Abstract
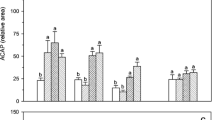
The Paramisgurnus dabryanus was exposed to 30 mmol L−1 NH4Cl solution and air to assessing the change of body ammonia and urea contents and the activities of alanine aminotransferase (ALT) and aspartate transaminase (AST). After 48 h of ammonia exposure, ammonia concentration in the plasma, brain, liver and muscle were 3.3-fold, 5.6-fold, 3.5-fold and 4.2-fold, respectively, those of the control values. Plasma, brain, liver and muscle ammonia concentrations increased to 2.2-fold, 3.3-fold, 2.5-fold and 2.9-fold, respectively, those of control values in response to 48 h of aerial exposure. Within the given treatment (ammonia or aerial exposure), there was no change in plasma, brain and liver urea concentrations between exposure durations. The plasma ALT activity was significantly affected by exposure time during aerial exposure, while the liver ALT activity was not affected by ammonia or aerial exposure. Exposure to NH4Cl or air had no effect on either plasma or liver AST activity. Our results suggested that P. dabryanus could accumulate quite high level of internal ammonia because of the high ammonia tolerance in its cells and tissues, and NH3 volatilization would be a possible ammonia detoxification strategy in P. dabryanus. Urea synthesis was not an effective mechanism to deal with environmental or internal ammonia problem. The significant increase of ALT activity in plasma during aerial exposure, indicating that alanine synthesis through certain amino acid catabolism may be subsistent in P. dabryanus.









Similar content being viewed by others
References
Arillo A, Margiocco C, Melodia F, Mensi P, Schenone G (1981) Ammonia toxicity mechanism in fish: studies on rainbow trout (Salmo gairdneri Rich.). Ecotox Environ Safe 5:316–308
Bergmeyer HU, Beutler HO (1985) Ammonia. In: Bergmeyer HU, Bergmeyer J, Grassl M (eds) Methods of enzymatic analysis, vol VIII. Academic Press, New York, pp. 454–461
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chew SF, Ip YK (2014) Excretory nitrogen metabolism and defence against ammonia toxicity in air-breathing fishes. J Fish Biol 84:603–638
Chew SF, Jin Y, Ip YK (2001) The loach Misgurnus anguillicaudatus reduces amino acid catabolism and accumulates alanine and glutamine during aerial exposure. Physiol Biochem Zool 74:226–237
Chew SF, Wong MY, Tam WL, Ip YK (2003) The snakehead Channa asiatica accumulates alanine during aerial exposure, but is incapable of sustaining locomotory activities on land through partial amino acid catabolism. J Exp Biol 206:693–704
Chew SF, Chan NKY, Tam WL, Loong AM, Hiong KC, Ip YK (2004) The African lungfish, Protopterus dolloi, increases the rate of urea synthesis despite a reduction in ammonia production during 40 days of aestivation in a mucus cocoon. J Exp Biol 207:777–786
Durand F, Regnault M (1998) Nitrogen metabolism of two portunid crabs, Carcinus maenas and Necora puber, during prolonged air exposure and subsequent recovery: a comparative study. J Exp Biol 201:2515–2528
Evans DH, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol Rev 85:97–177
Frick NT, Wright PA (2002) Nitrogen metabolism and excretion in the mangrove killifish Rivulus marmoratus II. Significant ammonia volatilization in a teleost during air-exposure. J Exp Biol 205:91–100
Hochachka PW, Guppy M (eds) (1987) Metabolic arrest and the control of biological time. Harvard University Press, London, pp. 101–119
Ip YK, Chew SF (2010) Ammonia production, excretion, toxicity, and defense in fish: a review. Front Physiol 1:134. doi:10.3389/fphy.2010.00134
Ip YK, Tay ASL, Lee KH, Chew SF (2004) Strategies for surviving high concentrations of environmental ammonia in the swamp eel Monopterus albus. Physiol Biochem Zool 77:390–405
Ip YK, Leong MWF, Sim MY, Goh GS, Chew SF (2005) Chronic and acute ammonia toxicity in mudskipper, Periophthalmodon schlosseri and Boleophthalmus boddaerti: brain ammonia and glutamine contents, and effects of methionine sulfoximine and MK801. J Exp Biol 208:1993–2004
Iwata K, Kajimura M, Sakamoto T (2000) Functional ureogenesis in the gobiid fish Mugilogobius abei. J Exp Biol 203:3703–3715
Jow LY, Chew SF, Lim CB, Anderson PM, Ip YK (1999) The marble goby Oxyeleotris marnorata activates hepatic glutamine synthetase and detoxifies ammonia to glutamine during air exposure. J Exp Biol 202:237–245
Loong AM, Hiong KC, Lee SLM, Wong WP, Chew SF, Ip YK (2005) Ornithine-urea cycle and urea synthesis in African lungfishes, Protopterus aethiopicus and Protopterus annectens, exposed to terrestrial conditions for 6 days. J Exp Zool 303A:354–365
Miñana MD, Hermenegildo C, Llansola M, Montoliu C, Grisolia S, Felipo V (1996) Carnitine and choline derivatives containing a trimethylamine group prevent ammonia toxicity in mice and glutamate toxicity in primary cultures of neurons. J Pharmacol Exp Ther 279:194–199
Paust LO, Foss A, Imsland AK (2011) Effects of chronic and periodic exposure to ammonia on growth, food conversion efficiency and blood physiology in juvenile Atlantic halibut (Hippoglossus hippoglossus L.). Aquaculture 315:400–406
Rahmahtullah M, Boyde TR (1980) Improvements in the determination urea using diacetyl monoxime; methods with and without deproteinization. Clin Chim Acta 107:3–9
Ramaswamy M, Reddy TG (1983) Ammonia and urea excretion in three species of air-breathing fish subjected to aerial exposure. Proc Indian Acad Sci (Anim Sci) 92:293–297
Randall DJ, Tsui TKN (2002) Ammonia toxicity in fish. Mar Pollut Bull 45:17–23
Randall DJ, Wilson JM, Peng KW, Kok TWK, Kuah SSL, Chew SF, Lam TJ, Ip YK (1999) The mudskipper, Periophthalmodon schlosseri, actively transports NH4 + against a concentration gradient. Am J Physiol-Regul Integr Comp Physiol 277:R1562–R1567
Saha N, Ratha BK (1989) Comparative study of ureogenesis in freshwater, aire-breathing teleosts. J Exp Zool 252:1–8
Sinha AK, Rasoloniriana R, Dasan AF, Pipralia N, Blust R, De Boeck G (2015) Interactive effect of high environmental ammonia and nutritional status on ecophysiological performance of European sea bass (Dicentrarchus labrax) acclimated to reduced seawater salinities. Aquat Toxicol 160:39–56
Steele SL, Yancey PH, Wright PA (2005) The little skate Raja erinacea exhibits an extrahepatic ornithine urea cycle in the muscle and modulates nitrogen metabolism during low-salinity challenge. Physiol Biochem Zool 78:216–226
Tay ASL, Chew SF, Ip YK (2003) The swamp eel Monopterus albus reduces endogenous ammonia production and detoxifies ammonia to glutamine during 144 h of aerial exposure. J Exp Biol 206:2473–2486
Tay ASL, Loong AM, Hiong KC, Lee SJ, Tng YYM, Wee NLJ, Lee SML, Wong WP, Chew SF, Wilson JM, Ip YK (2006) Active ammonia transport and excretory nitrogen metabolism in the climbing perch, Anabas testudineus, during 4 days of emersion or 10 minutes of forced exercise on land. J Exp Biol 209:4475–4489
Tng YYM, Chew SF, Wee NLJ, Wong FK (2009) Acute ammonia toxicity and the protective effects of methionine sulfoximine on the swamp eel, Monopterus albus. J Exp Zool 311A:676–688
Tsui TKN (2005) Mechanisms of ammonia tolerance in the oriental weatherloach, Misgurnus anguillicaudatus. Run Run Shaw Library, City University of Hong Kong, Hong Kong China
Tsui TKN, Randall DJ, Chew SF, Jin Y, Wilson JM, Ip YK (2002) Accumulation of ammonia in the body and NH3 volatilization from alkaline regions of the body surface during ammonia loading and exposure to air in the weather loach Misgurnus anguillicaudatus. J Exp Biol 205:651–659
Tsui TKN, Randall DJ, Hanson L, Farrell AP, Chew SF, Ip YK (2004) Dogmas and controversies in the handling of nitrogenous wastes: ammonia tolerance in the oriental weatherloach Misgurnus anguillicaudatus. J Exp Biol 207:1977–1983
Veauvy CM, McDonald MD, van Audekerke J, Vanhoutte G, van Camp N, van der Linden AT, Walsh PJ (2005) Ammonia affects brain nitrogen metabolism but not hydration status in the Gulf toadfish (Opsanus beta). Aquat Toxicol 74:32–46
Walsh PJ, Danulat E, Mommsen TP (1990) Variation in urea excretion in the gulf toadfish (Opsanus beta). Mar Biol 106:323–328
Wee NLJ, Tng YYM, Cheng HT, Lee SML, Chew SF, Ip YK (2007) Ammonia toxicity and tolerance in the brain of the African sharptooth catfish, Clarias gariepinus. Aquat Toxicol 82:204–213
Zhang L, Xiong D-M, Li B, Zhao Z-G, Fang W, Yang K, Fan Q-X (2012) Toxicity of ammonia and nitrite to yellow catfish (Pelteobagrus fulvidraco). J Appl Ichthyol 28:82–86
Zhang Y-L, Hu W-H, Wu Q-W, Wang F, Zhao Z-B, He H, Liu R-P, Fan Q-X (2015a) Ontogenetic changes in RNA, DNA and protein contents of Chinese loach, Paramisgurnus dabryanus (Dabry de Thiersant, 1872), larvae and juveniles. J Appl Ichthyol 31:876–882
Zhang Y-L, Wu Q-W, Hu W-H, Wang F, Zhao Z-B, He H, Shao W-H, Fan Q-X (2015b) Changes in digestive enzyme activities during larval development of Chinese loach Paramisgurnus dabryanus (Dabry de Thiersant, 1872). Fish Physiol Biochem 41:1577–1585
Zhang Y, Wu Q, Hu W, Wang F, Shao W, Zhang C, Zhao Z, He H, Fan Q, Gu Z (2016) Morphological changes and allometric growth in hatchery-reared Chinese loach Paramisgurnus dabryanus (Dabry de Thiersant, 1872). Chin J Oceanol Limnol 34:757–762
Acknowledgements
This work was funded by National Science and Technology Support Program, China (grant No. 2012BAD25B08; 2012BAD25B00).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, YL., Zhang, HL., Wang, LY. et al. Changes of ammonia, urea contents and transaminase activity in the body during aerial exposure and ammonia loading in Chinese loach Paramisgurnus dabryanus. Fish Physiol Biochem 43, 631–640 (2017). https://doi.org/10.1007/s10695-016-0317-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-016-0317-0