Abstract
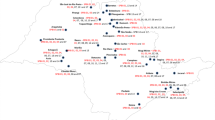
The fungal pathogen Leptosphaeria maculans (LM), causing blackleg in oilseed rape (OSR), develops gene-for-gene interactions with OSR cultivars. Thus, virulent races can cause severe OSR yield losses in genotypes that depend only on the major resistance genes. LM race spectra were determined using 644 isolates collected from four OSR growing areas in northern Germany in autumn 2011 and 2012. Avirulence alleles (AvrLm) and race spectra of the isolates was identified with OSR cultivars harbouring the major genes Rlm1, Rlm2, Rlm3, Rlm4, Rlm7, and Rlm9. Thirteen LM races were determined, whereby 85 % of the collected isolates belonged to race AvrLm(5)-(6)-7-(8) (virulent to Rlm1–4 and Rlm9) and 10 % belonged to race AvrLm1-(5)-(6)-7-(8) (virulent to Rlm2–4 and Rlm9). Only six of 644 isolates (0.9 %) showed virulence to Rlm7. Using tester isolates that identify Rlm7, we showed that Rlm7 was absent in the most sown cultivars in northern Germany, explaining the low frequency of isolates being virulent to Rlm7. However, blackleg disease incidence (DI) increased significantly in the cultivar Exocet (harbouring Rlm7) between 2011/12 and 2012/13, whereas DI was the same in a susceptible control cultivar across all sampled regions and years. Our results show that Rlm7 is the only known major resistance gene in German OSR cultivars that is still effective against LM, but increasing deployment in commercial cultivars may foster resistance breakdown in the future.


Similar content being viewed by others
References
Aubertot, J. N., West, J. S., Bousset-Vaslin, L., Salam, M. U., Barbetti, M. J., & Diggle, A. J. (2006). Improved resistance management for durable disease control: a case study of phoma stem canker of oilseed rape (Brassica napus). European Journal of Plant Pathology, 114, 91–106.
Balesdent, M.-H., Attard, A., Ansan-Melayah, D., Delourme, R., Renard, M., & Rouxel, T. (2001). Genetic control and host range of avirulence toward Brassica napus cultivars Quinta and Jet Neuf in Leptosphaeria maculans. Phytopathology, 91, 70–76.
Balesdent, M.-H., Attard, A., Kühn, M. L., & Rouxel, T. (2002). New avirulence genes in the phytopathogenic fungus Leptosphaeria maculans. Phytopathology, 92, 1122–1133.
Balesdent, M.-H., Barbetti, M. J., Li, H., Sivasithamparam, K., Gout, L., & Rouxel, T. (2005). Analysis of Leptosphaeria maculans race structure in a worldwide collection of isolates. Phytopathology, 95, 1061–1071.
Balesdent, M.-H., Louvard, K., Pinochet, X., & Rouxel, T. (2006). A large-scale survey of races of Leptosphaeria maculans occurring on oilseed rape in France. European Journal of Plant Pathology, 114, 53–66.
Balesdent, M.-H., Fudal, I., Ollivier, B., Bally, P., Granaubert, J., Chevre, A., Leflon, M., & Rouxel, T. (2013). The dispensable chromosome of Leptosphaeria maculans shelters an effector gene conferring avirulence towards Brassica rapa. New Phytologist, 198, 889–898.
Balesdent M.-H., Plissoneau C., Coudard, L., Daverdin, G., Le Meur, L., Carpezat, J., Leflon, M., Pinochet, X., Ermel, M., Brun, H., & Rouxel, T. (2015). Résistance du colza au phoma : où en est-on de l’efficacité de la résistance Rlm7 ? Phytoma, in press.
Brun, H., Chèvre, A.-M., Fitt, B. D. L., Powers, S., Besnard, A.-L., Ermel, M., et al. (2010). Quantitative resistance increases the durability of qualitative resistance to Leptosphaeria maculans in Brassica napus. New Phytologist, 185, 285–299.
Daverdin, G., Rouxel, T., Gout, L., Aubertot, J.-N., Fudal, I., Meyer, M., et al. (2012). Genome structure and reproductive behaviour influence the evolutionary potential of a fungal phytopathogen. PLoS Pathogens. doi:10.1371/journal.ppat.1003020.
Delourme, R., Chèvre, A.-M., Brun, H., Rouxel, T., Balesdent, M.-H., Dias, J. S., et al. (2006). Major gene and polygenic resistance to Leptosphaeria maculans in oilseed rape (Brassica napus). European Journal of Plant Pathology, 114, 41–52.
Delourme, R., Bousset, L., Ermel, M., Duffé, P., Besnard, A. L., Marquer, B., et al. (2014). Quantitative resistance affects the speed of frequency increase but not the diversity of the virulence alleles overcoming a major resistance gene to Leptosphaeria maculans in oilseed rape. Infection, Genetics and Evolution. doi:10.1016/j.meegid.2013.12.019.
Fitt, B. D. L., Brun, H., Barbetti, M. J., & Rimmer, S. R. (2006). World-wide importance of phoma stem canker (Leptosphaeria maculans and L. biglobosa) on oilseed rape (Brassica napus). European Journal of Plant Pathology, 114, 3–15.
Fudal, I., Ross, S., Gout, L., Blaise, F., Kühn, M.-L., Eckert, M. R., et al. (2007). Heterochromatin-like regions as ecological niches for avirulence genes in the Leptosphaeria maculans genome: map-based cloning of AvrLm6. Molecular Plant - Microbe Interactions, 20, 459–470.
Ghanbarnia, K., Fudal, I., Larkan, N. J., Links, M. G., Balesdent, M.-H., Profotova, B., Fernando, W. G. D., Rouxel, T., & Borhan, M. H. (2015). Rapid identification of the Leptosphaeria maculans avirulence gene AvrLm2 using an intraspecific comparative genomics approach. Molecular Plant Pathology, 15, 699–709.
Gout, L., Kühn, M.-L., Vincenot, L., Bernard-Samain, S., Cattolico, L., Barbetti, M. J., et al. (2007). Genome structure impacts molecular evolution at the AvrLm1 avirulence locus of the plant pathogen Leptosphaeria maculans. Environmental Microbiology, 9, 2978–2992.
Guo, X. W., Fernando, W. G. D., & Entz, M. (2005). Effects of crop rotation and tillage on blackleg disease of canola. Canadian Journal of Plant Pathology, 27, 53–56.
Howlett, B. J., Idnurum, A., & Pedras, M. S. C. (2001). Leptosphaeria maculans, the causal agent of blackleg disease of brassicas. Fungal Genetics and Biology, 33, 1–14.
Huang, Y. J., Karendeni-Dewage, C. S., & Fitt, B. D. L. (2014). Importance of Leptosphaeria biglobosa as a cause of phoma stem canker on winter oilseed rape in the UK. Aspects of Applied Biology, 127, 117–122.
Kuswinanti, T., Koopmann, B., & Hoppe, H.-H. (1999). Virulence pattern of aggressive isolates of Leptosphaeria maculans on an extended set of Brassica differentials. Journal of Plant Diseases and Protection, 106, 12–20.
Kutcher, H. R., Balesdent, M. H., Rimmer, S. R., Rouxel, T., Chèvre, A. M., Delourme, R., et al. (2010). Frequency of avirulence genes in Leptosphaeria maculans in western Canada. Canadian Journal of Plant Pathology, 32, 77–85.
Kutcher, H. R., Brandt, S. A., Smith, E. G., Ulrich, D., Malhi, S. S., & Johnston, A. M. (2013). Blackleg disease of canola mitigated by resistant cultivars and four-year crop rotations in western Canada. Canadian Journal of Plant Pathology, 35, 209–221.
Lancashire, P. D., Bleiholder, H., van den Boom, T., Langelüddeke, P., Stauss, R., Weber, E., et al. (1991). A uniform decimal code for growth stages of crops and weeds. Annals of Applied Biology, 119, 561–601.
Larkan, N. J., Lydiate, D. J., Parkin, I. A. P., Nelson, M. N., Epp, D. J., Cowling, W. A., et al. (2013). The Brassica napus blackleg resistance gene LepR3 encodes a receptor-like protein triggered by the Leptosphaeria maculans effector AVRLM1. New Phytologist, 197, 595–605.
Long, Y., Wang, Z., Sun, Z., Fernando, D. W. G., McVetty, P. B. E., & Li, G. (2011). Identification of two blackleg resistance genes and fine mapping of one of these two genes in a Brassica napus canola cultivar ‘Surpass 400’. TAG Theoretical and Applied Genetics, 122, 1223–1231.
Marcroft, S. J., Van de Wouw, A. P., Salisbury, P. A., Potter, T. D., & Howlett, B. J. (2012a). Effect of rotation of canola (Brassica napus) cultivars with different complements of blackleg resistance genes on disease severity. Plant Pathology, 61, 934–944.
Marcroft, S. J., Elliott, V. L., Cozijnsen, A. J., Salisbury, P., Howlett, B. J., & Van de Wouw, A. P. (2012b). Identifying resistance genes to Leptosphaeria maculans in Australian Brassica napus cultivars based on reactions to isolates with known avirulence genotypes. Crop and Pasture Science, 63, 338–350.
McDonald, B. A., & Linde, C. (2002). Pathogen population genetics, evolutionary potential, and durable resistance. Annual Review of Phytopathology, 40, 349–379.
NaLaMa-nT (2015). Homepage of the joint project Nachhaltiges Landmanagement im Norddeutschen Tiefland (NaLaMa-nT). URL: http://www.nalama-nt.de/modellregionen.html, visited on November 25, 2015.
Parlange, F., Daverdin, G., Fudal, I., Kühn, M.-L., Balesdent, M.-H., Blaise, F., et al. (2009). Leptosphaeria maculans avirulence gene AvrLm4-7 confers a dual recognition specificity by the Rlm4 and Rlm7 resistance genes of oilseed rape, and circumvents Rlm4-mediated recognition through a single amino acid change. Molecular Microbiology, 71, 851–863.
Rouxel, T., Penaud, A., Pinochet, X., Brun, H., Gout, L., Delourme, R., et al. (2003). A 10-year survey of populations of Leptosphaeria maculans in France indicates a rapid adaptation towards the Rlm1 resistance gene of oilseed rape. European Journal of Plant Pathology, 109, 871–882.
Shoemaker, R. A., & Brun, H. (2001). The teleomorph of the weekly aggressive segregate of Leptosphaeria maculans. Canadian Journal of Botany, 79, 412–419.
Sprague, S. J., Balesdent, M.-H., Brun, H., Hayden, H. L., Marcroft, S. J., Pinochet, X., et al. (2006). Major gene resistance in Brassica napus (oilseed rape) is overcome by changes in virulence of populations of Leptosphaeria maculans in France and Australia. European Journal of Plant Pathology, 114, 33–40.
Stachowiak, A., Olechnowicz, J., Jedryczka, M., Rouxel, T., & Balesdent, M.-H. (2006). Frequency of avirulence alleles in field populations of Leptosphaeria maculans in Europe. European Journal of Plant Pathology, 114, 67–75.
Statista 2015: Homepage Statista. Http: de.statista.com. Accessed 20 Apr 2015.
Tokunaga, J., & Bartnicki-Garcia, S. (1971). Cyst wall formation and endogenous carbohydrate utilization during synchronous encystment of Phytophthora cinnamomi zoospores. Archiv für Mikrobiologie, 19, 283–292.
Van de Wouw, A. P., & Howlett, B. (2012). Estimating frequencies of virulent isolates in field populations of a plant pathogenic fungus, Leptosphaeria maculans using high-throughput pyrosequencing. Journal of Applied Microbiology, 113, 1145–1153.
Van de Wouw, A. P., Marcroft, S. J., Barbetti, M. J., Hua, L., Salisbury, P. A., Gout, L., et al. (2009). Dual control of avirulence in Leptosphaeria maculans towards a Brassica napus cultivar with ‘sylvestris-derived’ resistance suggests involvement of two resistance genes. Plant Pathology, 58, 305–313.
Van de Wouw, A. P., Cozijnsen, A. J., Hane, J. K., Brunner, P. C., McDonald, B. A., Oliver, R. P., et al. (2010). Evolution of linked avirulence effectors in Leptosphaeria maculans is affected by genomic environment and exposure to resistance genes in host plants. PLoS Pathogens, 6, e1001180.
Van de Wouw, A. P., Lowe, R. G. T., Elliott, C. E., Dubois, D. J., & Howlett, B. J. (2014). An avirulence gene, AvrLmJ1, from the blackleg fungus, Leptosphaeria maculans, confers avirulence to Brassica juncea cultivars. Molecular Plant Pathology, 15, 523–530.
Williams, P. H., & Delwiche, P. A. (1979). Screening for resistance to blackleg of crucifers in the seedling stage. Proceedings Eurocarpia Conference: Breeding of Cruciferous Crops, Wageningen, Netherlands, 164–170.
Yu, F., Lydiate, D. J., & Rimmer, S. R. (2005). Identification of two novel genes for blackleg resistance in Brassica napus. Theoretical and Applied Genetics, 110, 969–979.
Yu, F., Lydiate, D. J., & Rimmer, S. R. (2008). Identification and mapping of a third blackleg resistance locus in Brassica napus derived from B. rapa subsp. sylvestris. Genome, 51, 64–72.
Yu, F., Gugel, R. K., Kutcher, H. R., Peng, G., & Rimmer, S. R. (2013). Identification and mapping of a novel blackleg resistance locus LepR4 in the progenies from Brassica napus × B. rapa subsp. sylvestris. Theoretical and Applied Genetics, 126, 307–315.
Acknowledgments
This work was supported by the Federal Ministry of Education and Research (BmBF) in Germany through the Project Management Jülich (PtJ), Germany (033L029B PT-J). Further we would like to thank Claudia Nordmann and Evelyn Vorbeck for excellent technical assistance. We kindly acknowledge Susanne Stein (Centre of Biodiversity and Sustainable Land Use, University of Goettingen, Germany) and Stefania Kupfer (Chamber of Agriculture in Brandenburg, Germany) for providing the data on the cropping density of OSR in the sampling regions. Further, we are grateful to Régine Delourme (Institute for Genetics, Environment and Plant Protection, INRA, Rennes, France) for providing the seeds of the Brassica differential set.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Winter, M., Koopmann, B. Race spectra of Leptosphaeria maculans, the causal agent of blackleg disease of oilseed rape, in different geographic regions in northern Germany. Eur J Plant Pathol 145, 629–641 (2016). https://doi.org/10.1007/s10658-016-0932-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-0932-8