Abstract
Understanding water stress signaling mechanisms and screening for tolerant cocoa cultivars are major challenges when facing prolonged dry and rainy seasons in cocoa-producing areas. While abscisic acid (ABA) and proline are supposed to enhance drought tolerance in cocoa, the role of polyamines remains unclear. The aim of this study was to investigate the biochemical response and phenological adaptation of cocoa (Theobroma cacao) on different soil moisture conditions, with a focus on short-term (20 days) and long-term (89 days) stress conditions, and to compare the performance of three cocoa cultivars. In a split plot design with four blocks, cocoa seedlings of an international high-yielding cultivar (TSH-565) and two locally selected cultivars (IIa-22 and III-06) from the drought-exposed Alto Beni region, Bolivia, were arranged in pots under a roof shelter (cultivar: three levels). The seedlings were exposed to strong (VERY DRY) and moderate (DRY) soil moisture deficits, water logging (WET) and regular irrigation (MOIST) that served as a control (moisture: four levels). We examined the growth performance and the levels of ABA, proline, and polyamines in the leaves. Growth was reduced already at a moderate drought, while severe drought enhanced seedling mortality. Severe drought increased the levels of ABA by 453% and of proline by 935%, inducing a short-term stress response; both compounds were degraded over the long-term period. The polyamine concentration was unrelated to soil moisture. The cocoa cultivars did not differ in their biochemical response to soil moisture stress (proline: p-value = 0.5, ABA: p-value = 0.3), but the local cultivar III-06 showed a stronger height growth increment than the international cultivar TSH-565 (237%, p-value = 0.002) under drought conditions.
Similar content being viewed by others
Introduction
Cocoa (Theobroma cacao L.) is produced in the humid tropics, where the trees are exposed to varying abiotic conditions. Climatic conditions are challenging cocoa cultivation: cocoa-producing regions worldwide are already affected by climate change and, consequently, by extreme weather events. Many regions are facing increasing temperatures, which, in turn, augment evapotranspiration and lead to changes in precipitation patterns (Läderach et al. 2013). The cocoa tree is sensitive to both water logging and drought (Wood and Lass 2001). Soils must have good drainage to avoid water logging, while rainfall should exceed 1250 mm (Zuidema et al. 2005) and be well distributed over the year. Prolonged and severe dry seasons affect growth and production (Läderach et al. 2013), and increase tree mortality (Gateau-Rey et al. 2018). Flooding is another environmental challenge for cocoa plantations, already common in lowlands and areas with heavy rainfall (Bertolde et al. 2010). The adaptation strategies of the cocoa production systems to reduce the impact of climate change have been widely discussed (Tscharntke et al. 2011; Vaast and Somarriba 2014), e.g., agroforestry systems that buffer climatic extremes and, therefore, protect the understory cocoa (Niether et al. 2018) and improve soil water infiltration (Tscharntke et al. 2011). Other efforts have focused on the identification and development of drought-tolerant cocoa cultivars (Apshara et al. 2013; Bae et al. 2008). Regional cultivation may contribute to adapting cocoa cultivars to local conditions (Araque et al. 2012; Ávila-Lovera et al. 2016), whereas international cultivars usually promise higher yields or pathogen resistance. Cocoa is an important cash crop. Especially small-scale farmers rely on the survival and productivity of their cocoa plantation for their family income (Hütz-Adams et al. 2016). This fact makes the adaptation of cultivars to local conditions and the design of resilient production systems important (Jacobi et al. 2015).
To define suitable breeding goals, the biochemical signaling of cocoa as a response to water stress in short- and long-term periods needs to be examined. Exposed to water stress, plants induce osmotic adjustment to maintain leaf turgor at low water potential values and protect cellular functioning (Almeida et al. 2002). The xylem water potential of cocoa leaves decreases in response to drought (Ávila-Lovera et al. 2016; Balasimha et al. 1991). Since maintenance of leaf turgor is an indicator of drought tolerance, the decrease indicates the drought susceptibility of cocoa in general, while more tolerant cocoa cultivars maintain a higher xylem water potential and reduce transpiration-mediated water loss by stomatal regulation (Almeida et al. 2016; Apshara et al. 2013; Araque et al. 2012). However, stomatal closure leads to a reduction in cocoa leaf mass and a decrease in starch concentration due to reduced photosynthetic activity (Almeida et al. 2016; Ávila-Lovera et al. 2016; Bae et al. 2009). Growth and development are consequently curtailed, especially in drought-sensitive cocoa cultivars (Santos et al. 2014). Maintaining high photosynthetic activity under dry conditions is also a desired trait for tolerance (Apshara et al. 2013). Flooding, as the opposite extreme to water deficit, causes hypoxia or anoxia in the soil, which inhibit water and nutrient uptake, and ultimately limit photosynthesis and reduce leave water potential in non-tolerant species like cocoa (Almeida et al. 2016; da Silva Branco et al. 2017).
Environmental stresses induce signaling cascades in plants that include messenger molecules and activation of gene transcription, resulting in a physiological response that depends on the severity of the stress and other environmental conditions (Tuteja and Sopory 2008). Drought activates the transcription of genes related to the biosynthesis of abscisic acid (ABA) (Santos et al. 2014). ABA is translocated within the plant and induces the ABA-dependent signal transduction pathway for stress response. In cocoa leaves, as in those of other plants, ABA stimulates osmotic adjustment, directly via stomatal closure (Balasimha 1983) or by activating gene expression in the biosynthetic pathways of other metabolites, e.g., the polyamines putrescine, spermidine and spermine (Bae et al. 2008). Polyamines are formed via ABA-dependent or independent pathways and are associated with drought responses (Alcázar et al. 2006; Bae et al. 2008). Particularly, accumulation of the diamine putrescine enhances tolerance to dehydration by controlling stomatal closure and reducing leave transpiration (Alet et al. 2011). ABA is further involved in the synthesis and accumulation of the amino acid proline during drought (Balasimha 1983). Proline fulfills several functions in cell protection, such as balancing the cells’ redox status and scavenging reactive oxygen species (reviewed in Verbruggen and Hermans 2008). The interaction of the different molecules involved in stress response is a recent research topic, and the complex connections are not yet completely understood (Pál et al. 2018). Both messenger molecules and osmotic adjustment differ in execution and intensity between species and cultivars, depending on stress tolerance and environmental conditions (Tuteja and Sopory 2008).
Dry seasons in the humid tropics, with little or no rain, can last for weeks or months. Short- and long-term strategies for adaptation to dry conditions are therefore crucial for cocoa trees to survive and maintain growth and development. Long-term stress experiments, especially involving cocoa seedlings and their leaf physiology, are scarce. However, they are necessary to understand the functioning of this crop under changing climatic conditions, and to support breeding goals. The aim of this study was to investigate the phenological adaptation and the physiological response of different cocoa cultivars to four soil moisture regimes, ranging from very dry to wet conditions, in an experimental set-up.
Due to the aforementioned sensibility of cocoa to drought and flooding (Wood and Lass 2001), we hypothesized (i) that the dry and wet conditions in our trial would reduce the growth of the seedlings, while moderate moist soil would be the best suitable condition for them, and (ii) that the plants would accumulate stress-related molecules of proline, ABA, and the polyamines putrescine, spermidine and spermine in the leaves in response to the soil moisture levels. We analyzed the biochemical response after a short-term (20 days) and a long-term (89 days) stress period under similar conditions to those of a prolonged dry season and a soil waterlogging period, expecting (iii) that the content of stress-related molecules in the leaves would remain high to maintain the functioning of the plant organs under the ongoing stress conditions. In this experimental set-up, we compared two locally selected cocoa cultivars and an international cultivar, all of them commonly used in the Alto Beni region, Bolivia, where the study was conducted. In this region, only 30% of the annual precipitation falls within the dry season. Trend analyses predict a rise in the mean temperature and a decrease in precipitation during the dry season (Niether et al. 2018), making the adaptation of crop cultivars to local conditions crucial. Finally, we assumed (iv) that the local selection of cultivars already seeks for adaptation to local climatic conditions, including water deficit and flooding, which is expressed by a higher survival rate and vegetative development of the local cultivars under stress.
Materials and methods
Plant material and experimental design
The study was conducted at Sara Ana, the experimental field station of the Research Institute of Organic Agriculture (Forschungsinstitut für Biologischen Landbau, FiBL) located in Alto Beni, Bolivia, a cocoa-producing region at the foothill of the Bolivian Andes (15°33’S, 67°19’W, 365 m a.s.l., 25.2 °C mean temperature, 83% mean relative humidity, 1439 mm annual precipitation, Niether et al. 2018). The experimental set-up with the cocoa seedlings was placed under a rainproof open-sided roof and a light shelter with 22% of photosynthetically active radiation reaching the seedlings, in accordance with previous experiments on cocoa seedlings (Almeida et al. 2016; da Silva Branco et al. 2017).
The cocoa seedlings used in this study were the popular internationally distributed cultivar TSH-565, from the Trinidad Selection Hybrid series, and the local cultivars IIa-22 and III-06, from an elite tree selection program by El Ceibo, the umbrella organization of organic cocoa producers’ cooperatives in Alto Beni, Bolivia. Producers in this region usually plant genetic material from both international and local cultivars, which makes it interesting to explore whether the selection of cultivars takes climatic suitability into consideration. The three cultivars of our study were selected according to their growth and production under field conditions (Schneider et al. 2017). Buds were taken from the same experimental field trial (Schneider et al. 2017) and grafted on four-month-old rootstocks of the cultivar IMC-67 growing in pots. To reduce water loss by evaporation, the soil was covered with rice husks. The seedlings were arranged in 3 × 4 treatment combinations of cultivars (three levels, main plot) and soil moisture (four levels, sub-plot) within a split-plot design with four blocks. One experimental unit (one treatment combination) consisted of four seedlings in individual pots to provide sufficient plant material for the repeated leaf sampling over the treatment period. This made up a total of 192 seedlings within 48 treatment combinations in four blocks.
Soil moisture and micro-environmental conditions during the experiment
The soil moisture was measured twice a week before the treatment started and in the course of the experiment, with a TDR-probe equipped with an H2-reader (IMKO, Germany). Initially, all plants were equally watered and kept at a high soil moisture level of 30% of volumetric water content (Table 1, see Online Resource Fig. 1). Three months after the grafting, the experiment started with the first sampling of leaves (day 0), followed by the soil moisture treatments. Over the time of the experiment, the plants were watered accordingly to maintain the soil moisture regimes. The MOIST condition was close to the initial condition (soil moisture was maintained at between 23 and 28% of volumetric water content); it decreased slightly during the first 20 days and was maintained at 27% over the long-term stress period. To obtain the DRY (soil moisture of 16–22% of volumetric water content) and VERY DRY (soil moisture of 10–15% of volumetric water content) conditions, the soil moisture was reduced in the first days until it reached the final water regime, and it was then maintained at 16% (DRY) and 13% (VERY DRY) during the long-term stress period. The temporal mean soil moisture during the first 20 days was higher than the final soil moisture, because it took some days to decrease the water content of the soil. The WET treatment was obtained with a 3 cm depth of stagnant water at the ground of the pot, simulating waterlogged soil with soil moisture of between 29 and 35% of volumetric water content. The soil moisture content was maintained at 33%, which was higher than the initial values before the onset of the water treatments.
Ambient climate, i.e. air temperature, relative humidity and radiation, varied over the course of the experiment during the rainy summer season. A data logger (Hobo Pro Series, Onset Computer Corporation, MA) recorded, hourly, the temperature and relative humidity at 1 m above the ground; extraterrestrial radiation was calculated from location data. Ambient temperature decreased slightly from a mean temperature of 26.8 °C, in the period before the onset of the experimental treatment, to a mean temperature of 25.7 °C during the long-term treatment period, while relative humidity rose from 81 to 88%. Radiation increased from October to December, but remained almost constant during the course of the water treatments from December to February.
Measurement of phenological adaptation and leaf sampling
Non-destructive measurements were conducted on day 40 and 89 of the experiment before leaf sampling. The plant development parameters measured included leaf number, stem diameter (at 5 cm below the inoculation site) and height (calculated as the sum of the length measured from the ground to the tip of one branch and the length of other branches measured from the furcation point to the tip of the branch). The growth increment from day 40 to day 89 was calculated for the leaf number, stem diameter and plant height to show the long-term development of the seedlings. The survival rate was determined from the initial number of seedlings and the number of dead seedlings at the end of the experiment.
Leaf sampling and determination of physiological stress response
Leaves were sampled in the morning of days 0, 20 and 89. Fully expanded leaves above the inoculation site were cut, immediately frozen in liquid nitrogen and transported to the Laboratorio de Calidad Ambiental (Laboratory of Environmental Quality) in La Paz, Bolivia, for freeze drying. Subsequently, they were sent to the University of Göttingen, Germany, where they were milled (ZM100, Retsch, Germany) and analyzed.
Proline
0.2 g of milled leaf samples were extracted in 3%-sulfosalicylic acid, as described by Bates et al. (1973). The reaction was started with ninhydrin (Carl Roth, Germany) in acetic acid and phosphoric acid. After one hour at 100 °C, the reaction was stopped on ice and toluol was added to separate the phases. The upper proline phase was immediately measured photometrically at 520 nm (8453 UV-Visible Spectrophotometer, Hewlett Packard, Germany). The proline concentration was calculated by the concentration of the standard reagent D-proline (Sigma-Aldrich, Germany) and is expressed in mg g− 1 (dry matter).
Abscisic acid (ABA)
Samples were extracted with a mixture of methanol, water and acetic acid (20:79:1) containing 25 ng ml1 of D6-(2Z,4E)-abscisic acid (Icon Services, USA) as an internal standard. ABA was re-extracted twice into diethyl ether, as described by Häffner et al. (2014): the solvent was removed under vacuum and the residue was dissolved in a mixture of methanol and water (1:1). ABA was detected by mass spectrometry (Sciex Qtrap 6500+) using LC-System Nexera X2 (Shimadzu, Germany), equipped with an RPcolumn (50 × 2 mm, Synergi Fusion, Phenomenx, Germany). ABA was eluted with a binary gradient containing 2 mmol ammonium acetate with 0.01% formic acid in bi-distilled water, as solvent A, and methanol, as solvent B. The gradient elution program (percentage of solvent A) was: 0 min, 25%; 4 min, 5%; 6 min, 5%; 6.01 min, 25%. The ABA content is expressed in µg g− 1 (dry matter).
Polyamines
Extraction and derivatization of polyamines from 0.1 g of milled cocoa leaves followed the method described by Smit et al. (2014), with the modification described by Niether et al. (2017). This included 10 µM of 1.7-diaminoheptan (Sigma-Aldrich, Germany) added to the extraction medium as an internal standard. Polyamines were analyzed via high-performance liquid chromatography (HPLC, LC-2000 Series, Jasco, Germany), as described by Smit et al. (2014), with HPLC-adjustments (Niether et al. 2017). The polyamines were eluted in the order of their retention times: internal standard diaminoheptan, putrescine, spermidine and spermine. The peaks were confirmed with the retention time of the reference compounds: spermine, spermidine (Sigma-Aldrich, Germany) and putrescine-dihydrochloride (Carl Roth, Germany). The limits of determination and quantification were defined according to Kromidas (2011). The concentrations of the different polyamines are expressed in µg g− 1 (dry matter).
Statistical analysis
We used linear mixed-effects models (lmerTest, Kuznetsova et al. 2016) within the statistical programming environment R (R Core Team 2018) to study the effects of the fixed factors “cultivar” (three levels) and “moisture” (soil moisture regime, four levels), and their interaction (moisture:cultivar), on the biochemical response variables. The block (replications, four levels) was always added as a random factor. In the first model, the fixed factor “stress period” (three levels: day 0 = before stress, day 20 = short-term stress, day 89 = long-term stress) was added, and in the second model the data were separated according to the sampling days. Linear mixed-effects models were also implemented to study the influence of cultivar, moisture and the moisture:cultivar interaction on the phenological variables. To determine differences in the survival rate, general mixed-effects models with family = binomial and the same factors as above were applied. In contrast to the other variables, we analyzed the survival of the four seedlings separately per treatment unit. Orthogonal contrasts were fixed in advance to compare the local cultivars to the international cultivar (IIa-22 vs. TSH-565, and TSH-565 vs. III-06), and the levels of the soil moisture regimes (VERY DRY vs. DRY, DRY vs. MOIST, and MOIST vs. WET). When necessary, outliers were removed and data were Box-Cox-transformed to meet the normality and homoscedasticity requirements. Data are usually presented as mean and standard error. The packages plyr (Wickham 2011) and ggplot2 (Wickham 2009) were used for data frame calculations and graphs.
Results
Phenological response to stress treatment
The best survival and plant growth of the cocoa seedlings across all cultivars was observed under the MOIST and WET soil moisture regimes; both survival and growth decreased from the MOIST to the DRY conditions. The differences between the DRY and VERY DRY conditions were not clearly pronounced for all parameters; leaf number and height increment were not different under those two regimes (Table 2). Height increment was 37.2 cm under the MOIST and 26.1 cm under the WET soil moisture regime, but only 15.0 and 17.8 cm under the DRY and VERY DRY treatments, respectively. Stem diameter and survival rate decreased further from DRY conditions, reaching their lowest values under the VERY DRY conditions. Leaf shedding and seedling mortality were observed on cocoa seedlings across all soil moisture conditions. While 79% of the cocoa seedlings withstood the MOIST conditions, the survival rate decreased to 71% and 69% under the DRY and WET conditions and even to 38% under the VERY DRY soil moisture conditions. 6% of the seedlings under WET conditions and 8% of the seedlings under MOIST conditions showed leaf shedding, but 29% of the seedlings of the DRY and 33% of the seedling of the VERY DRY soil moisture regimes lost leaves during the stress period (data not shown).
The cocoa cultivars varied in their stress adaptation to the different soil moisture regimes. While the local cultivars IIa-22 and III-06 showed their best stem diameter increment under the MOIST soil regime with 8.6 mm and 4.4 mm, respectively, TSH-565 had its highest increment (10.3 mm) under the WET conditions. The plant height increment of cultivar III-06 was 2.3-fold higher than that of TSH-565 across all soil moisture regimes. Additionally, the cultivars showed fluctuations in their soil moisture responses, e.g., the height increment under the VERY DRY conditions was higher than under DRY conditions for TSH-565 and III-06.
Biochemical response to soil moisture regimes
Proline
Before starting the experimental water treatment, i.e. when the seedlings were still under the same well-watered conditions, the concentration of proline in the leaves was on average 0.09 mg g− 1, ranging from 0.07 mg g− 1 in cultivar TSH-565 and 0.09 mg g− 1 in cultivar III-06 to 0.12 mg g− 1 in cultivar IIa-22 (Fig. 1; see also Online Resource Table 1). After the onset of the stress, the influence of the water treatments overlaid differences between cultivars. In the course of the experiment, the concentration of proline in cocoa leaves under the MOIST soil moisture conditions remained almost constant at 0.12 mg g− 1 in the short-term, and dropped to 0.09 mg g− 1 at the end of the three months of treatment (Table 3). After the short-term stress period, the concentration increased 1.5-fold to 0.13 mg g− 1 under the DRY conditions, and doubled to 0.19 mg g− 1 under the WET condition. After the long-term period, the concentration dropped again under both treatments, reaching values that were similar to those of the seedlings under the MOIST soil moisture regime (DRY: 0.09 mg g− 1, WET: 0.05 mg g− 1). The VERY DRY soil moisture conditions induced a 935% increase of the concentration of proline after the short-term soil moisture treatment, reaching 0.88 mg g− 1. The concentration decreased again to the initial one of 0.12 mg g− 1 after the long-term period, despite the ongoing moisture stress.
ABA
The concentration of ABA was 0.69 µg g− 1 in unstressed leaves before the onset of the soil moisture treatments, and increased 4.5-fold to 3.18 µg g− 1 when soil moisture was reduced during the short-term stress period in the VERY DRY treatment (Fig. 2; see also Online Resource Table 1). After the long-term stress period, the concentration of ABA decreased again, even dropping below the level measured before the onset of the stress (0.53 µg g− 1). Under the WET conditions, the concentration did not change after the short-term stress period, and only decreased slightly after the long-term stress period (0.58 µg g− 1). Differences in concentrations of ABA between cultivars in response to soil moisture regimes were not observed.
Polyamines
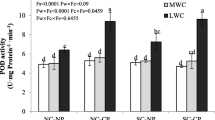
The influence of the soil moisture regimes (putrescine: p = 0.2; spermidine: p = 0.5; spermine: p = 0.6) and cultivars (putrescine: p = 0.2; spermidine: p = 0.6; spermine: p = 0.9) was imperceptible; instead the differences of the concentrations of all three polyamines between sampling periods overlaid all other effects irrespectively of the soil moisture treatment and the cultivars (p < 0.001 for all polyamines; Table 3). Before the onset of the stress treatment, putrescine was the most abundant polyamine (mean across all cultivars: 35.3 µg g− 1), followed by spermidine (18.3 µg g− 1) and spermine (8.1 µg g− 1; Fig. 3). The concentrations of putrescine and spermidine decreased across all soil moisture regimes and cultivars after the short-term and long-term periods. The concentration of spermine slightly increased to 150% of the initial value after the short-term stress period, and decreased again to 75% of that value after the long-term stress treatment. After both stress periods, spermine was the most abundant polyamine, followed by putrescine and spermidine.
After separating the dataset for statistical analysis into subsets for the various sampling periods, the soil moisture regime increased its effect on the level of polyamine (see Online Resource Table 2). At the end of the short-term stress period, the content of putrescine was 2-fold higher under the DRY (6.3 µg g− 1) and the VERY DRY (6.4 µg g− 1) regimes than under the MOIST (3.1 µg g− 1) soil moisture condition (p = 0.05). The content of spermine was slightly higher under the VERY DRY conditions than under the DRY ones (p = 0.05). After the long-term period, no differences in the concentrations of spermidine and putrescine were observed between soil moisture regimes, while the content of spermidine was lowest under the VERY DRY (2.2 µg g− 1) and DRY (2.0 µg g− 1) conditions and slightly higher under the MOIST (2.9 µg g− 1; p = 0.01) and the WET treatments (3.3 µg g− 1; p = 0.03). Before the onset of the stress, the content of putrescine in the leaves of TSH-565 (32.1 µg g− 1) was only 70% of the concentrations measured in IIa22 (45.7 µg g− 1; p = 0.02), but neither this difference nor any other differences between cultivars were observed in the other samplings.
Discussion
Biochemical response to short- and long-term moisture stress and phenological adaptation
Unfavorable soil moisture levels triggered biochemical responses in the leaves of cocoa seedlings. The concentrations of proline and ABA under the MOIST soil water regime over the short- and long-term stress periods were low, while the growth and survival of the cocoa seedlings were highest. This regime, with a high soil moisture content of 80% compared to the condition with stagnant water, was the most suitable one for the cocoa seedlings, and can therefore be referred to as the control condition (Ayegboyin and Akinrinde 2016; Santos et al. 2014).
When soil moisture decreased to 75% of the volumetric water content of the initial value (DRY) during the short-term period, the levels of proline in the leaves slightly increased. The biochemical response to soil water deficits was much stronger when soil moisture decreased to 62% of the initial value (VERY DRY) under the short-term treatment: proline accumulated in the leaves more than 9-fold and ABA more than 4-fold compared to the control condition. The effect of the soil moisture treatments DRY and VERY DRY was reflected very well by proline concentration even though the soil moisture increased on the sampling day. These results show that the proline levels established during the continuous treatment over the period of 20 days were not affected by spontaneous soil moisture changes. The parallel increases of the concentrations of ABA and proline after the short-term stress period can likely be accounted for the interaction of the biosynthetic pathways of both molecules during drought (Balasimha 1983). They were in line with a 9-fold increase of the levels of proline in cocoa seedlings seven days after withholding irrigation (Balasimha 1983), and an increase from day 3 to day 13 after stress induction (Bae et al. 2009). The accumulation of stress-induced molecules varies among species and plant organs and may reflect the ability of a plant to tolerate stressful conditions (Verbruggen and Hermans 2008). The concentration of proline in other plant species under osmotic stress increased 100-fold (Verbruggen and Hermans 2008), and the level of ABA in stressed cocoa embryos increased 25-fold as compared to the controls (Pence 1992). The relatively low accumulation rate of proline in cocoa seedlings compared to other plant species may be related to the susceptibility of cocoa to water deficit.
While the expression of genes associated with ABA biosynthesis was enhanced in tolerant cocoa cultivars when exposed to a 60-day-long drought treatment (Santos et al. 2014), the concentrations of proline and ABA in the leaves did not reach a high level, and then decreased again to or even below the level of unstressed cocoa seedlings. This may be caused by the degradation of accumulated ABA together with reduction of its synthesis, which is common when genes are repressed during a long-term treatment, as shown for tolerant cocoa cultivars (Santos et al. 2014). Another explanation of the decrease in the concentration of ABA may be conversion of ABA into ABA-glycosyl ester, as recently reported for Thellungiella salsuginea subjected to salt stress (Prerostova et al. 2017). The degradation of proline over a long-term treatment was described as a strategy to avoid proline toxicity in the leaves after proline accumulated to high concentrations over time (Verbruggen and Hermans 2008). Proline accumulation can be reduced when leaf turgor is maintained by ABA (Balasimha 1983). The reduction of both proline and ABA after a long-term water deficit may imply a different strategy aimed at cell protection and sustained cell functioning during the long-term water stress, or an impairment of the biosynthetic capacity of the plants because of the stress.
Under moderately dry conditions, the higher mortality and reduced vegetative growth of the seedlings, compared to those of the non-stressed seedlings, were accompanied by only a low increase in stress-related molecules, indicating that the damage of the plants was limited. Under strong water deficit, the damage of the plants resulted in a higher mortality than under moderate drought, which was comparable to observations of cocoa under severe stress made by other researchers (Alban et al. 2016; Ayegboyin and Akinrinde 2016). For its part, vegetative growth was not much reduced compared to the levels under moderate drought. This might be explained by the higher concentration of proline and ABA in the surviving seedlings under severe stress, which induced stomata closure resulting in limited photosynthesis and reduced growth (Ayegboyin and Akinrinde 2016). Some growth, at least at a low rate, has to be maintained even under very low moisture conditions to secure survival and sustain photosynthesis and a positive carbon balance over a long-term stress period (Joly and Hahn 1989). Although the water deficit triggered leaf shedding, which is an adaptational response reducing water loss by transpiration, the vegetative growth has not ceased (Table 2). Similar observations were reported for cocoa hybrids by Alban et al. (2015) and for drought-sensitive cocoa cultivars by Santos et al. (2014). Further strategies to reduce water loss reported in the literature include decrease of the leaf area (Ayegboyin and Akinrinde 2016) and reduction of the number of stomata per leaf area (Zanetti et al. 2016).
ABA accumulating in a rapid stress response is known to act as a growth inhibitor reducing cell functioning to a maintenance level. Proline, accumulated together with ABA in our experiments, is known to be synthesized by an ABA-dependent pathway and to help maintaining cell turgor at the onset of stress. Since the levels of stress molecules did not increase any further after the long-term stress period, the mechanism that sustained the survival of the cocoa seedlings over the whole stress period might have been induced already during the short-term stress. We hypothesize that, when the stress period continued, the reduction of vegetative growth that had already occurred as an adaptation to low moisture and high concentration of ABA may have been sufficient to sustain a maintenance level of plant functioning at continuously low water contents. In order to test this hypothesis, a study of gene expression (Santos et al. 2014) combined with an analysis of stress metabolites under long-term stress would be required.
Stagnant water over the long-term stress period simulated a flooded soil. No biochemical response to this situation was observed in the leaves, even though the reaction to water logging is regulated by ABA (Voesenek and Bailey-Serres 2015). The survival rate was slightly lower than under the control conditions (Table 2) and the vegetative growth, i.e. leaf and height increment, was reduced (Table 2), as previously shown by Bertolde et al. (2010) and da Silva Branco et al. (2017). In contrast, the stem diameter increment was higher under stagnant water than under the control conditions. This can also be the result of an adaptation to flooding, since stem diameter is positively correlated with the root biomass under flooding (Santos et al. 2018). Our contrasting findings regarding vegetative growth under drought and water logging support the observation by Almeida et al. (2016) that cocoa plants are more tolerant to flooding than to soil water deficit.
Polyamines in the water stress response of cocoa seedlings
In various plant species, drought induces an increase of polyamine concentrations (Alcázar et al. 2011). Also in cocoa, polyamines are supposed to play a role in response to soil moisture deficits (Bae et al. 2008), e.g., the concentration of spermine in cocoa beans decreases when the dry season prolongs, while those of putrescine and spermidine do not change (Niether et al. 2017). Bae et al. (2008) describe a drought-induced increase of the concentrations of all three polyamines after 13 days of treatment that was within the same range as polyamine concentrations after 20 days of treatment in the present study. But in contrast to the findings of Bae et al. (2008), the concentrations of putrescine and spermidine in this study were even higher before the treatment started (Fig. 3a, b). Furthermore, we did not observe change in the concentration of the three main polyamines in the leaves in response to the soil moisture regime. Instead, we found a general increase in the concentration of spermine at the beginning, as well as a decrease of the precursors of spermine, i.e. putrescine and spermidine, among the stressed and the control seedlings. These results can be explained by the conversion of putrescine to spermidine and subsequently to spermine, according to the biosynthetic pathway of polyamines in plants (Tuteja and Sopory 2008). These results do not support the role of polyamines in the drought tolerance of cocoa seedlings. We explain these findings by species-specific stress responses of cocoa leaves and the long duration of the stress period. Since foliar ABA and proline increased after 20 days of strong drought compared to non-stressed plants, but not after three months of ongoing stress, polyamines may have been involved in the stress response at a much earlier stage of the stress impact. This suggestion is inspired by the findings of Bae et al. (2008), who describe an increase in gene expression and polyamine levels during the first days after the stress started. Alcázar et al. (2011) propose homeostasis of polyamines during drought acclimation, which inhibits the accumulation of polyamines by constant synthesis, degradation, and back-conversion along the polyamine biosynthetic pathway. This might include a polyamine stress response that is not observed on the level of concentration, but rather on gene expression (Bae et al. 2008).
Since the variation of the polyamine levels was not explained by the soil moisture regimes, it is probably related to the general development of the plants in this case and might also been caused by other environmental effects during the treatment period, e.g., the decrease in the mean daily temperature and the simultaneous increase in relative humidity and extraterrestrial radiation (see Online Resource Fig. 1). These seasonal environmental changes were not strong, but we know that increasing radiation can affect the photosynthetic activity of young cocoa seedlings (Balasimha et al. 1991). Putrescine, among the other polyamines, has been shown to increase the efficiency of photosystem II (Ioannidis and Kotzabasis 2007), and a reduction of putrescine may come along with increasing light.
Conclusions
The mortality rate of cocoa seedlings increased with decreasing water availability, and vegetative growth was already reduced under moderate dry conditions, demonstrating the drought susceptibility of cocoa. Foliar ABA and proline accumulated as a response to short-term severe drought but the concentrations of both metabolites dropped during long-term treatments, implying an alternative mechanism of drought adaptation, such as the reduction of vegetative growth. Polyamine levels were controlled by a different environmental or plant-internal trigger over time that could not be identified. According to the biochemical data, we cannot recommend one cultivar over the others as better adapted to dry conditions. However, phenological performance favored local cultivars. The ability of different rootstock cultivars to take up water under dry conditions deserves further investigation.
References
Alban MKA, Apshara SE, Hebbar KB, Mathias TG, Séverin A (2015) Potential of antioxidant enzymes in depicting drought tolernace in cocoa (Theobroma cacao L.) genotypes at young age. Afr J Sci Res 4:18–23
Alban MKA, Apshara SE, Hebbar KB, Mathias TG, Séverin A (2016) Morpho-physiological criteria for assessment of two month old cocoa (Theobroma cacao L.) genotypes for drought tolerance. Ind J Plant Physiol 21:23–30. doi:https://doi.org/10.1007/s40502-015-0195-y
Alcázar R, Cuevas JC, Patron M, Altabella T, Tiburcio AF (2006) Abscisic acid modulates polyamine metabolism under water stress in Arabidopsis thaliana. Physiol Plant 128:448–455. doi:https://doi.org/10.1111/j.1399-3054.2006.00780.x
Alcázar R, Bitrián M, Bartels D, Koncz C, Altabella T, Tiburcio AF (2011) Polyamine metabolic canalization in response to drought stress in Arabidopsis and the resurrection plant Craterostigma plantagineum.. Plant Signal Behav 6:243–250. doi:https://doi.org/10.4161/psb.6.2.14317
Alet AI, Sanchez DH, Cuevas JC, del Valle S, Altabella T, Tiburcio AF, Marco F, Ferrando A, Espasandín FD, González ME, Ruiz OA, Carrasco P (2011) Putrescine accumulation in Arabidopsis thaliana transgenic lines enhances tolerance to dehydration and freezing stress. Plant Signal Behav 6:278–286. doi:https://doi.org/10.4161/psb.6.2.14702
Almeida J de, Tezara W, Herrera A (2016) Physiological responses to drought and experimental water deficit and waterlogging of four clones of cacao (Theobroma cacao L.) selected for cultivation in Venezuela. Agric Water Manag 171:80–88. doi:https://doi.org/10.1016/j.agwat.2016.03.012
Almeida AFd, Brito RCT, Aguilar MAG, Valle RR (2002) Water relation’s aspects of Theobroma cacao L. clones. Agrotrópica 14:35–44
Apshara SE, Rajesh MK, Balasimha D (2013) Assessment of morphological, physiological and molecular characteristics of cocoa accessions from Central and South America in relation to drought tolerance. J Plantation Crops 41:389–397
Araque O, Jaimez RE, Tezara W, Coronel I, Urich R, Espinoza W (2012) Comparative photosynthesis water relations growth and survival rates in juvenile criollo cacao cultivars Theobroma cacao during dry and wet seasons. Expl Agric 48:513–522. doi:https://doi.org/10.1017/S0014479712000427
Ávila-Lovera E, Coronel I, Jaimez R, Urich R, Pereya G, Araque O, Chacón I, Tezara W (2016) Ecophysiological traits of adult trees of criollo cocoa cultivars (Theobroma cacao L.) from a germplasm bank in Venezuela. Expl Agric 52:137–153. doi:https://doi.org/10.1017/S0014479714000593
Ayegboyin KO, Akinrinde EA (2016) Effect of water deficit imposed during the early developmental phase on photosynthesis of cocoa (Theobroma cacao L.). Agri Sci 07:11–19. doi:https://doi.org/10.4236/as.2016.71002
Bae H, Kim S, Kim MS, Sicher RC, Lary D, Strem MD, Natarajan S, Bailey BA (2008) The drought response of Theobroma cacao (cacao) and the regulation of genes involved in polyamine biosynthesis by drought and other stresses. Plant Physiol Biochem 46:174–188. doi:https://doi.org/10.1016/j.plaphy.2007.10.014
Bae H, Sicher RC, Kim MS, Kim S, Strem MD, Melnick RL, Bailey BA (2009) The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J Exp Bot 60:3279–3295. doi:https://doi.org/10.1093/jxb/erp165
Balasimha D (1983) Effect of abscisic acid and kinetin on growth and proline accumulation in cacao seedlings under water stress. Indian J Plant Physiol 26:139–142
Balasimha D, Daniel EV, Bhat PG (1991) Influence of environmental factors on photosynthesis in cocoa trees. Agric For Meteorol 55:15–21
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Bertolde FZ, Almeida AFd, Corrêa RX, Gomes FP, Gaiotto FA, Baligar VC, Lopes Loguerco L (2010) Molecular, physiological and morphological analysis of waterlogging tolerance in clonal genotypes of Theobroma cacao L. Tree Phys 30:56–67. doi:https://doi.org/10.1093/treephys/tpp101
da Silva Branco MC, Almeida AFd, Dalmolin ÂC, Ahnert D, Baligar VC (2017) Influence of low light intensity and soil flooding on cacao physiology. Sci Hortic 217:243–257. doi:https://doi.org/10.1016/j.scienta.2017.01.038
Gateau-Rey L, Tanner EVJ, Rapidel B, Marelli J, Royaert S (2018) Climate change could threaten cocoa production: Effects of 2015-16 El Niño-related drought on cocoa agroforests in Bahia, Brazil. PLoS ONE 13:e0200454. https://doi.org/10.1371/journal.pone.0200454
Häffner E, Karlovsky P, Splivallo R, Traczewska A, Diederichsen E (2014) ERECTA, salicylic acid, abscisic acid, and jasmonic acid modulate quantitative disease resistance of Arabidopsis thaliana to Verticillium longisporum. BMC Plant Biol 14:85. doi:https://doi.org/10.1186/1471-2229-14-85
Ioannidis NE, Kotzabasis K (2007) Effects of polyamines on the functionality of photosynthetic membrane in vivo and in vitro. Biochim Biophys Acta 1767:1372–1382. doi:https://doi.org/10.1016/j.bbabio.2007.10.002
Jacobi J, Schneider M, Bottazzi P, Pillco M, Calizaya P, Rist S (2015) Agroecosystem resilience and farmers’ perceptions of climate change impacts on cocoa farms in Alto Beni, Bolivia. Renew Agric Food Syst 30:170–183. doi:https://doi.org/10.1017/S174217051300029X
Joly RJ, Hahn DT (1989) Net C02 assimilation of cacao seedlings during periods of plant water deficit. Photosynth. Res. 21:151–159
Kromidas S (2011) Validierung in der Analytik, 2., nbsp;überarb. Aufl. Wiley-VCH, Weinheim
Kuznetsova A, Brockhoff PB, Bojesen Christensen RH (2016) lmerTest: tests in linear mixed effects models, Vienna, Austria. https://CRAN.R-project.org/package=lmerTest
Läderach P, Martinez-Valle A, Schroth G, Castro N (2013) Predicting the future climatic suitability for cocoa farming of the world’s leading producer countries, Ghana and Côte d’Ivoire. Clim Chang 119:841–854. https://doi.org/10.1007/s10584-013-0774-8
Marco F, Alcázar R, Tiburcio AF, Carrasco P (2011) Interactions between polyamines and abiotic stress pathway responses unraveled by transcriptome analysis of polyamine overproducers. OMICS 15:775–781. https://doi.org/10.1089/omi.2011.0084
Niether W, Smit I, Armengot L, Schneider M, Gerold G, Pawelzik E (2017) Environmental growing conditions in five production systems induce stress response and affect chemical composition of cocoa (Theobroma cacao L.) beans. J Agr Food Chem 65:10165–10173. https://doi.org/10.1021/acs.jafc.7b04490
Niether W, Armengot L, Andres C, Schneider M, Gerold G (2018) Shade trees and tree pruning alter throughfall and microclimate in cocoa (Theobroma cacao L.) production systems. Ann Forest Sci 75:e0149949. doi:https://doi.org/10.1007/s13595-018-0723-9
Pál M, Tajti J, Szalai G, Peeva V, Végh B, Janda T (2018) Interaction of polyamines, abscisic acid and proline under osmotic stress in the leaves of wheat plants. Sci Rep 8:12839. doi:https://doi.org/10.1038/s41598-018-31297-6
Pence VC (1992) Abscisic acid and the maturation of cacao embryos in vitro. Plant Physiol 98:1391–1395
Prerostova S, Dobreva PI, Gaudinova A, Hosek P, Soudek P, Knirsch V, Vankova R (2017) Hormonal dynamics during salt stress responses of salt-sensitive Arabidopsis thaliana and salt-tolerant Thellungiella salsuginea. Plant Sci 134:188–199. doi:https://doi.org/10.1016/j.plantsci.2017.07.020
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Santos EAD Almeida AFD, Silva Branco MCD, Santos ICD, Ahnert D, Baligar VC, Valle RR (2018) Path analysis of phenotypic traits in young cacao plants under drought conditions. PLoS ONE 13:e0191847. https://doi.org/10.1371/journal.pone.0191847
Santos ICD, Almeida AFD, Anhert D, Conceição ASd, Pirovani CP, Pires JL, Valle RR, Baligar VC (2014) Molecular, physiological and biochemical responses of Theobroma cacao L. genotypes to soil water deficit. PLoS ONE 9:e115746. https://doi.org/10.1371/journal.pone.0115746
Schneider M, Andres C, Trujillo G, Alcon F, Amurrio P, Perez E, Weibel F, Milz J (2017) Cocoa and total system yields of organic and conventional agroforestry vs. monoculture systems in a long-term field trial in Bolivia. Ex Agric 53:351–374. doi:https://doi.org/10.1017/S0014479716000417
Smit I, Pfliehinger M, Binner A, Grossmann M, Horst WJ, Lohnertz O (2014) Nitrogen fertilisation increases biogenic amines and amino acid concentrations in Vitis vinifera var. Riesling musts and wines. J Sci Food Agric 94:2064–2072. doi:https://doi.org/10.1002/jsfa.6525
Tscharntke T, Clough Y, Bhagwat SA, Buchori D, Faust H, Hertel D, Hölscher D, Juhrbandt J, Kessler M, Perfecto I, Scherber C, Schroth G, Veldkamp E, Wanger TC (2011) Multifunctional shade-tree management in tropical agroforestry landscapes - a review. J Appl Ecol 48:619–629. doi:https://doi.org/10.1111/j.1365-2664.2010.01939.x
Tuteja N, Sopory SK (2008) Chemical signaling under abiotic stress environment in plants. Plant Signal Behav 3:525–536
Vaast P, Somarriba E (2014) Trade-offs between crop intensification and ecosystem services: the role of agroforestry in cocoa cultivation. Agroforest Syst 88:947–956. https://doi.org/10.1007/s10457-014-9762-x
Verbruggen N, Hermans C (2008) Proline accumulation in plants: a review. Amino Acids 35:753–759. doi:https://doi.org/10.1007/s00726-008-0061-6
Voesenek LACJ, Bailey-Serres J (2015) Flood adaptive traits and processes: an overview. New Phytol 206:57–73. https://doi.org/10.1111/nph.13209
Wickham H (2009) ggplot2: Elegant graphics for data analysis. Use R. Springer, New York
Wickham H (2011) The split-apply-combine strategy for data analysis. J Stat Softw 40:1–29
Wood GAR, Lass RA (2001) Cocoa, 4th edn. Tropical agriculture series. Blackwell Science, Oxford
Zanetti LV, Milanez CRD, Novo Gama V, Galeas Aguilar MA, Spaggiari Souza CA, Campostrini E, MassiFerraz T, Mazzei Moura de Assis Figueiredo FA (2016) Leaf application of silicon in young cacao plants subjected to water deficit. Pesq Agropec Bras 51:215–223. https://doi.org/10.1590/S0100-204X2016000300003
Zuidema PA, Leffelaar PA, Gerritsma W, Mommer L, Anten NP (2005) A physiological production model for cocoa (Theobroma cacao): model presentation, validation and application. Agric Syst 84:195–225. https://doi.org/10.1016/j.agsy.2004.06.015
Acknowledgements
Open Access funding provided by Projekt DEAL. Many thanks are due to the Ecotop-team in Sara Ana, Bolivia, for technical and logistical support; to Jaime Chincheros Paniagua and the Laboratorio de Calidad Ambiental in La Paz, Bolivia, for sample preparation; and to Tobias Licha and his team at the Department of Applied Geology, University of Göttingen, Germany, for sample analysis. The study was funded by a grant from the Johannes-Hübner-Stiftung, Giessen, Germany.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niether, W., Glawe, A., Pfohl, K. et al. The effect of short-term vs. long-term soil moisture stress on the physiological response of three cocoa (Theobroma cacao L.) cultivars. Plant Growth Regul 92, 295–306 (2020). https://doi.org/10.1007/s10725-020-00638-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00638-9