Abstract
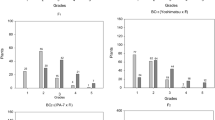
Bacterial wilt is a disease causing great losses to the tomato crop. Research investigating inheritance of resistance to this disease helps to improve the efficiency of breeding programs in the development of resistant cultivars. The objective of this study was to determine the inheritance of resistance to Ralstonia pseudosolanacearum in tomato plants. The parents Yoshimatsu and IPA-7; generations F1, F2, BC11, and BC21; and 43 F2:3 progenies were evaluated. The severity of bacterial wilt were assessed through a descriptive scale of scores at 10 and 20 days after inoculation. Inheritance of resistance to R. pseudosolanacearum in tomato involves two major genes with segregation independent of additive effects only, plus polygenes with additive and dominance effects wherein resistance is associated with recessive alleles. Selection of plants resistant to R. pseudosolanacearum is indicated mainly at 20 days after inoculation.


Similar content being viewed by others
References
Batista RO, Silva LC, Moura LM, Souza MH, Carneiro PCS, Carvalho Filho JLS, Carneiro JES (2017) Inheritance of resistance to fusarium wilt in common bean. Euphytica 2013:133. https://doi.org/10.1007/s10681-017-1925-1
Carvalho Filho JLS, Gomes LAA, Maluf WR, Oliveira RR, Costa DS, Ferreira S, Monteiro AB, Costa RR (2011) Resistance to Meloidogyne incognita race 1 in the lettuce cultivars Grand Rapids and Salinas-88. Euphytica (Wageningen) 182:199–208. https://doi.org/10.1007/s10681-011-0429-7
Coupat B, Chaumeille-dole F, Fall S, Prior P, Simonet P, Nesme X (2008) Natural transformation in the Ralstonia solanacearum species complex: number and size of DNA that can be transferred. FEMS Microbiol Ecol 66:14–24
Denny T (2006) Plant pathogenic Ralstonia species. In: Gnanamanickam SS (ed) Plant-associated bacteria. Springer, Dordrecht, pp 573–644
Elphinstone JG (2005) The current bacterial wilt situation: a global overview. In: Allen C, Prior P, Hayward AC (eds) Bacterial wilt disease and the R. solanacearum species complex. American Phytopathological Society Press, St. Paul, pp 9–28
Faostat (2016) Database results. http://www.fao.org/faostat/en/#data/QC/visualize. Accessed 14 April 2018
Felix KC, Souza EB, Michereff SJ, Mariano RLR (2012) Survival of R. solanacearum in infected tissues of Capsicum annuum and in soils of the state of Pernambuco, Brazil. Phytoparasitica 40:53–62
Ferrer ZA (1984) The nature of resistance in a tomato tolerant to Pseudomonas solanacearum. Phytopathology 74:1014
Garcia AL, Lima WG, Souza EB, Michereff SJ, Mariano RLR (2013) Characterization of Ralstonia solanacearum causing bacterial wilt bell pepper in the state Pernambuco, Brazil. J Plant Pathol 95:237–245
Graham KM, Yap TC (1976) Studies on bacterial wilt. I. Inheritance of resistance to Pseudomonas solanacearum in tomato. Malays Agric Res 5:1–8
Grimault V, Prior P, Anais G (1995) A monogenic dominant resistance of tomato to bacterial wilt in Hawaii 7996 is associated with plant colonization by Pseudomonas solanacearum. J Phytopathol 143:349–352. https://doi.org/10.1111/j.1439-0434.1995.tb00274.x
Hong Hai TT, Esch E, Wang JF (2008) Resistance to Taiwanese race 1 strains of R. solanacearum in wild tomato germplasm. Eur J Plant Pathol 122:471–479. https://doi.org/10.1007/s10658-008-9314-1
Huet G (2014) Breeding for resistances to R. solanacearum. Front Plant Sci 5:1–5
Instituto Brasileiro de Geografia e Estatística–IBGE (2017) Levantamento sistemático da produção agrícola (LSPA). Rio de Janeiro 29:1–79
Instituto Nacional de Meteorologia (Inmet) (2016) Dados meteorológicos da estação do Curado-Recife. http://www.inmet.gov.br/portal/. Accessed 03 Apr 2017
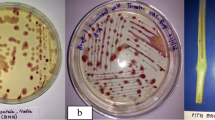
Kelman A (1954) The relationship of pathogenicity in Pseudomonas solanacearum to colony appearance on a tetrazolium medium. Phytopathology 44:693–695
Lima Neto AF, Silveira MA, Souza RM, Nogueira SR, André CMG (2002) Inheritance of bacterial wilt resistance in tomato plants cropped in naturally infested soils of the state of Tocantins. Crop Breed Appl Biotechnol 2:25–32
Lopes CA, Boiteux LS (2012) Melhoramento para resistência a doenças bacterianas. In: Fritse-Neto R, Borém A (eds) Melhoramento de plantas para condições de estresses bióticos. Editora UFV, Viçosa, pp 61–88
Lopes CA, Mendonça JL (2014) Enxertia em tomateiro para o controle da murcha-bacteriana. Circular técnica 131, Embrapa
Lopes CA, Boiteux LS, Eschemback V (2015) Eficácia relativa de porta enxertos comerciais de tomateiro no controle da murcha bacteriana. Hortic Bras 33:125–130. https://doi.org/10.1590/S0102-053620150000100020
Mather K, Jinks JL (1982) Biometrical genetics, 3rd edn. Cambridge University Press, Cambridge
McDonald BA, Linde C (2002) Pathogen population genetics, evolutionary potential, and durable resistance. Annu Rev Phytopathol 40:349–379
Menezes CB, Maluf WR, Azevedo SM, Faria MV, Nascimento IR, Nogueira DW, Gomes LAA, Bearzoti E (2005) Inheritance of parthenocarpy in summer squash (Cucurbita pepo L.). Genet Mol Res 4:39–46
Menezes CB, Maluf WR, Faria MV, Azevedo SM, Resende JTV, Figueira AR, Gomes LAA (2015) Inheritance of resistance to papaya ringspot virus-watermelon strain (PRSV-W) in Whitaker summer squash line. Crop Breed Appl Biotechnol 15:203–209
Monma S, Sakata Y, Matsunaga H (1997) Inheritance and selection efficiency of bacterial wilt resistance in tomato. JARQ 31:195–204
Mood AM, Graybill FA, Boes DC (1974) Introduction to the theory of statistics, 3rd edn. McGraw-Hill Kogakusha, Tokio
Nick C, Silva DJH (2016) Melhoramento de tomate. In: Nick C, Borém A (eds) melhoramento de hortaliças. Editora UFV, Viçosa, pp 396–431
Nielsen LW, Haynes FL (1960) Resistance in Solanum tuberosum to Pseudomonas solanacearum. Am Potato J 37:260–267
Noda H (2008) Melhoramento de hortaliças em climas desfavoráveis: o desafio do desenvolvimento de cultivares adaptadas a Amazônia. Melhoramento do Tomateiro para o Tropico Úmido Brasileiro. Hortic Bras. 25(1) (CD-ROM)
Oliveira WF, Giordano LB, Lopes CA (1999) Herança da resistência em tomateiro à murcha-bacteriana. Fitopatologia Brasileira 24:49–53
Persley GJ, Batugal P, Gaparin D, Vander Zaag P (1985) Summary of discussion and recommendations. In: ACIAR proceedings, Canberra, Australia, vol 13, pp 7–13
Ramalho APR, Abreu AFB, Santos JB, Nunes JAR (2012) Aplicações de genética quantitativa no melhoramento de plantas autógamas. Editora UFLA, Lavras
Safni I, Cleenwerck I, De Vos P, Fegan M, Sly L, Kappler U (2014) Polyphasic taxonomic revision of the Ralstonia solanacearum species complex: proposal to emend the descriptions of Ralstonia solanacearum and Ralstonia syzygii and reclassify current R. syzygii strains as Ralstonia syzygii subsp. Syzygii subsp. nov., R. solanacearum phylotype IV strains as Ralstonia syzygii subsp. indonesiensis subsp. nov., banana blood disease bacterium strains as Ralstonia syzygii subsp. celebesensis subsp. nov. and R. solanacearum phylotype I and III strains as Ralstonia pseudosolanacearum sp. nov. Int J Syst Evol Microbiol 64:3087–3103
Santiago TR, Lopes CA, Caetano-Anollés G, Mizubuti ESG (2016) Phylotype and sequevar variability of Ralstonia solanacearum in Brazil, an ancient centre of diversity of the pathogen. Plant Pathol 66:383–392
Sharma KC, Sharma LK (2015) Genetic studies of bacterial wilt resistance in tomato crosses under mid-hill conditions of Himachal Pradesh. J Hill Agric 6:136–137. https://doi.org/10.5958/2230-7338.2015.00026.9
Sigma Plot (2008) For Windows, version, 10.0. Systat Software, San Jose
Silva JA (2003) Estimadores de máxima verossimilhança em misturas de densidade normais: uma aplicação em genética. Tese de mestrado, Universidade Federal de Lavras, Lavras, Minas Gerais, Brasil
Silveira MS, Queiróz MA, Lima JAA, Nunes JHS, Nascimento AKQ, Lima Neto IS (2015) Herança da resistência ao papaya ringspot virus em melancia. Rev Caatinga 28:128–136. https://doi.org/10.1590/1983-21252015v28n315rc
Thakur AK, Kohli UK, Kumar M (2004) Inheritance of resistance to bacterial wilt in tomato (Lycopersicon esculentum Mill.). Indian J Genet Plant Breed 64:79–80
Vencovsky R (1987) Herança quantitativa. In: Paterniani E (ed) Melhoramento e produção de milho no Brasil. Fundação Cargill, Piracicaba, pp 1204–1214
Wicker E, Lefeuvre P, De Cambiaire JC, Lemaire C, Poussier S, Prior P (2012) Contrasting recombination patterns and demographic histories of the plant pathogen Ralstonia solanacearum inferred from MLSA. Int Soc Microb Ecol J 6:961–974
Wright S (1934) The results of crosses between inbred strains of guinea pigs, differing in number of digits. Genetics 19:537–551
Acknowledgements
To the Agronomic Institute of Pernambuco for the support in the stage of the hybrids and to the Laboratory of Phytobacteriology of the Federal Rural University of Pernambuco for the support in the stages of inoculation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
da Silva Costa, K.D., dos Santos, A.M.M., dos Santos, P.R. et al. Inheritance of resistance to Ralstonia pseudosolanacearum in tomato. Euphytica 214, 137 (2018). https://doi.org/10.1007/s10681-018-2217-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-018-2217-0