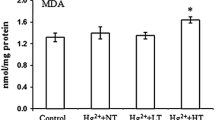
Abstract
Obscure puffer (Takifugu obscurus) is an anadromous fish widely distributed around the coastal and inland rivers in East Asia. T. obscurus often encounters fluctuations in temperature and salinity. This study aimed to investigate the effect of the interactions of temperature and salinity on survival and oxidative stress response of newly hatched T. obscurus larvae. A combination of three temperatures (19, 25, and 31 °C) and three salinities (0, 10, and 20 ppt) was applied for 96 h under laboratory conditions. The newly hatched larvae could not tolerate 31 °C for 96 h. No death was recorded at other temperatures during this experiment. Malondialdehyde concentrations increased significantly after 6 h of exposure to high salinity (10 and 20 ppt) and then decreased until the end of the experiment at each temperature. The highest superoxide dismutase activity was observed under the exposure to 20 ppt for 24 h at 31 °C. Na+/K+–ATPase activity significantly increased as salinity increased, especially at low temperatures. With the prolong of exposure time, the integrated biomarker response (IBR) values showed an increase until 48 h and then declined at 96 h in most treatments. The largest IBR value appeared when larvae were exposed to the highest temperature and salinity for 24 h. Our study indicated that high temperature with high salinity may negatively affect the early development of T. obscurus and their combined effects should be considered in the larvae culture.




Similar content being viewed by others
References
Abele D, Puntarulo S (2004) Formation of reactive species and induction of antioxidant defence systems in polar and temperate marine invertebrates and fish. Comp Biochem Physiol A Mol Integr Physiol 138:405–415
Akatsu S, Al-Abdul-Elah KM, Teng SK (2009) Effects of salinity and water temperature on the survival and growth of brown spotted grouper larvae (Epinephelus tauvina, Seranidae). J World Aquacult Soc 14:624–635
Amado LL, Monserrat JM (2010) Oxidative stress generation by microcystins in aquatic animals: why and how. Environ Int 36:226–235
Basha PS, Rani AU (2003) Cadmium-induced antioxidant defense mechanism in freshwater teleost Oreochromis mossambicus (tilapia). Ecotoxicol Environ Saf 56:218–221
Baylon JC (2010) Effects of salinity and temperature on survival and development of larvae and juveniles of the mud crab, Scylla serrata (Crustacea: Decapoda: Portunidae). J World Aquacult Soc 41:858–873
Beliaeff B, Burgeot T (2002) Integrated biomarker response: a useful tool for ecological risk assessment. Environ Toxicol Chem 21:1316–1322
Bœuf G, Payan P (2001) How should salinity influence fish growth? Comp Biochem Physiol C 130:411–423
Bushnell PG, Brill RW (1992) Oxygen transport and cardiovascular responses in skipjack tuna (Katsuwonus pelamis) and yellowfin tuna (Thunnus albacares) exposed to acute hypoxia. J Comp Physiol B 162:131–143
Bystriansky JS, Richards JG, Schulte PM, Ballantyne JS (2006) Reciprocal expression of gill Na+/K+-ATPase α-subunit isoforms α1a and α1b during seawater acclimation of three salmonid fishes that vary in their salinity tolerance. J Exp Biol 209:1848–1858
Cheng W, Chen JC (2000) Effects of pH, temperature and salinity on immune parameters of the freshwater prawn Macrobrachium rosenbergii. Fish Shellfish Immunol 10:387–391
Choi CY, An KW, An MI (2008) Molecular characterization and mRNA expression of glutathione peroxidase and glutathione S-transferase during osmotic stress in olive flounder (Paralichthys olivaceus). Comp Biochem Physiol A Mol Integr Physiol 149:330–337
Damiens G, Gnassia-Barelli M, Loquèo F, Roméo M, Salbert V (2007) Integrated biomarker response index as a useful tool for environmental assessment evaluated using transplanted mussels. Chemosphere 66:574–583
Doyotte A, Cossu C, Jacquin MC, Babut M, Vasseur P (1997) Antioxidant enzymes, glutathione and lipid peroxidation as relevant biomarkers of experimental or field exposure in the gills and the digestive gland of the freshwater bivalve Unio tumidus. Aquat Toxicol 39:93–110
Fielder DS, Bardsley WJ, Allan GL, Pankhurst PM (2005) The effects of salinity and temperature on growth and survival of Australian snapper, Pagrus auratus larvae. Aquaculture 250:201–214
Han KN, Yoshimatsu T (1997) Early developmental stages in laboratory-reared puffer fish, Takifugu rubripes. Sci Bull Fac Agr Kyushu Univ 51:157–165
Itai P (1999) Effects of salinity on swimming performance, routine activity and standard metabolic rate on the euryhaline killifish, Aphanius Dispar. Comp Biochem Physiol A Mol Integr Physiol 124:S82
Johnston IA, Vieira V, Temple GK (2001) Functional consequences population differences in the developmental plasticity of muscle to temperature in Atlantic herring Clupea harengus. Mar Ecol Prog Ser 213:285–300
Khatooni MM, Amiri BM, Hoseinifar SH, Jafari V, Makhdomi N (2011) Acclimation potential of Acipenser persicus post-larvae to abrupt or gradual increase in salinity. J Appl Ichthyol 27:528–532
Kim JH, Dahms HU, Rhee JS, Lee YM, Lee J, Han KN, Lee JS (2010a) Expression profiles of seven glutathione S-transferase (GST) genes in cadmium-exposed river pufferfish (Takifugu obscurus). Comp Biochem Physiol C 151:99–106
Kim JH, Rhee JS, Lee JS, Dahms HU, Lee J, Han KN, Lee JS (2010b) Effect of cadmium exposure on expression of antioxidant gene transcripts in the river pufferfish, Takifugu obscurus (Tetraodontiformes). Comp Biochem Physiol C 152:473–479
Kinne O (1970) Marine ecology: a comprehensive integrated treatise on life in oceans and coastal waters volume I: environmental factors. Wiley, London, pp 683–1244
Kong X, Wang G, Li S (2012) Effects of low temperature acclimation on antioxidant defenses and ATPase activities in the muscle of mud crab (Scylla paramamosain). Aquaculture 370:144–149
Kumlu M, Eroldogan OT, Aktas M (2000) Effects of temperature and salinity on larval growth, survival and development of Penaeus semisulcatus. Aquaculture 188:167–173
Li JJ, Wang J, Yang LJ, Chen YF, Yang Z (2014) Changes in plasma osmolality and Na/K ATPase activity of juvenile obscure puffer Takifugu obscurus following salinity challenge. Biochem Syst Ecol 56:111–117
Liu Y, Wang WN, Wang AL, Wang JM, Sun RY, Liu Y, Wang WN, Wang AL, Wang JM, Sun RY (2007) Effects of dietary vitamin E supplementation on antioxidant enzyme activities in Litopenaeus vannamei (Boone, 1931) exposed to acute salinity changes. Aquaculture 265:351–358
Lushchak VI (2010) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30
Lushchak VI, Bagnyukova TV (2006a) Temperature increase results in oxidative stress in goldfish tissues. 1. Indices of oxidative stress. Comp Biochem Physiol C 143:30–35
Lushchak VI, Bagnyukova TV (2006b) Temperature increase results in oxidative stress in goldfish tissues. 2. Antioxidant and associated enzymes. Comp Biochem Physiol C 143:36–41
Madeira D, Narciso L, Cabral HN, Vinagre C, Diniz MS (2013) Influence of temperature in thermal and oxidative stress responses in estuarine fish. Comp Biochem Physiol A Mol Integr Physiol 166:237–243
Martínez-Alvarez RM, Hidalgo MC, Domezain A, Morales AE, García-Gallego M, Sanz A (2003) Physiological changes of sturgeon Acipenser naccarii caused by increasing environmental salinity. J Exp Biol 205:3699–3706
Mobasheri A, Avila J, Cózar-Castellano I, Brownleader MD, Trevan M, Francis MJO, Lamb JF, Martín-Vasallo P (2000) Na+, K+-ATPase isozyme diversity; comparative biochemistry and physiological implications of novel functional interactions. Biosci Rep 20:51–91
Munari M, Matozzo V, Marin MG (2011) Combined effects of temperature and salinity on functional responses of haemocytes and survival in air of the clam Ruditapes philippinarum. Fish Shellfish Immunol 30:1024–1030
Nagar LR, Shenkar N (2016) Temperature and salinity sensitivity of the invasive ascidian Microcosmus exasperatus Heller, 1878. Aquat Invasions 11:33–43
Nurdiani R, Zeng C (2007) Effects of temperature and salinity on the survival and development of mud crab, Scylla serrata (Forsskål), larvae. Aquac Res 38:1529–1538
Pack EC, Lee SH, Kim CH, Lim CH, Sung DG, Kim MH, Park KH, Lim KM, Choi DW, Kim SW (2014) Effects of environmental temperature change on mercury absorption in aquatic organisms with respect to climate warming. J Toxicol Environ Health 77:1477–1490
Parihar MS, Dubey AK (1995) Lipid peroxidation and ascorbic acid status in respiratory organs of male and female freshwater catfish Heteropneustes fossilis exposed to temperature increase. Comp Biochem Physiol C 112:309–313
Patterson JT, Mims SD, Wright RA (2013) Effects of body mass and water temperature on routine metabolism of American paddlefish Polyodon spathula. J Fish Biol 82:1269–1280
Pirozzi I, Booth MA (2009) The effect of temperature and body weight on the routine metabolic rate and postprandial metabolic response in mulloway, Argyrosomus japonicus. Comp Biochem Physiol A Mol Integr Physiol 154:110–118
Rombough P (2007) The functional ontogeny of the teleost gill: which comes first, gas or ion exchange? Comp Biochem Physiol A Mol Integr Physiol 148:732–742
Sajadi H, Abraham J, Grundy MA, Gerhard GS (2004) The effects of temperature reduction on gene expression and oxidative stress in skeletal muscle from adult zebrafish. Comp Biochem Physiol C 138:363–373
Saoud IP, Kreydiyyeh S, Chalfoun A, Fakih M (2007) Influence of salinity on survival, growth, plasma osmolality and gill Na+–K+–ATPase activity in the rabbitfish Siganus rivulatus. J Exp Mar Biol Ecol 348:183–190
Shi YH, Zhang GY, Zhu YZ, Liu JZ (2010) Effects of photoperiod, temperature, and salinity on growth and survival of obscure puffer Takifugu obscurus larvae. Aquaculture 309:103–108
Shi YH, Zhang GY, Liu JZ, Zhu YZ, Jia-Bo XU, Zhu XD (2011) Effects of temperature and salinity on oxygen consumption of tawny puffer Takifugu flavidus juvenile. Aquac Res 42:301–307
Spanopoulos-Hernández M, Martínez-Palacios CA, Vanegas-Pérez RC, Rosas C, Ross LG (2005) The combined effects of salinity and temperature on the oxygen consumption of juvenile shrimps Litopenaeus stylirostris (Stimpson, 1874). Aquaculture 244:341–348
Sun HJ, Lü L, Minter EJA, Chen YF, Yang Z, Montagnes DJS (2012) Combined effects of ammonia and microcystin on survival, growth, antioxidant responses, and lipid peroxidation of bighead carp Hypophthalmythys nobilis larvae. J Hazard Mater 221:213–219
Tsangaris C, Hatzianestis I, Catsiki VA, Kormas KA, Strogyloudi E, Neofitou C, Andral B, Galgani F (2011) Active biomonitoring in Greek coastal waters: application of the integrated biomarker response index in relation to contaminant levels in caged mussels. Sci Total Environ 412:359–365
Venkatesan P (1981) Influence of temperature and salinity variations on an aquatic bug population in a tropical pond. Hydrobiologia 79:33–50
Vinagre C, Madeira D, Narciso L, Cabral HN, Diniz M (2012) Effect of temperature on oxidative stress in fish: lipid peroxidation and catalase activity in the muscle of juvenile seabass, Dicentrarchus labrax. Ecol Indic 23:274–279
Wang J, Li ZH, Chen YF, Yang Z (2015a) The combined effect of temperature and pH on embryonic development of obscure puffer Takifugu obscurus and its ecological implications. Biochem Syst Ecol 58:1–6
Wang J, Li JJ, Xu N, Li J, Li ZH, Chen YF, Yang Z (2015b) Responses of Takifugu obscurus fertilized eggs and larvae to increased ammonia exposure. Environ Sci Pollut Res 22:15976–15984
Wang J, Zhu XX, Huang X, Gu L, Chen YF, Yang Z (2016) Combined effects of cadmium and salinity on juvenile Takifugu obscurus: cadmium moderates salinity tolerance; salinity decreases the toxicity of cadmium. Sci Rep 6:30968
Wang J, Tang HX, Zhang XX, Xue XF, Zhu XX, Chen YF, Yang Z (2018) Mitigation of nitrite toxicity by increased salinity is associated with multiple physiological responses: a case study using an economically important model species, the juvenile obscure puffer (Takifugu obscurus). Environ Pollut 232:137–145
Yadwad VB, Kallapur VL, Basalingappa S (1990) Inhibition of gill Na+/K+-ATPase activity in dragonfly larva, Pantala flavesens, by endosulfan. Bull Environ Contam Toxicol 44:585–589
Yamanoue Y, Miya MK, Miyazawa S, Tsukamoto N, Doi H, Takahashi H, Mabuchi K, Nishida M, Sakai H (2009) Explosive speciation of Takifugu: another use of fugu as a model system for evolutionary biology. Mol Biol Evol 26:623–629
Yang Z, Chen YF (2003) Induced ovulation using LHRHa in anadromous obscure puffer Takifugu obscurus cultured entirely in freshwater. Fish Physiol Biochem 29:323–326
Yang Z, Chen YF (2004) Induced ovulation in obscure puffer Takifugu obscurus by injections of LHRH-a. Aquac Int 12:215–223
Yang Z, Chen YF (2005) Effect of temperature on incubation period and hatching success of obscure puffer Takifugu obscurus (Abe) eggs. Aquaculture 246:173–179
Yang Z, Chen YF (2006) Salinity tolerance of embryos of obscure puffer Takifugu obscurus. Aquaculture 253:393–397
Yang Z, Chen YF (2008) Differences in reproductive strategies between obscure puffer Takifugu obscurus and ocellated puffer Takifugu ocellatus during their spawning migration. J Appl Ichthyol 24:569–573
Yang Z, Yang JX (2004) Effect of photoperiod on the embryonic development of obscure puffer (Takifugu obscurus). J Freshw Ecol 19:53–58
Zydlewski J, Mccormick SD (1997) The ontogeny of salinity tolerance in the American shad, Alosa sapidissima. Can J Fish Aquat Sci 54:182–189
Acknowledgements
This investigation was funded by the National Natural Science Foundation of China (31370429) and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All experiments were approved by and carried out in accordance with the guidelines of the Institutional Animal Care and Use Committees (IACUC) of Nanjing Normal University, Nanjing, China (Research permit number: SYXK2015-0028).
Rights and permissions
About this article
Cite this article
Wang, J., Hou, X., Xue, X. et al. Interactive effects of temperature and salinity on the survival, oxidative stress, and Na+/K+–ATPase activity of newly hatched obscure puffer (Takifugu obscurus) larvae. Fish Physiol Biochem 45, 93–103 (2019). https://doi.org/10.1007/s10695-018-0537-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-018-0537-6