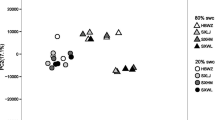
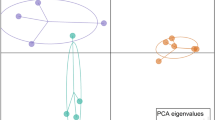
Abstract
Dehydrins are one of the major stress-induced gene families, and the expression of dehydrin 6 (Dhn6) is strictly related to drought in barley. In order to investigate how the evolution of the Dhn6 gene is associated with adaptation to environmental changes, we examined 48 genotypes of wild barley, Hordeum spontaneum, from “Evolution Canyon” at Mount Carmel, Israel. The Dhn6 sequences of the 48 genotypes were identified, and a recent insertion of 342 bp at 5′UTR was found in the sequences of 11 genotypes. Both nucleotide and haplotype diversity of single nucleotide polymorphism in Dhn6 coding regions were higher on the AS (“African” slope or dry slope) than on the ES (“European” slope or humid slope), and the applied Tajima D and Fu-Li test rejected neutrality of SNP diversity. Expression analysis indicated that the 342 bp insertion at 5′UTR was associated with the earlier up-regulation of Dhn6 after dehydration. The genetic divergence of amino acids sequences indicated significant positive selection of Dhn6 among the wild barley populations. The diversity of Dhn6 in microclimatic divergence slopes suggested that Dhn6 has been subjected to natural selection and adaptively associated with drought resistance of wild barley at “Evolution Canyon”.



Similar content being viewed by others
References
Allagulova CR, Gimalov FR, Shakirova FM, Vakhitov VA (2003) The plant dehydrins: structure and putative functions. Biochem (Moscow) 68:945–951
Bartels D, Souer E (2004) Molecular responses of higher plants to dehydration. In: Hirt H, Shinozaki K (eds) Plant responses to abiotic stress. Springer, Berlin, pp 9–38
Britten RJ (1996) DNA sequence insertion and evolutionary variation in gene regulation. Proc Natl Acad Sci USA 93:9374–9377
Campbell SA, Close TJ (1997) Dehydrins: genes, proteins and associations with phenotypic traits. New Phytol 137:61–74
Chenna R, Sugawara H, Koike T, Lopez R, Gibson TJ, Higgins DG, Thompson JD (2003) Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 31:3497–3500
Choi DW, Zhu B, Close TJ (1999) The barley (Hordeum vulgare L) dehydrin multigene family: sequences, allele types, chromosome assignments, and expression characteristics of 11 Dhn genes of cv. Dicktoo. Theor Appl Genet 98:1234–1247
Close TJ (1996) Dehydrins: emergence of a biochemical role of a family of plant dehydration proteins. Physiol Plant 97:795–803
Close TJ (1997) Dehydrins: a commonality in the response of plants to dehydration and low temperature. Physiol Plant 100:291–296
Close TJ, Choi DW, Venegas M, Salvi S, Tuberosa R, Ryabushkina N, Turuspekov Y, Nevo E (2000) Allelic variation in wild and cultivated barley at the Dhn4 locus, which encodes a major drought-induced and seed protein, DHN4. (Abstract). In: 8th international Barley genetics symposium, Adelaide, South Australia
Comeron JM (1999) K-estimator: calculation of the number of nucleotide substitutions per site and the confidence intervals. Bioinformatics 15:763–764
Cronin JK, Bundock PC, Henry RJ, Nevo E (2007) Adaptive climatic molecular evolution in wild barley at the Isa defense locus. Proc Natl Acad Sci USA 104:2773–2778
Fu YX, Li WH (1993) Statistical tests of neutrality of mutations. Genetics 133:693–709
Gupta PK, Sharma PK, Balyan HS, Roy JK, Sharma S, Beharav A, Nevo E (2002) Polymorphism at rDNA loci in barley and its relation with climatic variables. Theor Appl Genet 104:473–481
Heckathorn SA, Mueller JK, LaGuidice S, Zhu B, Barrett T, Blair B, Dong Y (2004) Chloroplast small heat-shock proteins protect photosynthesis during heavy metal stress. Am J Bot 91:1312–1318
Huang X, Lu G, Zhao Q, Liu X, Han B (2008) Genome-wide analysis of transposon insertion polymorphisms reveals intraspecific variation in cultivated rice. Plant Physiol 148:25–40
Hudson RR (2000) A new statistic for detecting genetic differentiation. Genetics 155:2011–2014
Hudson RR, Slatkin M, Maddison WP (1992) Estimation of levels of gene flow from DNA sequence data. Genetics 132:583–589
Ivandic V, Hackett CA, Zhang ZJ, Staub JE, Nevo E, Thomas WTB, Forster BP (2000) Phenotypic responses of wild barley to experimentally imposed water stress. J Exp Bot 51:2021–2029
Kawaura K, Mochida K, Enju A, Totoki Y, Toyoda A, Sakaki Y, Kai C, Kawai J, Hayashizaki Y, Seki M, Shinozaki K, Ogihara Y (2009) Assessment of adaptive evolution between wheat and rice as deduced from full-length common wheat cDNA sequence data and expression patterns. BMC Genomics 10:e271
Koag MC, Fenton RD, Wilkens S, Close TJ (2003) The binding of maize DHN1 to lipid vesicles. Gain structure and lipid specificity. Plant Physiol 131:309–316
Kosova K, Holkova L, Prasil IT, Prasilova P, Bradacova M, Vitamvas P, Capkova V (2008) Expression of dehydrin 5 during the development of frost tolerance in barley (Hordeum vulgare). J Plant Physiol 165:1142–1151
Kreitman M (2000) Methods to detect selection in populations with applications to the human. Annu Rev Genomics Hum Genet 1:539–559
Li YC, Krugman T, Fahima T, Beiles A, Nevo E (1998) Genetic diversity of alcohol dehydrogenase 3 in wild barley population at the "Evolution Canyon" microsite, Nahal Oren, Mt. Carmel, Israel. Barley Genet Newsl 28:58–60
Martin J, Geromanos S, Tempst P, Hartl FU (1993) Identification of nucleotide-binding regions in the chaperonin proteins GroEL and GroES. Nature 366:279–282
Morrell PL, Clegg MT (2007) Genetic evidence for a second domestication of barley (Hordeum vulgare) east of the Fertile Crescent. Proc Natl Acad Sci USA 104:3289–3294
Morrell PL, Lundy KE, Clegg MT (2003) Distinct geographic patterns of genetic diversity are maintained in wild barley (Hordeum vulgare ssp. spontaneum) despite migration. Proc Natl Acad Sci USA 100:10812–10817
Morrell PL, Toleno DM, Lundy KE, Clegg MT (2005) Low levels of linkage disequilibrium in wild barley (Hordeum vulgare ssp. spontaneum) despite high rates of self-fertilization. Proc Natl Acad Sci USA 102:2442–2447
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Nevo E (1995) Asian, African and European biota meet at ‘Evolution Canyon’, Israel: local tests of global biodiversity and genetic diversity patterns. P Roy Soc B-Biol Sci 262:149–155
Nevo E (1997) Evolution in action across phylogeny caused by microclimatic stresses at "Evolution Canyon". Theor Pop Biol 52:231–243
Nevo E (2001) Evolution of genome-phenome diversity under environmental stress. Proc Natl Acad Sci USA 98:6233–6240
Nevo E (2004) Population genetic structure of wild barley and wheat in the Near East Fertile Crescent: regional and local adaptive patterns. In: Gupta PK, Varshney RK (eds) Cereal genomics. Kluwer, Dordrecht, pp 135–163
Nevo E (2006) ‘Evolution Canyon’: a microcosm of life’s evolution. Isr J Ecol Evol 52:183–184
Nevo E (2009) Evolution in action across life at “Evolution Canyons”, Israel. Trends Evol Biol 1:1–3
Nevo E, Apelbaum-Elkaher I, Garty J, Beiles A (1997) Natural selection causes microscale allozyme diversity in wild barley and a lichen at ‘Evolution Canyon’ Mt Carmel, Israel. Heredity 78:373–382
Nevo E, Travleev A, Belova NA, Tsatskin A, Pavlicek T, Kulik AF, Tsvetkova NN, Yemshanov DC (1998) Edaphic interslope and valley bottom differences at "Evolution Canyon", Lower Nahal Oren, Mount Carmel, Israel. Catena 33:241–254
Nevo E, Fragman O, Dafni A, Beiles A (1999) Biodiversity and interslope divergence of vascular plants caused by microclimatic differences at ‘Evolution Canyon’, Lower Nahal Oren, Mount Carmel, Israel. Isr J Plant Sci 47:49–59
Nevo E, Beharav A, Meyer RC, Hackett CA, Forster BP, Russell JR, Powell W (2005) Genomic microsatellite adaptive divergence of wild barley by microclimatic stress in ‘Evolution Canyon’, Israel. Biol J Linn Soc 84:205–224
Owuor ED, Fahima T, Beiles A, Korol AB, Nevo E (1997) Population genetics response to microsite ecological stress in wild barley Hordeum spontaneum. Mol Ecol 6:1177–1187
Pavlicek T, Sharon D, Kravchenko V, Saaroni H, Nevo E (2003) Microclimatic interslope differences underlying biodiversity contrasts in ‘Evolution Canyon’, Mt Carmel, Israel. Isr J Earth Sci 52:1–9
Peleg O, Brunak S, Trifonov E, Nevo E, Bolshoy A (2002) RNA secondary structure and sequence conservation in C1 region of human immunodeficiency virus type 1 env gene. AIDS Res Hum Retrov 18:867–878
Qian G, Liu Y, Ao D, Yang F, Yu M (2008) Differential expression of dehydrin genes in hull-less barley (Hordeum vulgare ssp. vulgare) depending on duration of dehydration stress. Can J Plant Sci 88:899–906
Rodriguez EM, Svensson JT, Malatrasi M, Choi DW, Close TJ (2005) Barley Dhn13 encodes a KS-type dehydrin with constitutive and stress responsive expression. Theor Appl Genet 110:852–858
Rorat T (2006) Plant dehydrins. Tissue location, structure and function. Cell Mol Biol Lett 11:536–556
Rozas J, Sánchez-DelBarrio JC, Messeguer X, Rozas R (2003) DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 19:2496–2497
Shen Q, Uknes SJ, Ho TH (1993) Hormone response complex in a novel abscisic acid and cycloheximide-inducible barley gene. Biol Chem 268:23652–23660
Suprunova T, Krugman T, Fahima T, Chen G, Shams I, Korol A, Nevo E (2004) Differential expression of dehydrin genes in wild barley, Hordeum spontaneum, associated with resistance to water deficit. Plant Cell Environ 27:1297–1308
Svensson AS, Johansson FI, Moller IM, Rasmusson AG (2002) Cold stress decreases the capacity for respiratory NADH oxidation in potato leaves. FEBS Lett 517:79–82
Tajima F (1989) Statistical methods to test for nucleotide mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Tommasini L, Svensson JT, Rodriguez EM, Wahid A, Malatrasi M, Kato K, Wanamaker S, Resnik J, Close TJ (2008) Dehydrin gene expression provides an indicator of low temperature and drought stress: transcriptome-based analysis of Barley (Hordeum vulgare L.). Funct Integr Genomic 8:387–405
Werner-Fraczek JE, Close TJ (1998) Genetic studies of Triticeae dehydrins: assignment of seed proteins and a regulatory factor to map positions. Theor Appl Genet 97:220–226
Yang Z, Wong WSW, Nielsen R (2005) Bayes empirical Bayes inference of amino acid sites under positive selection. Mol Biol Evol 22:1107–1118
Yang ZJ, Li GR, Chang ZJ, Zhou JP, Ren ZL (2006) Characterization of a partial amphiploid between Triticum aestivum cv. Chinese Spring and Thinopyrum intermedium ssp. trichophorum. Euphytica 149:11–17
Yang Z, Zhang T, Bolshov A, Beharav A, Nevo E (2009) Adaptive microclimatic structural and expressional dehydrin 1 evolution in wild barley, Hordeum spontaneum, at ‘Evolution Canyon’, Mount Carmel, Israel. Mol Ecol 18:2063–2075
Zou C, Lehti-Shiu MD, Thomashow M, Shiu S (2009) Evolution of stress-regulated gene expression in duplicate genes of Arabidopsis thaliana. PLoS Genet 5:e1000581
Acknowledgments
This research was supported by National Natural Science Foundation of China (31171542), the Fundamental Research Funds for the Central Universities, grant NCET-06-0810 from the Chinese Ministry of Education, the Israel Discount Bank Chair of Evolutionary Biology, and the Ancell-Teicher Research Foundation for Molecular Genetics and Evolution. The authors thank Mrs. Robin Barasch-Permut for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10709_2012_9641_MOESM2_ESM.tif
Figure S1. “Evolution Canyon” I, Lower Nahal Oren, Mount Carmel, Israel. Note the plant formation on opposite slopes. The green lush, “European”, temperate, and cool-mesic north-facing slope (NFS) sharply contrasts the NFS with the open park forest of warm-xeric, tropical, and “African”-savannoid south-facing slope (SFS): (a) “cross section” view (b) air view with the seven assigned stations (TIFF 1987 kb)
10709_2012_9641_MOESM3_ESM.tif
Figure S2. DNA conservation along the Dhn6 alignment sequence of wild barley, Hordeum spontaneum. The conservation is measured by Kullback–Leibler information content (TIFF 130 kb)
10709_2012_9641_MOESM4_ESM.tif
Figure S3. Dendrogram of the genetic relationships of 48 genotypes of wild barley from seven populations representing the AS, ES, and VB in “Evolution Canyon” I, Israel. These genotypes were obtained from the Dhn6 alignment sequence, calculated nucleotide diversity (p-distance) values (see scale), using the NJ method from bootstrap analysis (1000 replicates). The stars showed sequences with 342bp insertion (TIFF 128 kb)
10709_2012_9641_MOESM5_ESM.tif
Figure S4. The amino acid sequences translated from Dhn6 N-terminal region and alignment by Clustal X. The locations of predicted Y-segments are boxed, and the repetitive φ-segments are underlined (TIFF 1148 kb)
Rights and permissions
About this article
Cite this article
Yang, Z., Zhang, T., Li, G. et al. Adaptive microclimatic evolution of the dehydrin 6 gene in wild barley at “Evolution Canyon”, Israel. Genetica 139, 1429–1438 (2011). https://doi.org/10.1007/s10709-012-9641-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-012-9641-1