Abstract
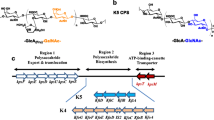
Escherichia coli strain K4 expresses a chondroitin (CH)-polymerizing enzyme (K4CP) that contains two glycosyltransferase active domains. K4CP alternately transfers glucuronic acid (GlcA) and N-acetyl-galactosamine (GalNAc) residues using UDP-GlcA and UDP-GalNAc donors to the nonreducing end of a CH chain acceptor. Here we generated two K4CP point mutants substituted at the UDP-sugar binding motif (DXD) in the glycosyltransferase active domains, which showed either glycosyltransferase activity of the intact domain and retained comparable activity after immobilization onto agarose beads. The mutant enzyme-immobilized beads exhibited an addition of GlcA or GalNAc to GalNAc or GlcA residue at the nonreducing end of CH oligosaccharides and sequentially elongated pyridylamine-conjugated CH (PA-CH) chain by the alternate use. The sequential elongation up to 16-mer was successfully achieved as assessed by fluorescent detection on a gel filtration chromatography and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) and MALDI potential lift tandem TOF mass spectrometry (MALDI-LIFT-TOF/TOF MS/MS) analyses in the negative reflection mode. This method provides exactly defined CH oligosaccharide derivatives, which are useful for studies on glycosaminoglycan functions.






Similar content being viewed by others
Abbreviations
- CH:
-
chondroitin
- CS:
-
chondroitin sulfate
- GalNAc:
-
N-acetyl-D-galactosamine
- GlcA:
-
D-glucuronic acid
- GalNAc-T:
-
GalNAc transferase
- GlcA-T:
-
GlcA transferase
- K4CP:
-
CH polymerase from Escherichia coli strain K4
- MALDI-TOF MS:
-
matrix-assisted laser desorption/ionization and time-of-flight mass spectrometry
- MALDI-LIFT-TOF/TOF MS/MS:
-
matrix-assisted laser desorption/ionization and potential lift tandem time-of-flight mass spectrometry
- m/z :
-
mass-per-charge ratio
- PA:
-
pyridylamine
References
Handel, T.M., Johnson, Z., Crown, S.E., Lau, E.K., Proudfoot, A.E.: Regulation of protein function by glycosaminoglycans—as exemplified by chemokines. Annu. Rev. Biochem. 74, 385–410 (2005)
Haltiwanger, R.S., Lowe, J.B.: Role of glycosylation in development. Annu. Rev. Biochem. 73, 491–537 (2004)
Mizuguchi, S., Uyama, T., Kitagawa, H., Nomura, K.H., Dejima, K., Gengyo-Ando, K., Mitani, S., Sugahara, K., Nomura, K.: Chondroitin proteoglycans are involved in cell division of caenorhabditis elegans. Nature 423, 443–8 (2003)
Gowda, D.C.: Role of chondroitin-4-sulfate in pregnancy-associated malaria. Adv. Pharmacol. 53, 375–400 (2006)
Watanabe, H., Yamada, Y., Kimata, K.: Roles of aggrecan, a large chondroitin sulfate proteoglycan, in cartilage structure and function. J. Biochem (Tokyo) 124, 687–93 (1998)
Ashikari-Hada, S., Habuchi, H., Kariya, Y., Itoh, N., Reddi, A.H., Kimata, K.: Characterization of growth factor-binding structures in heparin/heparan sulfate using an octasaccharide library. J. Biol. Chem. 279, 12346–54 (2004)
Stern, R., Asari, A.A., Sugahara, K.N.: Hyaluronan fragments: An information-rich system. Eur. J. Cell Biol. 85, 699–715 (2006)
Maeda, N., Fukazawa, N., Hata, T.: The binding of chondroitin sulfate to pleiotrophin/heparin-binding growth-associated molecule is regulated by chain length and oversulfated structures. J. Biol. Chem. 281, 4894–902 (2006)
Sotogaku, N., Tully, S.E., Gama, C.I., Higashi, H., Tanaka, M., Hsieh-Wilson, L.C., Nishi, A.: Activation of phospholipase c pathways by a synthetic chondroitin sulfate-E tetrasaccharide promotes neurite outgrowth of dopaminergic neurons. J. Neurochem. 103, 749–60 (2007)
Tully, S.E., Mabon, R., Gama, C.I., Tsai, S.M., Liu, X., Hsieh-Wilson, L.C.: A chondroitin sulfate small molecule that stimulates neuronal growth. J. Am. Chem. Soc. 126, 7736–7 (2004)
Tamura, J., Tokuyoshi, M.: Synthesis of chondroitin sulfate E hexasaccharide in the repeating region by an effective elongation strategy toward longer chondroitin oligosaccharide. Biosci. Biotechnol. Biochem. 68, 2436–43 (2004)
Lidholt, K., Fjelstad, M.: Biosynthesis of the Escherichia coli K4 capsule polysaccharide. A parallel system for studies of glycosyltransferases in chondroitin formation. J. Biol. Chem. 272, 2682–7 (1997)
Ninomiya, T., Sugiura, N., Tawada, A., Sugimoto, K., Watanabe, H., Kimata, K.: Molecular cloning and characterization of chondroitin polymerase from Escherichia coli strain K4. J. Biol. Chem. 277, 21567–75 (2002)
Jing, W., DeAngelis, P.L.: Analysis of the two active sites of the hyaluronan synthase and the chondroitin synthase of Pasteurella multocida. Glycobiology 13, 661–71 (2003)
DeAngelis, P.L., Oatman, L.C., Gay, D.F.: Rapid chemoenzymatic synthesis of monodisperse hyaluronan oligosaccharides with immobilized enzyme reactors. J. Biol. Chem. 278, 35199–203 (2003)
Sugiura, N., Shimokata, S., Watanabe, H., Kimata, K.: MS analysis of chondroitin polymerization: Effects of Mn2+ ions on the stability of UDP-sugars and chondroitin synthesis. Anal. Biochem. 365, 62–73 (2007)
Highsmith, S., Garvin Jr., J.H., Chipman, D.M.: Mechanism of action of bovine testicular hyaluronidase. mapping of the active site. J. Biol. Chem. 250, 7473–80 (1975)
Hayashi, S.: Study on the degradation of glycosaminoglycans by canine liver lysosomal enzymes. The contributions of hyaluronidase, β-glucuronidase, sulfatase, and β-N-acetylhexosaminidase in the case of chondroitin 4-sulfate. J. Biochem. (Tokyo) 83, 149–57 (1978)
Takagaki, K., Takeda, Y., Nakamura, T., Daidouji, K., Narita, H., Endo, M.: Analysis of glycosaminoglycans by high-performance liquid chromatography. J. Biochem. Biophys. Methods 28, 313–20 (1994)
Laemmli, U.K.: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–5 (1970)
Hase, S., Ibuki, T., Ikenaka, T.: Reexamination of the pyridylamination used for fluorescence labeling of oligosaccharides and its application to glycoproteins. J. Biochem. (Tokyo) 95, 197–203 (1984)
Tomiya, N., Takahashi, N.: Contribution of component monosaccharides to the coordinates of neutral and sialyl pyridylaminated N-glycans on a two-dimensional sugar map. Anal. Biochem. 264, 204–10 (1998)
Hirabayashi, J., Arata, Y., Kasai, K.: Reinforcement of frontal affinity chromatography for effective analysis of lectin-oligosaccharide interactions. J. Chromatogr. A 890, 261–71 (2000)
Okamoto, M., Takahashi, K., Doi, T., Takimoto, Y.: High-sensitivity detection and postsource decay of 2-aminopyridine-derivatized oligosaccharides with matrix-assisted laser desorption/ionization mass spectrometry. Anal. Chem. 69, 2919–26 (1997)
Iwafune, M., Kakizaki, I., Nakazawa, H., Nukatsuka, I., Endo, M., Takagaki, K.: A glycomic approach to proteoglycan with a two-dimensional polysaccharide chain map. Anal. Biochem. 325, 35–40 (2004)
Plaas, A.H., Hascall, V.C., Midura, R.J.: Ion exchange HPLC microanalysis of chondroitin sulfate: Quantitative derivatization of chondroitin lyase digestion products with 2-aminopyridine. Glycobiology 6, 823–9 (1996)
Minamisawa, T., Suzuki, K., Hirabayashi, J.: Systematic identification of N-acetylheparosan oligosaccharides by tandem mass spectrometric fragmentation. Rapid Commun. Mass Spectrom. 20, 267–74 (2006)
Domon, B., Costello, C.E.: A systematic nomenclature for carbohydrate fragmentations in FAB-MS/MS spectra of glycoconjugates. Glycoconj. J. 5, 397–409 (1988)
Nakata, H.: A rule to account for mass shifts in fragmentations of even-electron organic ions in mass specrometry. J. Mass Spectrom. Soc. Jpn. 50, 173–88 (2002)
Minamisawa, T., Suzuki, K., Maeda, H., Shimokata, S., Sugiura, N., Kimata, K., Hirabayashi, J.: Characterization of isomeric unsulfated glycosaminoglycan oligosaccharides by mass spectrometry/mass spectrometry. J. Mass Spectrom. Soc. Jpn. 55, 1–6 (2007)
Tawada, A., Masa, T., Oonuki, Y., Watanabe, A., Matsuzaki, Y., Asari, A.: Large-scale preparation, purification, and characterization of hyaluronan oligosaccharides from 4-mers to 52-mers. Glycobiology 12, 421–6 (2002)
Takagaki, K., Munakata, H., Majima, M., Kakizaki, I., Endo, M.: Chimeric glycosaminoglycan oligosaccharides synthesized by enzymatic reconstruction and their use in substrate specificity determination of streptococcus hyaluronidase. J. Biochem. (Tokyo) 127, 695–702 (2000)
Acknowledgements
We are grateful to Hiroshi Maeda and Yuniko Shibata, Seikagaku Corporation for kindly providing desulfated CH. We thank Minoru Fukayama, Aichi Medical University and Takashi Nirasawa and Nobuyuki Shimura, Bruker Daltonics for assistance in MALDI-TOF MS and MALDI-LIFT-TOF/TOF MS/MS analyses. This work was supported by a special research fund from Seikagaku Corporation by the New Energy and Industrial Technology Development Organization (NEDO) and by the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sugiura, N., Shimokata, S., Minamisawa, T. et al. Sequential synthesis of chondroitin oligosaccharides by immobilized chondroitin polymerase mutants. Glycoconj J 25, 521–530 (2008). https://doi.org/10.1007/s10719-008-9105-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-008-9105-0