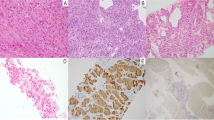
Abstract
The majority of pancreatic neuroendocrine tumors (PNETs) are sporadic while 10–15% are attributable to one of several familial cancer syndromes. Hereditary forms are more commonly associated with Multiple Endocrine Neoplasia Type I and von Hippel Lindau Syndrome. However, patients with Tuberous sclerosis complex also have an increased incidence of PNETs. More often this has been reported in patients with TSC2 variants. In this case report, we summarize the literature regarding PNETs associated with Tuberous sclerosis complex, as well as present a case of a patient with a TSC1 variant and a PNET. This case highlights the association of TSC1 gene variants with these tumors and emphasizes the importance of considering such diagnoses in this patient population.





Similar content being viewed by others
References
Northrup H, Koenig M, Pearson D, Au K (2015) Tuberous sclerosis complex. In: GeneReviews at GeneTests: medical genetics information resource, University of Washington, Seattle, pp 1997–2016. https://www.ncbi.nlm.nih.gov/books/NBK1220/
Santos L, Brcic I, Unterweger G, Riddell R, Langner C (2015) Hamartomatous polyposis in tuberous sclerosis complex: case report and review of the literature. Pathol-Res Pract 211(12):1025–1029
Kumar V, Abbas A, Aster J (2015) Tuberous sclerosis complex. In: Robbins and cotran pathologic basis of disease 9th edn. Elsevier, Philadelphia
Lodish MB, Stratakis CA (2010) Endocrine tumours in neurofibromatosis type 1, tuberous sclerosis and related syndromes. Best Pract Res Clin Endocrinol Metab 24(3):439–449
Northrup H, Krueger DA (2013) International tuberous sclerosis complex consensus group. Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 international tuberous sclerosis complex consensus conference. Pediatr Neurol 49:243–254
Sancak O, Nellist M, Goedblood M, Elfferich P, Wouters C, Maat-Kievit A, Zonnenberg B, Verhoef S, Halley D, van den Ouweland A (2005) Mutational analysis of the TSC1 and TSC2 genes in a diagnostic setting: genotype-phenotype correlations and comparison of diagnostic DNA techniques in tuberous sclerosis complex. Eur J Hum Genet 13:731–741
Dabora SL, Jozwiak S, Franz DN, Roberts PS, Nieto A, Chung J, Choy YS, Reeve MP, Thiele E, Egelhoff JC, Kasprzyk-Obara J, Domanska-Pakiela D, Kwiatkowski DJ (2001) Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1 disease in multiple organs. Am J Hum Genet 68:64–80
Jansen FE, Braams O, Vincken KL, Algra A, Anbeek P, Jennekens-Schinkel A, Halley D, Zonnenberg BA, van den Ouweland A, van Huffelen AC, van Nieuwenhuizen O, Nellist M (2008) Overlapping neurologic and cognitive phenotypes in patients with TSC1 or TSC2 mutations. Neurology 70:908–915
Jansen AC, Sancak O, D’Agostino MD, Badhwar A, Roberts P, Gobbi G, Wilkinson R, Melanson D, Tampieri D, Koenekoop R, Gans M, Maat-Kievit A, Goedbloed M, van den Ouweland AM, Nellist M, Pandolfo M, McQueen M, Sims K, Thiele EA, Dubeau F, Andermann F, Kwiatkowski DJ, Halley DJ, Andermann E (2006) Unusually mild tuberous sclerosis phenotype is associated with TSC2 R905Q mutation. Ann Neurol 60:528–539
Kothare SV, Singh K, Chalifourx JR, Staley BA, Weiner HL, Menzer K, Devinsky O (2014) Severity of manifestations in tuberous sclerosis complex in relation to genotype. Epilepsia 55(7):1025–1029
Rosset C, Netto CB, Ashton-Prolla P (2017) TSC1 and TSC2 gene mutations and their implications for treatment in tuberous sclerosis complex: a review. Genet Mol Biol. doi:10.1590/1678-4685-gmb-2015-0321
Lewis JC, Thomas HV, Murphy KC, Sampson JR (2004) Genotype and psychological phenotype in tuberous sclerosis. J Med Genet 41:203–207
Numis AL, Major P, Montenegro MA, Muzykewica DA, Pulsifer MB, Thiele EA (2011) Identification of risk factors for autism spectrum disorders in tuberous sclerosis complex. Neurology 76:981–987
Al-Saleem T, Wessner LL, Scheithauer BW, Patterson K, Roach ES, Dreyer SJ, Fujikawa K, Bjornsson J, Bernstein J, Henske EP (1998) Malignant tumors of the kidney, brain and soft tissues in children and young adults with the tuberous sclerosis complex. Cancer 83:2016–2208
Yang P, Cornejo KM, Sadow PM, Cheng L, Wang M, Xiao Y, Jiang Z, Oliva E, Jozwiak S, Nussbaum RL, Feldman AS, Paul E, Thiele EA, Yu JJ, Henske EP, Kwiatkowski DJ, Young RH, Wu CL (2014) Renal cell carcinoma in tuberous sclerosis complex. Am J Surg Pathol 38:895–909
Larson AM, Hedgire SS, Deshpande V, Stemmer-Rachamimov AO, Harisinghani MG, Ferrone CR, Shah U, Thiele EA (2012) Pancreatic neuroendocrine tumors in patients with tuberous sclerosis complex. Clin Genet 82:558–563
Diaz Diaz D, Ibarrola C, Gomez-Sanz R, Perez Hurtado B, Salazar Tabares J, Colina Ruizdelgado F (2012) Neuroendocrine tumor of the pancreas in a patient with tuberous sclerosis: a case report and review of the literature. Int J Surg Pathol 20(4):390–395
Gustafson S, Eng S (2008) Patient with multifocal renal cell carcinoma, pancreatic neuroendocrine tumor and TSC1 Q343X mutation, TSC2 SNP and PTEN c.210-7 ~ 3del5 http://www.ashg.org/2008meeting/abstracts/fulltext/f20544.htm. Accessed 14 April 2017
Krueger DA, Northrup H (2013) Tuberous sclerosis complex surveillance and management of the 2012 International tuberous sclerosis complex consensus conference. Pediatr Neurol 49(4):255–265
Dworakowska D, Grossman AB (2009) Are neuroendocrine tumors a feature of tuberous sclerosis? A systematic review. Endocr Relat Cancer 16(1):45–58
Arva NC, Pappas JG, Bhatla T, Raetz EA, Macari M, Ginsburg HB, Hajdu CH (2012) Well differentiated pancreatic neuroendocrine carcinoma in tuberous sclerosis: case report and review of the literature. Am J Surg Pathol 36(1):149–153
Laplante M, Sabatini DM (2012) mTOR signaling in growth and disease. Cell 149(2):274–293
Cinglarlini SS, Bonomi M, Corbo V, Scarpa A, Tortora (2012) Profiling mTOR pathway in neuroendocrine tumors. Target Oncol 7(3):183–188
Koc G, Sugimoto S, Kuperman R, Kammen B, Karakas SP (2016) Pancreatic tumors in children and young adults with tuberous sclerosis complex. Pediatr Radiol 47:39–45
Bombardierei R, Moavero R, Roberto D, Cerminara C, Curatolo P (2013) Pancreatic Neuroendocrine tumor in a child with a tuberous sclerosis complex 2 (TSC2) mutation. Endocr Pract 19(5):e124–e128
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not identify any conflicts of interest.
Rights and permissions
About this article
Cite this article
Mortaji, P., Morris, K.T., Samedi, V. et al. Pancreatic neuroendocrine tumor in a patient with a TSC1 variant: case report and review of the literature. Familial Cancer 17, 275–280 (2018). https://doi.org/10.1007/s10689-017-0029-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-017-0029-3