Abstract
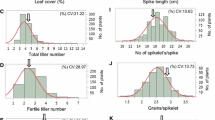
The domestication syndrome of wheat was based on genetically-mediated mutation(s) in reproductive traits with no due consideration to traits that comprise the plant “phenome” as an abstract expression of phenotypic architecture and a central component of the plant phenotype. The first of multi-part study reports on genetically and phenotypically variable germplasm source of diploid einkorn (Triticum monococcum L. subsp. monococcum) and tetraploid emmer wheat [Triticum turgidum subsp. dicoccon (Schrank) Thell.]. The germplasm was phenotyped for architectural (plant, tillers, leaves) and reproductive (spike, spikelet, and kernel) components of its phenome to quantify, describe, and contrast plant architecture of both species, and estimate the level of genetic divergence of emmer from einkorn due to polyploidy. Inter-specific variation for all architectural and reproductive components exhibited significant differences; while, intra-specific phenotypic variation estimates in both species decreased as follows: kernels > spikelets > spikes; and smaller values were estimated for leaves > plants > tillers. Diploid and tetraploid architectural modules, expressed as loadings on principal components, were significantly different; einkorn and emmer were 97.1 and 89.8 correctly classified, respectively, and were significantly separated at a multivariate level (Mahalanobis D2 = 127.5; p < 0.001). More traits exhibited larger magnitude and expressed larger genotypic variation due to polyploidy; however, phenotypic and genotypic variation, as well as heritability of these traits displayed different patterns, with large differences found for leaf area index and tillers per plant between species. Reduced major axis analysis of inter-specific functional relationships suggested that phenotypic and reproductive traits were divided almost equally between allometric and isometric patterns. Paired comparisons between species suggested that relationships between spikelet and kernel traits in each species were close (r = 0.54–0.97; p < 0.05). However, the magnitude of trait estimates in pairwise comparisons between ploidy levels were not in unison; the magnitude of almost all spikelet traits, but not kernel traits in emmer exceeded their counterparts in einkorn. Polyploidy caused phenotypic increases in traits that can support larger grain yield, including stems, peduncles, leaves and spikes, but not tillers; whereas, the species varied in the magnitude and distribution of genetic variances across their multivariate phenome space. Indicators were identified for the development of novel hulled wheat idiotypes with larger yield potential and wide adaptation.



Similar content being viewed by others
Abbreviations
- CAI:
-
Compact (or horizontal) architecture index
- Do :
-
Fractal dimension
- ECa:
-
Apparent soil electrical conductivity
- FLA:
-
Flag leaf area
- FL SLW:
-
Flag leaf specific leaf weight
- FT:
-
Number of fertile tillers per plant
- GCV:
-
Genotypic coefficient of variation
- GDD:
-
Growing degree days
- GLM:
-
General linear model
- G(Sp):
-
Genotypes within species
- h 2 :
-
Heritability estimate (narrow sense)
- LAI:
-
Leaf Area Index
- MATR:
-
Major axis tests and routines
- MSL:
-
Main stem length
- OLR:
-
Ordinary linear regression
- PCV:
-
Phenotypic coefficient of variation
- PL:
-
Plant length
- PLA:
-
Penultimate leaf area
- PL SLW:
-
Specific leaf weight of penultimate leaf
- PSPD:
-
Percent significant pairwise mean comparison differences
- PTQ:
-
Photothermal quotient
- RMA:
-
Reduced major axis
- SLW:
-
Specific leaf weight (dry weight per unit leaf area)
- T(Sp):
-
Traits within species
- TTP:
-
Total number of tillers per plant
- VAI:
-
Vertical architecture index
- Y:
-
Year
References
Baret F, de Solan B, Lopez-Lazano R, Ma K, Weiss M (2010) GAI estimates of row crops from downward looking digital photos taken perpendicular to rows at 57.5◦ zenith angle: theoretical considerations based on 3D architecture models and application to wheat crops. Agric For Meteorol 150:1393–1401
Barthélémy D, Caraglio Y (2007) Plant architecture: a dynamic, multilevel and comprehensive approach to plant form, structure and ontogeny. Ann Bot 99:375–407
Chateil C, Goldringer I, Tarall L, Kerbiriou C, Le Viol I, Ponge J-F, Salmon S, Gachet S, Porcher E (2013) Crop genetic diversity benefits farmland biodiversity in cultivated fields. Agric Ecosyst Environ 171:25–32
Chen D, Neumann K, Friedel S, Kilian B, Chen M, Altmann T, Klukas C (2014) Dissecting the phenotypic components of crop plant growth and drought responses based on high-throughput image analysis. Plant Cell 26:4636–4655. www.plantcell.org/cgi/doi/. https://doi.org/10.1105/tpc.114.129601
Chomicki G, Coiro M, Renner S (2017) Evolution and ecology of plant architecture: integrating insights from the fossil record, extant morphology, developmental genetics and phylogenies. Ann Bot 120:855–891. https://doi.org/10.1093/aob/mcx113
Das Choudhury S, Bashyam S, Qiu Y, Samal A, Awada T (2018) Holistic and component plant phenotyping using temporal image sequence. Plant Methods 14:35. https://doi.org/10.1186/s13007-018-0303-x
De Vita P, Riefolo C, Codianni P, Cattivelli L, Fares C (2006) Agronomic and qualitative traits of T. turgidum ssp. dicoccum genotypes cultivated in Italy. Euphytica 150:195–205
Donald C (1968) The breeding of crop ideotypes. Euphytica 17:385–403
Duan T, Chapman S, Holland E, Rebetzke G, Guo Y, Zheng B (2016) Dynamic quantification of canopy structure to characterize early plant vigor in wheat genotypes. J Exp Bot 67:4523–4534. https://doi.org/10.1093/jxb/erw227
Dubcovsky J, Dvorak J (2007) Genome plasticity a key factor in the success of polyploid wheat under domestication. Science 316:1862. https://doi.org/10.1126/science.1143986
Feldman M, Kislev M (2007) Domestication of emmer wheat and evolution of free-threshing tetraploid wheat. Isr J Plant Sci 55:207–221
Ferrante A, Cartelle J, Savin R, Slafer G (2017) Yield determination, interplay between major components and yield stability in a traditional and a contemporary wheat across a wide range of environments. Field Crops Res 203:114–127
Giraldo P, Royo C, González M, Carrillo J, Ruiz M (2016) Genetic diversity and association mapping for agro-morphological and grain Quality traits of a structured collection of durum wheat landraces including subsp. durum, t urgidum and dicoccon. PLoS ONE 11:0166577. https://doi.org/10.1371/journal.pone.0166577
Gonzalez-Navarro O, Griffiths S, Molero G, Reynolds M, Slafer G (2016) Variation in developmental patterns among elite wheat lines and relationships with yield, yield components and spike fertility. Field Crops Res 196:294–304
Guttieri M, Baenziger P, Frels K, Carver B, Arnall B, Waters B (2015) Variation for grain mineral concentration in a diversity panel of current and historical Great Plains hard winter wheat germplasm. Crop Sci 55:1035–1052
Hammer K (1984) Das Domestikationssyndrom. Kulturpflanze 32:11–34
Hinterthuer A (2017) Can ancient grains find their way in modern agriculture? CSA News Magazine, pp 4–8. Available at: https://dl.sciencesocieties.org/publications/csa/articles/62/4/4
Iriondo J, Milla R, Volis S, Rubio de Casas R (2017) Reproductive traits and evolutionary divergence between Mediterranean crops and their wild relatives. Plant Biol. https://doi.org/10.1111/plb.12640
Jaradat A (2018) Statistical modeling of phenotypic plasticity under abiotic stress in Triticum durum L. and Triticum aestivum L. genotypes. Agronomy 8:139. https://doi.org/10.3390/agronomy8080139
Kissoudis C, van de Wiel C, Visser R, van der Linden G (2016) Future-proof crops: challenges and strategies for climate resilience improvement. Curr Opin Plant Biol 30:47–56
Konvalina P, Capouchova I, Stenho Z (2012) Agronomically important traits of emmer wheat. Plant Soil Environ 58:341–346
Krahmer J, Ganpudi A, Abbas A, Romanowski A, Halliday K (2018) Phytocrome, carbon sensing, metabolism, and plant growth plasticity. Plant Physiol 176:1039–1048
Kumar U, Lazaa M, Soulié J-C, Pascoa R, Mendeza K, Dingkuhn M (2017) Analysis and simulation of phenotypic plasticity for traits contributing to yield potential in twelve rice genotypes. Field Crops Res 202:94–107. https://doi.org/10.1016/j.fcr.2016.04.037/
Li P-F, Cheng Z-G, Ma B-L, Palta Kong H-Y, Mo F, Wang J-Y, Zhu Y, Lv G-C, Batool A, Bai X, Li F-M, Xiong C (2014) Dryland wheat domestication changed the development of aboveground architecture for a well-structured canopy. PLoS ONE 9:e95825. https://doi.org/10.1371/journal.pone.0095825
Li X, Wang X, Peng Y, Wei H, Zhu X, Chang S, Li M, Li T, Huang H (2017) Quantitative descriptions of rice plant architecture and their application. PLoS ONE 12(5):e0177669. https://doi.org/10.1371/journal.pone.0177669
Longin C, Würschum T (2016) Back to the future: tapping into ancient grains for food diversity. Trends Plant Sci 21:731–737. https://doi.org/10.1016/j.tplants.2016.05.005
Longin C, Ziegler J, Schweiggert R, Koehler P, Carle R, Würschum T (2015) Comparative study of hulled (einkorn, emmer, and spelt) and naked wheats (durum and bread wheat): agronomic performance and quality traits. Crop Sci 56:302–311. https://doi.org/10.2135/cropsci2015.04.0242
Marino S, Tognetti R, Alvino A (2009) Crop yield and grain quality of emmer populations grown in central Italy, as affected by nitrogen fertilizer. Eur J Agron 31:233–240
Mauro-Herrera M, Doust A (2016) Development and genetic control of plant architecture and biomass in the Panicoid grass, Setaria. PLoS ONE 11(3):e0151346. https://doi.org/10.1371/journal.pone.0151346
Messier J, Lechowicz M, McGill B, Violle C, Enquist B (2017) Interspecific integration of trait dimensions at local scales: the plant phenotype as an integrated network. J Ecol https://doi.org/10.1111/1365-2745.12755
Milla R, Osborne CP, Turcotte MM, Violle C (2015) Plant domestication through an ecological lens. Trends Ecol Evol 30:463–469. https://doi.org/10.1016/j.tree.2015.06.006
Mondini L, Grausgruber H, Pagnotta M (2014) Evaluation of European emmer wheat germplasm for agro-morphological, grain quality traits and molecular traits. Genet Resour Crop Evol 61:69–87. https://doi.org/10.1007/s10722-013-0016-y
Münzbergová Z, Skuhrovec J (2016) Contrasting effects of ploidy level on seed production in a diploid-tetraploid system. AoB Plants 9: plw077. https://doi.org/10.1093/aobpla/plw077
Niklas KJ, Hammond ST (2014) Assessing scaling relationships: uses, abuses, and alternatives. Int J Plant Sci 175:754–763. https://doi.org/10.1086/677238
Oliveira H, Jones H, Leigh F, Lister D, Jones M, Pena-Chocarro L (2011) Phylogeography of einkorn landraces in the Mediterranean basin and Central Europe: population structure and cultivation history. Archaeol Anthropol Sci 3:327–341. https://doi.org/10.1007/s12520-011-0076-x
Payne W (2014) Developments from analysis of variance through to generalized linear models and beyond. Ann Appl 164:11–17
Pigliucci M (2003) Phenotypic integration: studying the ecology and evolution of complex phenotypes. Ecology Lett 6:265–272
Poorter H, Niklas K, Reich P, Oleksyn J (2012) Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol 193:30–50. https://doi.org/10.1111/j.1469-8137.2011.03952.x
Preece C, Livarda A, Christin P-A, Wallace M, Martin G, Charles M, Jones G, Rees M, Osborne C (2017) How did the domestication of Fertile Crescent grain crops increase their yields? Funct Ecol 31:387–397. https://doi.org/10.1111/1365-2435.12760
Qin X-L, Niklas K, Qi L, Yiong Y-C, Li F-M (2012) The effects of domestication on the scaling of below- vs. aboveground biomass in four selected wheat (Triticum; Poaceae) genotypes. Am J Bot 99:1112–1117
Rahman M, Ahsan M, Gillani Z, Chen M (2017) Digital biomass accumulation using high-throughput plant phenotype data analysis. J Integr Bioinform 20170028. https://doi.org/10.1515/jib-2017-0028
Robert C, Garin G, Abichou M, Houles V, Pradal C, Fournier C (2018) Plant architecture and foliar senescence impact the race between wheat growth and Zymoseptoria tritici epidemics. Ann Bot 121:975–989. https://doi.org/10.1093/aob/mcx192
Rötter R, Tao F, Höhn J, Palosuo T (2015) Use of crop simulation modelling to aid ideotype design of future cereal cultivars. J Exp Bot 66:3463–3476. https://doi.org/10.1093/jxb/erv098
Roucou A, Violle C, Fort F, Roumet P, Ecarnot M, Vile D (2018) Shifts in plant functional strategies over the course of wheat domestication. J Appl Ecol 55:25–37. https://doi.org/10.1111/1365-2664.13029
SAS Institute Inc (2016) JMP® Pro Version 13.2.0. SAS Institute Inc., Cary
Schindlin J, Rueden C, Hiner M, Eliceiri K (2015) The ImageJ ecosystems: an open platform for biomedical image analysis. Mol Reprod Dev 82:518–529. https://doi.org/10.1002/mrd.22489
Semenov M, Stratonovitch P (2013) Designing high-yielding wheat ideotypes for a changing climate. Food Energy Secur 2:185–196. https://doi.org/10.1002/fes3.34
Soil Conservation Service (USDA-SCS) (1971) Soil survey stevens county, Minnesota. USDA SCS, Washington, DC
StatCorp (2017) Stata statistical software, release 15.1. StatCorp LLC, College Station
Teichmann T, Muhr M (2015) Shaping plant architecture. Front Plant Sci 6:233. https://doi.org/10.3389/fpls.2015.00233
Troccoli A, Codianni P (2005) Appropriate seeding rate of einkorn, emmer, and spelt grown under rainfed conditions in southern Italy. Eur J Agron 22:293–300
Unal G (2009) Some physical and nutritional properties of hulled wheats. Turkish J Agron 15:58–64
Walter G, Aguirre J, Blows M, Ortiz-Barrientos D (2017) Evolution of genetic variance during adaptive radiation. bioRxiv. http://dx.doi.org/10.1101/097642
Wang J, Turner N, Liu Y, Siddique K, Xiong Y (2017) Effects of drought stress on morphological, physiological and biochemical characteristics of wheat species differing in ploidy level. Funct Plant Biol 44:219–234. https://doi.org/10.1071/FP16082
Warton D, Duursma R, Falseter D, Taskinen S (2012) SMATR 3—an R package for estimation and inference about allometric lines. Methods Ecol Evol 3:257–259. https://doi.org/10.1111/j.2041-210X.2011.00153.x
Xie Q, Mayes S, Sparkes D (2015) Spelt as a genetic resource for yield component improvement in bread wheat. Crop Sci 55:2753–2765
Zaharieva M, Monneveux P (2014) Cultivated einkorn wheat (Triticum monococcum L. subsp. monococcum): the long life of a founder crop of agriculture. Genet Resour Crop Evol https://doi.org/10.1007/s10722-014-0084-7
Zaharieva M, Ayana G, Al Hakimi A, Misra S, Monneveux P (2010) Cultivated emmer wheat (Triticum dicoccon Schrank), an old crop with promising future: a review. Genet Resour Crop Evol 57:937–962
Acknowledgements
This research was supported by USDA Project Number: 5060-21220-005-D. Thanks are due to support staff at the North Central Soil Conservation Research Lab, Morris, MN. USDA is an equal-opportunity provider and employer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jaradat, A.A. Comparative assessment of einkorn and emmer wheat phenomes: I. Plant architecture. Genet Resour Crop Evol 66, 491–512 (2019). https://doi.org/10.1007/s10722-018-0729-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-018-0729-z