Abstract
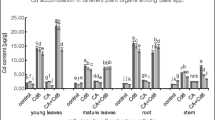
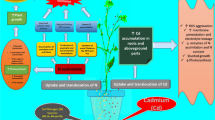
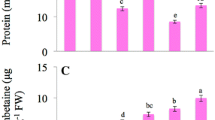
In a pot-soil culture ameliorative effect of sulphur (S) (0 or 40 mg S kg−1 soil) on cadmium (Cd) (0, 25, 50 and 100 mg Cd kg−1 soil)-induced growth inhibition and oxidative stress in mustard (Brassica campestris L.) cultivar Pusa Gold was studied. Cadmium at 100 mg kg−1 soil caused maximum increase in the contents of Cd and thiobarbituric acid reactive substances (TBARS) in leaves. Maximum reductions in growth (plant dry mass, leaf area), chlorophyll content, net photosynthetic rate (PN) and the contents of ascorbate (AsA), glutathione (GSH) were observed with 100 mg Cd kg−1 soil compared to control. The application of S helped in reducing Cd toxicity, which was greater for 25 and 50 mg Cd kg−1 soil) compared to 100 mg Cd kg−1 soil. Addition of S to Cd-treated plants showed decrease in Cd and TBARS content in leaves and restoration of growth and photosynthesis through increase in the contents of AsA and GSH. Net photosynthetic rate and plant dry mass were strongly and positively correlated with the contents of AsA and GSH. It is suggested that S may ameliorate Cd toxicity and protects growth and photosynthesis of mustard involving AsA and GSH.



Similar content being viewed by others
Abbreviations
- AsA:
-
Ascorbate
- Cd:
-
Cadmium
- Chl:
-
Chlorophyll
- DW:
-
Dry weight
- DAS:
-
Days after sowing
- DTNB:
-
5′,5′-Dithiobis-2-nitrobenzoic acid
- DTT:
-
1,4-dithiothreitol
- EDTA:
-
Ethylene diamine tetraacetate
- FW:
-
Fresh weight
- GR:
-
Glutathione reductase
- GSH:
-
Glutathione
- NPT:
-
Non-protein thiol
- PN :
-
Net photosynthetic rate
- PCs:
-
Phytochelatins
- ROS:
-
Reactive oxygen species
- TBARS:
-
Thiobarbituric acid reactive substances
References
Ahmad A, Khan I, Anjum NA, Diva I, Abrol YP, Iqbal M (2005) Effect of timing of sulphur fertilizer application on growth and yield of rapeseed (Brassica campestris L.). J Plant Nutr 28:1049–1059
Anderson ME (1985) Determination of glutathione and glutathione disulfides in biological samples. Meth Enzymol 113:548–570
Anjum NA, Umar S, Ahmad A, Iqbal M (2007a) Responses of components of antioxidant system in moongbean [Vigna radiata (L.) Wilczek] genotypes to cadmium stress. Commun Soil Sci Plant Anal (in press)
Anjum NA, Umar S, Iqbal M, Khan NA (2007b) Growth stage variation of the response to cadmium toxicity in Brassica campestris L. J Plant Interact. doi:1080/17429140171823164
Anjum NA, Umar S, Singh S, Nazar R, Khan NA (2008) Sulphur assimilation and cadmium tolerance in plants. In: Khan NA, Singh S, Umar S (eds) Abiotic stress and sulphur assimilation in plants. Springer-Verlag, Berlin, pp 271–302
Arvind P, Prasad MNV (2005) Cadmium and zinc interactions in a hydroponic system using Ceratophylum demersum L.: adaptive ecophysiology, biochemistry and molecular toxicology. Braz J Plant Physiol 17:3–20
Asada K (1999) The water–water cycle in chloroplasts: scavenging of active oxygen and dissipation of excess photons. Annu Rev Plant Physiol Plant Mol Biol 50:601–639
Chaoui A, Mazhoudi S, Ghorbal MH, El-Ferjani E (1997) Cadmium and zinc induction of lipid: lipid peroxidation and antioxidant enzyme activities in bean (Phaseolus vulgaris L.). Plant Sci 127:139–147
Chen Z, Gallie DR (2004) The ascorbic acid redox state controls guard cell signaling and stomatal movement. Plant Cell 16:1143–1162
Chien HF, Lin CC, Wang JW, Chen CT, Kao CH (2002) Changes in ammonium ion content and glutamine synthetase activity in rice leaves caused by excess cadmium are a consequence of oxidative damage. Plant Growth Regul 36:41–47
Cobbett CS (2000a) Phytochelatins and their roles in heavy metal detoxification. Plant Physiol 123:825–832
Cobbett CS (2000b) Phytochelatin biosynthesis and function in heavy metal detoxification. Curr Opin Plant Biol 3:211–216
Dhindsa RH, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence correlated with increased level of membrane permeability, lipid per oxidation and decreased level of SOD and CAT. J Exp Bot 32:93–101
Dixit V, Pandey V, Shyam R (2001) Differential oxidative responses to cadmium in roots and leaves of pea (Pisum sativum L. cv. Azad). J Exp Bot 52:1101–1109
Douglas KT (1987) Mechanism of action of glutathione-dependent enzymes. In: Meister A (ed) Advances in enzymology. John-Wiley, New York, pp 103–167
Ernst WHO (1998) Sulphur metabolism in higher plants: potential for phytoremediation. Biodegradation 9:311–318
Foyer CH (1993) Ascorbic acid. In: Alscher RG, Hess JL (eds) Antioxidants in higher plants. CRC Press, Boca Raton, pp 31–58
Foyer CH, Halliwell B (1976) The presence of glutathione and glutathione reductase in chloroplasts: a proposed role in ascorbic acid metabolism. Planta 133:21–25
Foyer CH, Lopez-Delgado H, Dat JF, Scott IM (1997) Hydrogen peroxide- and glutathione-associated mechanisms of acclimatory stress tolerance and signaling. Physiol Plant 100:241–254
Freedman JH, Ciriolo MR, Peisach J (1989) The role of glutathione in copper metabolism and toxicity. J Biol Chem 264:5598–5605
Gallego SM, Benavides MP, Tomaro ML (1996) Effect of heavy metal ion excess on sunflower leaves: evidence for involvement of oxidative stress. Plant Sci 121:151–159
Gong H, Zhu X, Chen K, Wang S, Zhang C (2005) Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci 169:313–321
Grill E, Winnacker E-L, Zenk MH (1985) Phytochelatins: the principal heavy metal complexing peptides of higher plants. Science 230:674–676
Hall JL (2002) Cellular mechanisms for heavy metal detoxification and tolerance. J Exp Bot 53:1–11
Hancock RD, Viola R (2005) Biosynthesis and catabolism of L-ascorbic acid in plants. Crit Rev Plant Sci 24:167–188
Hassan JM, Wang Z, Zhang G (2005) Sulphur alleviates growth inhibition and oxidative stress caused by cadmium toxicity in rice. J Plant Nutr 28:1785–1800
Hiscox JH, Israelstam GF (1979) A method for extraction of chlorophyll from leaf tissues without maceration. Can J Bot 57:1332–1334
Hsu YT, Kao CH (2004) Cadmium toxicity is reduced by nitric oxide in rice leaves. Plant Growth Regul 42:227–238
Inouhe M (2005) Phytochelatins. Braz J Plant Physiol 17:65–78
Kang YJ, Enger MD (1987) Effect of cellular glutathione depletion on cadmium-induced cytotoxicity in human lung carcinoma cells. Cell Biol Toxicol 3:347–360
Khan NA, Samiullah, Singh S, Nazar R (2007) Activities of antioxidative enzymes, sulphur assimilation, photosynthetic activity and growth of wheat (Triticum aestivum) cultivars differing in yield potential under cadmium stress. J Agron Crop Sci 193:435–444
Kunert KJ, Foyer CH (1993) Thiol/disulphide exchange in plants. In: De Kok LJ (ed) Sulphur nutrition and assimilation in higher plants. SPB Academic Publishing. The Hague, The Netherlands, pp 139–151
Law ME, Charles SA, Halliwell B (1983) Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts: the effect of hydrogen peroxide and of paraquat. Biochem J 210:899–903
May MJ, Vernoux T, Leaver C, Van-Montagu M, Inze D (1998a) Glutathione homeostasis in plants: implications for environmental sensing and plant development. J Exp Bot 49:649–667
May MJ, Vernoux T, Sanchez-Fernandez R, Van Montagu M, Inze D (1998b) Evidence for posttranscriptional activation of γ-glutamylcysteine synthetase during plant stress responses. Proc Natl Acad Sci USA 95:12049–12054
McLaughlin MJ, Parker DR, Clarke JM (1999) Metals and micronutrients: food safety issues. Field Crops Res 60:143–163
Metwally A, Safronova VI, Belimov AA, Dietz KJ (2005) Genotypic variation of the response to cadmium toxicity in Pisum sativum L. J Exp Bot 56:167–178
Mishra S, Srivastava S, Tripathy RD, Govindaranjan R, Kuriakose SV, Prasad MNV (2006) Phytochelatin synthesis and response of antioxidants during cadmium stress in Bacopa monieri L. Plant Physiol Biochem 44:25–37
Mobin M, Khan NA (2007) Photosynthetic activity, pigment composition and antioxidative response of two mustard (Brassica juncea) cultivars differing in photosynthetic capacity subjected to cadmium stress. J Plant Physiol 164:601–610
Nikiforova V, Freitag J, Kempa S, Adamik M, Hesse H, Hoefgen R (2003) Transcriptome analysis of sulphur depletion in Arabidopsis thaliana: interlacing of biosynthetic pathways provides response specificity. Plant J 33:633–650
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Noctor G, Gomez LA, Vanacker H, Foyer CH (2002) Interactions between biosynthesis, comparmentation and transport in the control of glutathione homeostasis and signaling. J Exp Bot 53:1283–1304
Ozturk L, Eker S, Ozkutlu F, Cakmak I (2003) Effect of cadmium on growth and concentrations of cadmium, ascorbic acid and sulfhydril groups in durum wheat cultivars. Turk J Agric Forest 27:161–168
Qadir S, Qureshi MI, Javed S, Abdin MZ (2004) Genotypic variation in phytoremediation potential of Brassica juncea cultivars exposed to Cd stress. Plant Sci 167:1171–1181
Rausch T, Wachter A (2005) Sulphur metabolism: a versatile platform for launching defence operations. Trends Plant Sci 10:503–509
Rauser WE (1995) Phytochelatins and related peptides: structure, biosynthesis and function. Plant Physiol 109:1141–1149
Saito K, Kurosawa M, Tatsuguchi K, Takagi Y, Murakoshi I (1994) Modulation of cysteine biosynthesis in the chloroplasts of transgenic tobacco overexpressing cysteine synthses [O-acetylserine(thiol)lyase]. Plant Physiol 106:887–895
Sharma SS, Kaul S, Metwally A, Goyal KC, Finkemeier I, Deitz KJ (2004) Cadmium toxicity to barley (Hordeum vulgare) as affected by varying Fe nutritional status. Plant Sci 166:1287–1295
Singh S, Anjum NA, Khan NA, Nazar R (2008) Metal-binding peptides and antioxidant defence system in plants: significance in cadmium tolerance. In: Khan NA, Singh S (eds) Abiotic stress and plant responses. IK International, New Delhi, pp 159–189
Singhal RK, Anderson ME, Meister A (1987) Glutathione, a first line defence against cadmium toxicity. FASEB J 1:220–223
Thangavel P, Long S, Minocha R (2007) Changes in phytochelatins and their biosynthetic intermediates in red spruce (Picea rubens Sarg.) cell suspension culture under cadmium and zinc stress. Plant Cell Tissue Organ Cult 88:201–216
Wagner G (1993) Accumulation of cadmium in crop plants and its consequences to human health. Adv Agron 51:173–211
Wu FB, Zhang GP (2002) Alleviation of cadmium-toxicity by application of zinc and ascorbic acid in barley. J Plant Nutr 25:2745–2761
Xiang C, Werner BL, Christensen EM, Oliver DJ (2001) The biological functions of glutathione revisited in Arabidopsis transgenic plants with altered glutathione levels. Plant Physiol 126:564–574
Zhang GP, Motohiro F, Hitoshi S (2002) Influence of cadmium on mineral concentrations and yield components in wheat genotypes differing in Cd tolerance at seedling stage. Field Crops Res 77:93–99
Zhao FJ, Evans EJ, Bilsborrow PE, Syers JK (1993) Influence of S and N on seed yield and quality of low glucosinolate oilseed rape (Brassica napus L.). J Sci Food Agric 63:29–37
Acknowledgements
Authors are indebted to Dr. R. K. Katiyar, Director, National Research Centre on Plant Biotechnology (NRCPB) of the Indian Agricultural Research Institute (IARI), New Delhi, India for providing authentic seeds of mustard for the study. The first author is obliged to Hamdard National Foundation, New Delhi, India for financial assistance in the form of Research Fellowship (DSW/HNF-18/2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anjum, N.A., Umar, S., Ahmad, A. et al. Sulphur protects mustard (Brassica campestris L.) from cadmium toxicity by improving leaf ascorbate and glutathione. Plant Growth Regul 54, 271–279 (2008). https://doi.org/10.1007/s10725-007-9251-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-007-9251-6