Y, Li W, Zhang J, Wang T, Church GM (2020) Porcine genome engineering for xenotransplantation. Adv Drug Deliv Rev. https://doi.org/10.1016/j.addr.2020.04.001
Liu Y, Qin L, Tong R, Liu T, Ling C, Lei T, Zhang D, Wang Y, Deng S (2020) Regulatory changes in China on xenotransplantation and related products. Xenotransplantation:e12601. doi:https://doi.org/10.1111/xen.12601
Kwon I, Park C-G, Lee S (2020) Regulatory aspects of xenotransplantation in Korea. Xenotransplantation:e12602. doi:https://doi.org/10.1111/xen.12602
Cooper DKC, Pierson RN, Hering BJ, Mohiuddin MM, Fishman JA, Denner J, Ahn C, Azimzadeh AM, Buhler LH, Cowan PJ, Hawthorne WJ, Kobayashi T, Sachs DH (2017) Regulation of clinical xenotransplantation-time for a reappraisal. Transplantation 101(8):1766–1769. https://doi.org/10.1097/TP.0000000000001683
Cooper DKC, Wijkstrom M, Hariharan S, Chan JL, Singh A, Horvath K, Mohiuddin M, Cimeno A, Barth RN, LaMattina JC, Pierson RN (2017) Selection of patients for initial clinical trials of solid organ xenotransplantation. Transplantation 101(7):1551–1558. https://doi.org/10.1097/TP.0000000000001582
Lee A, Hudson AR, Shiwarski DJ, Tashman JW, Hinton TJ, Yerneni S, Bliley JM, Campbell PG, Feinberg AW (2019) 3D bioprinting of collagen to rebuild components of the human heart. Science (New York, NY) 365(6452):482–487. https://doi.org/10.1126/science.aav9051
Dasgupta Q, Black LD (2019) A fresh slate for 3D bioprinting. Science (New York, NY) 365(6452):446–447. https://doi.org/10.1126/science.aay0478
Dal Sasso E, Bagno A, Scuri STG, Gerosa G, Iop L (2019) The biocompatibility challenges in the total artificial heart evolution. Annu Rev Biomed Eng 21:85–110. https://doi.org/10.1146/annurev-bioeng-060418-052432
Murry CE, MacLellan WR (2020) Stem cells and the heart-the road ahead. Science (New York, NY) 367(6480):854–855. https://doi.org/10.1126/science.aaz3650
Davis DR (2019) Cardiac stem cells in the post-Anversa era. Eur Heart J 40(13):1039–1041. https://doi.org/10.1093/eurheartj/ehz098
Desgres M, Menasché P (2019) Clinical translation of pluripotent stem cell therapies: challenges and considerations. Cell Stem Cell 25(5):594–606. https://doi.org/10.1016/j.stem.2019.10.001
Mancini D, Goldstein D, Taylor S, Chen L, Gass A, DeLair S, Pinney S (2017) Maximizing donor allocation: a review of UNOS region 9 donor heart turn-downs. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 17(12):3193–3198. https://doi.org/10.1111/ajt.14499
Que W, Hu X, Fujino M, Terayama H, Sakabe K, Fukunishi N, Zhu P, Yi SQ, Yamada Y, Zhong L, Li XK (2019) Prolonged cold ischemia time in mouse heart transplantation using supercooling preservation. Transplantation. https://doi.org/10.1097/tp.0000000000003089
Pegg DE (2010) The relevance of ice crystal formation for the cryopreservation of tissues and organs. Cryobiology 60(3 Suppl):S36–S44. https://doi.org/10.1016/j.cryobiol.2010.02.003
de Vries RJ, Yarmush M, Uygun K (2019) Systems engineering the organ preservation process for transplantation. Curr Opin Biotechnol 58:192–201. https://doi.org/10.1016/j.copbio.2019.05.015
Ardehali A, Esmailian F, Deng M, Soltesz E, Hsich E, Naka Y, Mancini D, Camacho M, Zucker M, Leprince P, Padera R, Kobashigawa J (2015) Ex-vivo perfusion of donor hearts for human heart transplantation (PROCEED II): a prospective, open-label, multicentre, randomised non-inferiority trial. Lancet (London, England) 385(9987):2577–2584. https://doi.org/10.1016/S0140-6736(15)60261-6
Chan JL, Kobashigawa JA, Reich HJ, Ramzy D, Thottam MM, Yu Z, Aintablian TL, Liou F, Patel JK, Kittleson MM, Czer LS, Trento A, Esmailian F (2017) Intermediate outcomes with ex-vivo allograft perfusion for heart transplantation. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 36(3):258–263. https://doi.org/10.1016/j.healun.2016.08.015
Stamp NL, Shah A, Vincent V, Wright B, Wood C, Pavey W, Cokis C, Chih S, Dembo L, Larbalestier R (2015) Successful heart transplant after ten hours out-of-body time using the TransMedics organ care system. Heart, lung & circulation 24(6):611–613. https://doi.org/10.1016/j.hlc.2015.01.005
Dalle Ave AL, Shaw DM, Bernat JL (2016) Ethical issues in the use of extracorporeal membrane oxygenation in controlled donation after circulatory determination of death. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 16(8):2293–2299. https://doi.org/10.1111/ajt.13792
Funding
This research was supported by Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (2019-I2M-1-002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Funding information was added.
Rights and permissions
About this article
Cite this article
Shu, S., Ren, J. & Song, J. Cardiac xenotransplantation: a promising way to treat advanced heart failure. Heart Fail Rev 27, 71–91 (2022). https://doi.org/10.1007/s10741-020-09989-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-020-09989-x