Abstract
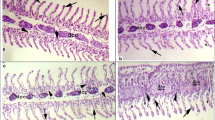
The effects of Roundup (glyphosate-based herbicide) on peripheral blood parameters and hematopoietic tissue cellular composition and activity in common carp juveniles were evaluated. The fish were exposed for 7 days at 0.0, 0.1, 0.5, or 5.0 mg/L of glyphosate. In fish exposed to Roundup, hematological alterations were observed; however, most of them were not directly related to the herbicide concentration. An increase in Ht and MCV, and decrease in Hb, MCH, and MCHC compared to the control were observed. Fish exposed to Roundup showed also a reduction in WBC and oxidative metabolic activity of phagocytes (NBT) compared to the control. The fish exposed to 0.1 and 5.0 mg/L showed increased glucose values, whereas in those subjected to 0.5 mg/L blood glucose concentration declined compared to the control. Cholesterol significantly increased at 0.1 mg/L and decreased at 5.0 mg/L. Analysis of head kidney hematopoietic tissue revealed that Roundup at concentrations 0.5 and 5.0 mg/L caused a significant increase in the rate of cell proliferation accompanied by an increase in frequency of early blast cells. No significant differences occurred in percentages of most cell lineages but the frequency of monocytoid, eosinophilic, and basophilic lineage cells significantly increased in the herbicide-exposed fish compared to the control. The obtained results revealed that sublethal concentrations of Roundup that may occur in polluted natural waters caused a slight anemic and significant immunosuppressive response in common carp juveniles. On the other hand, they indicate a considerable compensatory potential of carp hematopoietic system.
Similar content being viewed by others
References
Anton FA, Labora E, De Ariz M (1994) Acute toxicity of the herbicide glyphosate to fish. Chemosphere 82:745–753
Ayoola SO (2008) Histopathological effects of glyphosate on juvenile African catfish (Clarias gariepinus). JAES 4:362–367
Barton BA (2002) Stress in fishes: a diversity of responses with particular reference to changes in circulating corticosteroids. Integ Comp Biol 42:517–525
Benachour N, Séralini GE (1997) Glyphosate formulations induce apoptosis and necrosis in human umbilical, embryonic, and placental cells. Chem Res Toxicol 22:97–105
Binukumari S, Vasanthi J (2013) The toxic effect of pesticide dimethoate 30% EC on the protein metabolism of the fresh water fish, Labeorohita. IJCMAS 2:79
Brausch JM, Smith PN (2007) Toxicity of three polyethoxylated tallowamine surfactant formulations to laboratory and field collected fairy shrimp, Thamnocephalus platyurus. Arch Environ Contam Toxicol 52:217–221
Cavas T, Konen S (2007) Detection of cytogenetic and DNA damage in peripheral erythrocytes of goldfish (Carassius auratus) exposed to a glyphosate formulation using the micronucleus test and the comet assay. Mutagenesis 22:263–268
Fijan N (2002a) Morphogenesis of blood cell lineages in channel catfish. J Fish Biol 60:999–1014
Fijan N (2002b) Composition of main haematopoietic compartments in normal and bled channel catfish. J Fish Biol 60:1142–1154
Folmar LC, Sander HO, Julin AM (1979) Toxicity of the herbicide glyphosate and several of its formulations to fish and aquatic invertebrate. Arch Environ Contam Toxicol 8:269–278
Franz JE, Mao MK, Sikorski JA (1997) Glyphosate: a unique global herbicide. ACS 4:65–97
Gholami-Seyedkolaei SJ, Mirvaghefi A, Farahmand H, Kosari AA (2013) Effect ofaglyphosate-based herbicidein Cyprinus carpio: assessment of acetylcholinesterase activity, hematological responses and serum biochemical parameters. Ecotox Environ Safe 98:135–141
Giesy JP, Dobson S, Solomon KR (2000) Ecotoxicological risk assessment for Roundup herbicide. Rev Environ Contam Toxicol 167:35–120
Gimsing AL, Borggaard OK, Jacobsen OS, Aamand J, Sorensen J (2004) Chemical and microbiological soil characteristics controlling glyphosate mineralization in Danish surface soils. Appl Soil Ecol 27:233–242
Glusczak L, Miron DS, Crestani M, Fonseca MB, Pedron FA, Duarte MF, Vieira VLP (2006) Effect of glyphosate herbicide on acetylcholinesterase activity and metabolic and hematological parameters in piava (Leporinus obtusidens). Ecotox Environ Safe 65:237–241
Glusczak L, Loro VL, Pretto A, Moreas BS, Raabe A, Duarte MF, da Fonseca MB, de Menezes CC, Valladão DM (2011) Acute exposure to glyphosate herbicide affects oxidative parameters in piava (Leporinus obtusidens). Arch Environ Contam Toxicol 61:624–630
Guilherme S, Santos MA, Barroso C, Gaivão I, Pacheco M (2010) Differential genotoxicity of Roundup® formulation and its constituents in blood cells of fish (Anguilla anguilla): considerations on chemical interactions and DNA damaging mechanisms. Ecotoxycology 21:1381–1390
Henderson M, Gervais JA, Luukinen B, Buhl K, Stone D (2010) Glyphosate technical fact sheet. National pesticide information center). Oregon State University Extension Services. Available: <http://npic.orst.edu/factsheets/glyphotech.html/> Updated on September 2010
Hildebrand LD, Sullivan DS, Sullivan TP (1982) Experimental studies of rainbow trout populations exposed to field applications of Roundup herbicide. Arch Environ Contam Toxicol 11:93–98
Jiraungkoorskul W, Upatham ES, Kruatrachue M, Sahaphong S, Vichasri-Grams S, Pokethitiyook P (2002) Histopathological effects of Roundup, a glyphosate herbicide, on Nile tilapia (Oreochromis niloticus). Sci Asia 28:121–127
Kavitha C, Malarvizhi A, Kumaran SS, Ramesh M (2010) Toxicological effects of arsenate exposure on hematological, biochemical and liver transaminases activity in an Indian major carp, Catla catla. Food Chem Toxicol 48:2848–2854
Khan A, Shah N, Gul A, Us-Sahar N, Ismail A, Aziz MF, Farooq M, Adnan M, Rizwan M (2016) Comparative study of toxicological impinge of glyphosate and atrazine (Herbicide) on stress biomarkers: blood biochemical and hematological parameters of the freshwater Common Carp (Cyprinus carpio). Pol J Environ Stud 25:1995–2001. https://doi.org/10.15244/pjoes/62698
Kondera E, Witeska M (2013) Cadmium and copper reduce hematopoietic potential in common carp (Cyprinus carpio L.) head kidney. Fish Physiol Biochem 39:755–764. https://doi.org/10.1007/s10695-012-9738-6
Kondera E (2011) Haematopoiesis in the head kidney of common carp (Cyprinus carpio L): a morphological study. Fish Physiol Biochem 37:355–362
Kotsanis N, Iliopoulou-Georgudaki J, Kapata-Zoumbos K (2000) Changesin selected hematological parameters at early stages of the rainbow trout, Oncorhynchus mykiss, subjected to metal toxicants: arsenic, cadmium and mercury. J Appl Ichthyol 16:276–278
Kreutz LC, Barcellos LJG, Marteninghe A, dos Santos ED, Zanatta R (2010) Exposure to sublethal concentration of glyphosate or atrazine-based herbicides alters the phagocytic function and increases the susceptibility of silver cat fish fingerlings (Rhamdia quelen) to Aeromonas hydrophila challenge. Fish Shellfish Immunol 29:694–697
Kreutz LC, Barcellos LJG, de Faria Valle S, de Oliveira Silva T, Anziliero D, dos Santos ED, Pivato M, Zanatta R (2011) Altered hematological and immunological parameters in silver catfish (Rhamdia quelen) following short term exposure to sublethal concentration of glyphosate. Fish Shellfish Immunol 30:51–57
Langiano VC, Martinez CBR (2008) Toxicity and effects of a glyphosate-based herbicide on the Neotropical fish Prochilodus lineatus. CBP 147:222–231
LeÁvesque A, Rahe JE (1992) Herbicide interactions with fungal root pathogens, with special reference to glyphosate. Annu Rev Phytopathol 30:579–602
Mallory-Smith CA, Ratzinger EJ Jr (2003) Revised classification of herbicides by sites of action for weed resistance management strategies. Weed Technol 17:605–619
Modesto KA, Martinez CBR (2010) Effects of Roundup Transorb on fish: hematology, antioxidant defenses and acetylcholinesterase activity. Chemosphere 81:781–787
Neskovic NK, Poleksic V, Elezovic I, Karan V, Budimir M (1996) Biochemical and histopathological effects of glyphosate on carp, Cyprinus carpio L. Bull Environ Contam Toxicol 56:295–302
Okomoda VT, Ataguba GA, Ayuba VO (2013) Hematological response of Clarias gariepinus fingerlings exposed to acute concentrations of Sunsate®. J Stress Physiol Biochem 9:271–278
Olurin KB, Olojo EAA, Mbaka GO, Akindele AT (2006) Histopathological responses of the gill and liver tissues of Clarias gariepinus fingerlings to the herbicide, glyphosate. Afr J Biotechnol 5:2480–2487
Orun I, Dogru MI, Erdogan K, Dogru A, Yuksel E, Talas ZS (2013) Effects of acute and chronic exposure to glyphosate on common carp (Cyprinus carpio L.) hematological parameters: the beneficial effect of propolis. Fresenius Environ Bull 22:2504–2509
Relyea RA (2006) The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities: response. Ecol Appl 16:2027–2034
Salbego J, Pretto A, Gioda CR, Cavalheiro de Menezes C, Lazzari R, Neto JR, Baldisserotto B, Loro VL (2010) Herbicide formulation with glyphosate affects growth, acetylcholinesterase activity, and metabolic and hematological parameters in piava (Leporinus obtusidens). Arch Environ Contam Toxicol 58:740–745. https://doi.org/10.1007/s00244-009-9464-y
Schuette AJ (1998) Environmental fate of glyphosate, in: Environmental Monitoring & Pest Management, Department of Pesticide Regulation, Sacramento, CA9 5824-5624, Available: http://www.cdpr.ca.gov/docs/emon/pubs/fatememo/glyphos.pdf/ Updated on November 1998
Sopińska A, Grochoła A, Niezgoda J (2000) Influence of water polluted with herbicide Roundap on the organism of fish. Med Wet 56:593–597 [In Polish]
Stoyanova S, Yancheva V, Iliev I, Vasileva T, Bivolarski V, Velcheva I, Georgieva E (2015) Glyphosate induces morphological and enzymatic changes in common carp (CyprinuscarpioL.) liver. Bulg J Agric Sci 21:409
Studnicka M, Siwicki AK, Ryka B (1985) Phagocytic ability of neutrophils in carp (Cyprinus carpio L.) Isr J Aquacult Bamidgeh 37:123–128
Svobodova Z, Groch L, Flajshans M, Vykusova B, Machova J (1997) The effect of long-term therapeutic bath of malachite green on common carp Cyprinus carpio L. Acta Vet Brno 66:111–117
Svobodova Z, Pravda D, Palackova J (1991) Unified methods of haematological examination of fish VURH Vodnany, 31
Svobodova Z, Vykusova B, Machova J (1994) The effects of pollutants on selected haematological and biochemical parameters in fish. In: Müller,R, Lloyd,R (Eds.), Sublethal and chronic effects of pollutants on fresh water fish. FAO Fishing NewsBooks, Great Britain, pp. 39–52
Szarek J, Siwicki A, Andrzejewska A, Terech-Majewska E, Banaszkiewicz T (2000) Effects of the herbicide Roundups on the ultrastructural pattern of hepatocytes in carp (Cyprinus carpio). Mar Environ Res 50:263–266
Tu MC, Hurd R, Robison J, Randall M (2001) Glyphosate weed control methods handbook, The Nature Conservancy, pp. 7e1–7e10, Available: http://www.invasive.org/gist/products/handbook/14Glyphosate.pdf/>. Updates on November 2001
U.S. EPA. Pesticides Industry Sales and usage: 2006 and 2007 market estimates; U.S. Environmental Protection Agency: Washington, DC, 2011
Wendelaar Bonga SE (1997) The stress response in fish. Physiol Rev 77:591–625
Acknowledgments
This work was financed from the science grant 387/14/S granted by the Ministry of Science and Higher Education.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kondera, E., Teodorczuk, B., Ługowska, K. et al. Effect of glyphosate-based herbicide on hematological and hemopoietic parameters in common carp (Cyprinus carpio L). Fish Physiol Biochem 44, 1011–1018 (2018). https://doi.org/10.1007/s10695-018-0489-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-018-0489-x