Abstract
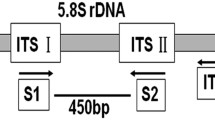
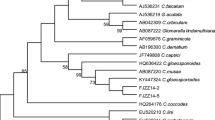
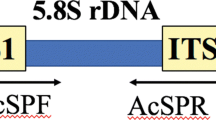
Mycosphaerella nawae is the causal agent of circular leaf spot of persimmon. A polymerase chain reaction (PCR) based protocol was developed for M. nawae-specific identification from pure culture, or infected symptomatic and asymptomatic persimmon tissues. Variation among the internal transcribed spacer regions (ITS) of the ribosomal DNA (rDNA) sequences of potentially related fungal species in persimmon orchards was analyzed for a primer pair design. Specificity was confirmed using multiple isolates of these species, other fungal pathogens that cause foliar diseases in persimmon and contaminants commonly obtained in the isolation process. The detection threshold for M. nawae DNA was lowered from 50 pg to 500 fg when nested-PCR was evaluated instead of single PCR. The nested-PCR protocol developed in this study showed its suitability to be applied for the specific detection of M. nawae from three types of naturally infected persimmon tissues: from lesions in fresh leaves, from pseudothecia present in lesions in leaf litter, and from infected asymptomatic leaves. The protocol can be useful for routine diagnosis, disease monitoring programs and for epidemiological research.


Similar content being viewed by others
References
Alaniz, S., Armengol, J., García-Jiménez, J., Abad-Campos, P., & León, M. (2009). A Multiplex PCR system for the specific detection of Cylindrocarpon liriodendri, C. macrodidymum, and C. pauciseptatum from grapevine. Plant Disease, 93, 821–825.
Arnal, L., & del Río, M. A. (2003). Removing astringency by carbon dioxide and nitrogen-enriched atmospheres in persimmon fruit cv. Rojo Brillante. Journal of Food Science, 68, 1516–1518.
Berbegal, M., Armengol, J., & García-Jiménez, J. (2011). Evaluation of fungicides to control circular leaf spot of persimmon caused by Mycosphaerella nawae. Crop Protection, 30, 1461–1468.
Berbegal, M., Pérez-Sierra, A., Armengol, J., Park, C. S., & García-Jiménez, J. (2010). First report of circular leaf spot of persimmon caused by Mycosphaerella nawae in Spain. Plant Disease, 94, 374.
Crous, P. W., Hong, J. W., Wingfield, B. D., & Wingfield, M. J. (2001). ITS rDNA phylogeny of selected Mycosphaerella species and their anamorphs occurring on Myrtaceae. Mycological Research, 105, 425–431.
David, J. C. (2000). Pseudocercospora kaki. IMI descriptions of fungi and bacteria: 144, sheet 1439. Wallingford: CAB International.
Food and Agriculture Organization of the United Nations (FAO) (2009). Crop production database FAOSTAT. http://faostat3.fao.org/home/index.html. Accessed 14 Sept 2012
Gardes, M., & Bruns, T. D. (1993). ITS primers with enhanced specificity for basidiomycetes-applications to the identification of mycorrhizae and rusts. Molecular Ecology, 2, 113–118.
Ikata, S., & Hitomi, T. (1929). Studies on circular leaf spot of persimmon caused by Mycosphaerella nawae. Special Bulletin of the Okayama Prefecture Agricultural Experiment Station, 33, 1–36 (In Japanese).
Johanson, A., & Jeger, M. J. (1993). Use of PCR for detection of Mycosphaerella fijiensis and M. musicola, the causal agents of Sigatoka leaf spots in banana and plantain. Mycological Research, 97, 670–674.
Kang, S. W., Kwon, J. H., & Kim, H. K. (1997). High sporulating medium for Cercospora kaki causing persimmon angular leaf spot. Korean Journal of Plant Pathology, 13, 69–71.
Kang, S. W., Kwon, J. H., Lee, Y. S., & Park, C. S. (1993). Effects of meteorological factors on perithecial formation and release of ascospores of Mycosphaerella nawae from the overwintered persimmon. Rural Development Administration Journal of Agricultural Science, 35, 337–343 (In Korean).
Kularatne, H. A. G., Lawrie, A. C., Barber, P. A., & Keane, P. J. (2004). A specific primer PCR and RFLP assay for the rapid detection and differentiation in planta of some Mycosphaerella species associated with foliar diseases of Eucalyptus globulus. Mycological Research, 108, 1476–1493.
Kwon, J. H., Jeong, S. G., & Chung, B. K. (2007). Survey of overwintering potential of anthracnose of sweet persimmon caused by Colletotrichum gloeosporioides. Research in Plant Disease, 13, 204–206 (In Korean).
Kwon, J. H., Kang, S. W., Park, C. S., & Kim, H. K. (1998). Identification of the imperfect stage of Mycosphaerella nawae causing circular leaf spot of persimmon in Korea. Korean Journal of Plant Pathology, 14, 397–401.
Kwon, J. H., & Park, C. S. (2004). Ecology of disease outbreak of circular leaf spot of persimmon and inoculum dynamics of Mycosphaerella nawae. Research in Plant Disease, 10, 209–216 (In Korean).
Nieto-Feliner, G., & Roselló, J. A. (2007). Better the devil you know? Guidelines for insightful utilization of nrDNA ITS in species-level evolutionary studies in plants. Molecular Phylogenetics and Evolution, 44, 911–919.
Thompson, J. D., Higgins, D. G., & Gibson, T. J. (1994). CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Research, 22, 4673–4680.
Truett, G. E., Heeger, P., Mynatt, R. L., Truett, A. A., Walker, J. A., & Warman, M. L. (2000). Preparation of PCR-quality mouse genomic DNA with hot sodium hydroxide and Tris (HotSHOT). Biotechniques, 29, 52–54.
Tuset, J. J., Hinarejos, C., & Mira, J. L. (1999). First report of leaf blight on sweet persimmon tree by Pestalotiopsis theae in Spain. Plant Disease, 83, 11.
Vicent, A., Bassimba, D. D. M., Hinarejos, C., & Mira, J. L. (2012). Inoculum and disease dynamics of circular leaf spot of persimmon caused by Mycosphaerella nawae under semi-arid conditions. European Journal of Plant Pathology, 134, 289–299.
Vicent, A., Bassimba, D. D. M., & Intrigliolo, D. S. (2011). Effects of temperature, water regime and irrigation on the release of ascospores of Mycosphaerella nawae, causal agent of circular leaf spot of persimmon. Plant Pathology, 60, 890–908.
White, T. J., Burns, T., Lee, S., & Taylor, J. W. (1990). Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetic. In M. A. Innis, D. H. Gelfand, J. J. Sninsky, & T. J. White (Eds.), PCR protocols, a guide to methods and applications (pp. 315–322). San Diego: Academics Press.
Acknowledgements
M. Berbegal was a contract holder of the “Campus de Excelencia Internacional” program of the Universitat Politècnica de Valencia. This research was financially supported by Fundación Agroalimed (Consellería de Agricultura, Pesca i Alimentació, Generalitat Valenciana). We thank J. Armengol for critically reading the manuscript prior to submission.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berbegal, M., Mora-Sala, B. & García-Jiménez, J. A nested-polymerase chain reaction protocol for the detection of Mycosphaerella nawae in persimmon. Eur J Plant Pathol 137, 273–281 (2013). https://doi.org/10.1007/s10658-013-0237-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-013-0237-0