Abstract
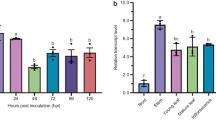
This study examined the role of indole-3-acetic acid (IAA) during the early stage of infection of rapeseed (Brassica napus L.) by the obligate biotrophic protist Plasmodiophora brassicae Woron., which causes clubroot disease. Treatment of infected B. napus seedlings with exogenous IAA promoted the development of clubroot disease, including an increase in the number and size of galls, whereas treatment with N-1-naphthylphthalamic acid (NPA) attenuated the disease and altered the location and size of the root nodules, resulting in a reduced disease index. An increased level of free IAA was detected in the roots at 3 and 7 days after inoculation (DAI) compared with the control, suggesting the activation of IAA signaling in the early stage of P. brassicae infection. Moreover, IAA treatment caused no significant change in the germination rate of the resting spores but resulted in enhanced root hair infection. RT–PCR and quantitative real-time PCR (qPCR) were used to analyze the expression of IAA homeostasis-related genes (3, 7, and 10 DAI) to identify gene expression patterns in the roots and leaves of B. napus after inoculation. Five IAA biosynthesis-related genes examined in this study were induced in the roots after inoculation with P. brassicae but not in the leaves. The rapid induction of BnAAO4 expression at 3 DAI may be responsible for the overproduction of IAA during the early stages of infection. This study suggests that IAA acts as a signaling molecule that putatively stimulates root hair infection in the early response of B. napus to P. brassicae infection.





Similar content being viewed by others
References
Ando, S., Tsushima, S., Tagiri, A., Kamachi, S., Konagaya, K., Hagio, T., et al. (2006). Increase in BrAO1 gene expression and aldehyde oxidase activity during clubroot development in Chinese cabbage (Brassica rapa L.). Molecular Plant Pathology, 7, 223–234.
Ando, S., Tsushima, S., Kamachi, S., Konagaya, K., & Tabei, Y. (2008). Alternative transcription initiation of the nitrilase gene (BrNIT2) caused by infection with Plasmodiophora brassicae Woron. in Chinese cabbage (Brassica rapa L.). Plant Molecular Biology, 68, 557–569.
Asano, T., Kageyama, K., & Hyakumachi, M. (1999). Surface disinfestation of resting spores of Plasmodiophora brassicae used to infect hairy roots of Brassica spp. Phytopathology, 89, 314–319.
Bari, R., & Jones, J. D. (2009). Role of plant hormones in plant defence responses. Plant Molecular Biology, 69, 473–488.
Cao, T., Srivastava, S., Rahman, M. H., Kav, N. N. V., Hotte, N., Deyholos, M. K., et al. (2008). Proteome-level changes in the roots of Brassica napus as a result of Plasmodiophora brassicae infection. Plant Science, 174, 97–115.
Castlebury, L. A., Maddox, J. V., & Glawe, D. A. (1994). A technique for the extraction and purification of viable Plasmodiophora brassicae resting spores from host root tissue. Mycologia, 86, 13.
Deora, A., Gossen, B. D., & Mcdonald, M. R. (2012). Effect of host resistance on infection by Plasmodiophora brassicae in canola. Canadian Journal of Plant Pathology, 34, 329–330.
Devos, S., & Prinsen, E. (2006). Plant hormones: a key in clubroot development. Communications of Agricultural &Appied Biological Science, 71, 869–872.
Devos, S., Vissenberg, K., Verbelen, J. P., & Prinsen, E. (2005). Infection of Chinese cabbage by Plasmodiophora brassicae leads to a stimulation of plant growth: impacts on cell wall metabolism and hormone balance. New Phytologist, 166, 241–250.
Devos, S., Laukens, K., Deckers, P., Van Der Straeten, D., Beeckman, T., Inze, D., et al. (2006). A hormone and proteome approach to picturing the initial metabolic events during Plasmodiophora brassicae infection on Arabidopsis. Molecular Plant Microbe Interaction, 19, 1431–1443.
Ding, X., Cao, Y., Huang, L., Zhao, J., Xu, C., Li, X., et al. (2008). Activation of the indole-3-acetic acid-amido synthetase GH3-8 suppresses expansin expression and promotes salicylate- and jasmonate-independent basal immunity in rice. Plant Cell, 20, 228–240.
Dixon, G. R. (2009). The occurrence and economic impact of Plasmodiophora brassicae and clubroot disease. Journal of Plant Growth Regulation, 28, 194–202.
Donald, E. C., Jaudzems, G., & Porter, I. J. (2008). Pathology of cortical invasion by Plasmodiophora brassicae in clubroot resistant and susceptible Brassica oleracea hosts. Plant Pathology, 57, 201–209.
Feng, J., Hwang, S. F., & Strelkov, S. E. (2013). Studies into primary and secondary infection processes by Plasmodiophora brassicae on canola. Plant Pathology, 62, 177–183.
Friberg, H., Lagerlof, J., & Ramert, B. (2005). Germination of Plasmodiophora brassicae resting spores stimulated by a non-host plant. European Journal of Plant Pathology, 113, 275–281.
Grsic, S., Kirchheim, B., Pieper, K., Fritsch, M., Hilgenberg, W., & Ludwig-Muller, J. (1999). Induction of auxin biosynthetic enzymes by jasmonic acid and in clubroot diseased Chinese cabbage plants. Physiologia Plantarum, 105, 521–531.
Grsic-Rausch, S., Kobelt, P., Siemens, J. M., Bischoff, M., & Ludwig-Muller, J. (2000). Expression and localization of nitrilase during symptom development of the clubroot disease in Arabidopsis. Plant Physiology, 122, 369–378.
Hayashi, K. (2012). The interaction and integration of auxin signaling components. Plant Cell Physiology, 53, 965–975.
Hwang, S. F., Ahmed, H. U., Zhou, Q., Strelkov, S. E., Gossen, B. D., Peng, G., et al. (2011). Influence of cultivar resistance and inoculum density on root hair infection of canola (Brassica napus) by Plasmodiophora brassicae. Plant Pathology, 60, 820–829.
Hwang, S. F., Ahmed, H. U., Zhou, Q., Strelkov, S. E., Gossen, B. D., Peng, G., et al. (2012). Assessment of the impact of resistant and susceptible canola on Plasmodiophora brassicae inoculum potential. Plant Pathology, 61, 945–952.
Ji, H. W., Ren, L., Chen, K. R., Xu, L., Liu, F., Sun, C. C., et al. (2013). Identification of physiological races of clubroot and resistance of rape cultivars to Plasmodiophora brassicae. Chinese Journal of Crop Science, 35, 301–306.
Jubault, M., Lariagon, C., Taconnat, L., Renou, J. P., Gravot, A., Delourme, R., et al. (2013). Partial resistance to clubroot in Arabidopsis is based on changes in the host primary metabolism and targeted cell division and expansion capacity. Functional & Integrative Genomics, 13, 191–205.
Kageyama, K., & Asano, T. (2009). Life cycle of Plasmodiophora brassicae. Journal of Plant Growth Regulation, 28, 203–211.
Kavanagh, J. A., & Williams, P. H. (1981). Indole auxins in Plasmodiophora infected cabbage roots and hypocotyls. Transactions of the British Mycological Society, 77, 125–129.
Kim, J. I., Murphy, A. S., Baek, D., Lee, S. W., Yun, D. J., Bressan, R. A., et al. (2011). YUCCA6 over-expression demonstrates auxin function in delaying leaf senescence in Arabidopsis thaliana. Journal of Experimental Botany, 62, 3981–3992.
Li, Y. H., Wei, F., Dong, X. Y., Peng, J. H., Liu, S. Y., & Chen, H. (2011). Simultaneous analysis of multiple endogenous plant hormones in leaf tissue of oilseed rape by solid-phase extraction coupled with high-performance liquid chromatography-electrospray ionisation tandem mass spectrometry. Phytochemical Analysis, 22, 442–449.
Ludwig-Muller, J. (2009a). Glucosinolates and the clubroot disease: defense compounds or auxin precursors? Phytochemistry Reviews, 8, 135–148.
Ludwig-Muller, J. (2009b). Plant defence - what can we learn from clubroots? Australasian Journal of Plant Pathology, 38, 318–324.
Ludwig-Muller, J., & Schuller, A. (2008). What can we learn from clubroots: alterations in host roots and hormone homeostasis caused by Plasmodiophora brassicae. European Journal of Plant Pathology, 121, 291–302.
LudwigMuller, J., Epstein, E., & Hilgenberg, W. (1996). Auxin-conjugate hydrolysis in Chinese cabbage: characterization of an amidohydrolase and its role during infection with clubroot disease. Physiologia Plantarum, 97, 627–634.
Ludwig-Muller, J., Pieper, K., Ruppel, M., Cohen, J. D., Epstein, E., Kiddle, G., et al. (1999). Indole glucosinolate and auxin biosynthesis in Arabidopsis thaliana (L.) Heynh. glucosinolate mutants and the development of clubroot disease. Planta, 208, 409–419.
Ludwig-Muller, J., Prinsen, E., Rolfe, S. A., & Scholes, J. D. (2009). Metabolism and plant hormone action during clubroot disease. Journal of Plant Growth Regulation, 28, 229–244.
Luo, H. C., Chen, G. K., Liu, C. P., Huang, Y., & Xiao, C. G. (2014). An improved culture solution technique for Plasmodiophora brassicae infection and the dynamic infection in the root hair. Australasian Journal of Plant Pathology, 43, 53–60.
Mashiguchi, K., Tanaka, K., Sakai, T., Sugawara, S., Kawaide, H., Natsume, M., et al. (2011). The main auxin biosynthesis pathway in Arabidopsis. Proceedings of the National Academy of Sciences of the United States of America, 108, 18512–18517.
Neuhaus, K., Grsic-Rausch, S., Sauerteig, S., & Ludwig-Muller, J. (2000). Arabidopsis plants transformed with nitrilase 1 or 2 in antisense direction are delayed in clubroot development. Journal of Plant Physiology, 156, 756–761.
Niwa, R., Nomura, Y., Osaki, M., & Ezawa, T. (2008). Suppression of clubroot disease under neutral pH caused by inhibition of spore germination of Plasmodiophora brassicae in the rhizosphere. Plant Pathology, 57, 445–452.
Park, J. E., Park, J. Y., Kim, Y. S., Staswick, P. E., Jeon, J., Yun, J., et al. (2007). GH3-mediated auxin homeostasis links growth regulation with stress adaptation response in Arabidopsis. Journal of Biological Chemistry, 282, 10036–10046.
Rashid, A., Ahmed, H. U., Xiao, Q., Hwang, S. F., & Strelkov, S. E. (2013). Effects of root exudates and pH on Plasmodiophora brassicae resting spore germination and infection of canola (Brassica napus L.) root hairs. Crop Protection, 48, 16–23.
Ren, L., Jia, J. G., Li, M., Liu, F., Cheng, Y. G., Chen, K. R., et al. (2012). Distribution of rapeseed clubroot disease in Hubei Province and evaluation of yield loss. Agricultural of Science & Technology, 13, 775–777.
Rieu, I., & Powers, S. J. (2009). Real-time quantitative RT-PCR: design, calculations, and statistics. Plant Cell, 21, 1031–1033.
Saini, S., Sharma, I., Kaur, N., & Pati, P. K. (2013). Auxin: a master regulator in plant root development. Plant Cell Report, 32, 741–757.
Schuller, A., & Ludwig-Muller, J. (2006). A family of auxin conjugate hydrolases from Brassica rapa: characterization and expression during clubroot disease. New Phytologist, 171, 145–157.
Searle, L. M., Chamberlain, K., Rausch, T., & Butcher, D. N. (1982). The conversion of 3-indolemethylglucosinolate to 3-indoleacetonitrile by myrosinase and its relevance to the clubroot disease of the cruciferae. Journal of Experimental Botany, 33, 935–942.
Seo, M., & Koshiba, T. (2002). Complex regulation of ABA biosynthesis in plants. Trends of Plant Science, 7, 41–48.
Seo, M., Aoki, H., Koiwai, H., Kamiya, Y., Nambara, E., & Koshiba, T. (2004). Comparative studies on the Arabidopsis aldehyde oxidase (AAO) gene family revealed a major role of AAO3 in ABA biosynthesis in seeds. Plant and Cell Physiology, 45, 1694–1703.
Siemens, J., Keller, I., Sarx, J., Kunz, S., Schuller, A., Nagel, W., et al. (2006). Transcriptome analysis of Arabidopsis clubroots indicate a key role for cytokinins in disease development. Molecular Plant Microbe Interaction, 19, 480–494.
Siemens, J., Graf, H., Bulman, S., In, O., & Ludwig-Muller, J. (2009). Monitoring expression of selected Plasmodiophora brassicae genes during clubroot development in Arabidopsis thaliana. Plant Pathology, 58, 130–136.
Sundelin, T., Jensen, D. F., & Lubeck, M. (2011). Identification of expressed genes during infection of Chinese cabbage (Brassica rapa subsp. pekinensis) by Plasmodiophora brassicae. Journal of Eukaryotic Microbiology, 58, 310–314.
Vandenbussche, F., Smalle, J., Le, J., Saibo, N. J., De Paepe, A., Chaerle, L., et al. (2003). The Arabidopsis mutant alh1 illustrates a cross talk between ethylene and auxin. Plant Physiology, 131, 1228–1238.
Williams, P. H. (1966). A system for the determination of races of Plasmodiophora brassicae that infect cabbage and rutabaga. Phytopathology, 56, 624–626.
Acknowledgments
Financial support from the National Project for Agricultural Technology System (CARS-13) and the Natural Science Foundation of China grants (31401720 and 31501617) is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, L., Ren, L., Chen, K. et al. Putative role of IAA during the early response of Brassica napus L. to Plasmodiophora brassicae . Eur J Plant Pathol 145, 601–613 (2016). https://doi.org/10.1007/s10658-016-0877-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-0877-y