Abstract
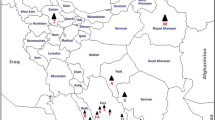
Symptoms reminiscent of phytoplasma infection were observed in four provinces (governorates) of Egypt in fields of eggplants, tomato plants and squash. Diseased plants exhibited stunting, leaf yellows and flower development abnormalities. PCR amplification of 16SrDNA with phytoplasma-specific primer pairs confirmed the phytoplasma presence. Sequencing and phylogenetic analysis indicated that all phytoplasmas had the same partial 16SrDNA sequence, assigning them to the 16SrII-D phytoplasma subgroup. Disease incidence was about 1% among the 20 squash fields surveyed and equally varied from 4% to 15% in the 20 eggplant fields and in the 40 tomato fields inspected. The widespread distribution of this phytoplasma in annual solanaceous and cucurbit crops suggests a wider plant host range including wild plants that could act as reservoir and insist on the need for a insect vector survey. A finer genetic differentiation of Egyptian 16SrII-D phytoplasma strains from different geographical origins and different host plants should help to better trace such epidemics.


Similar content being viewed by others
References
Al-Subhi, A. M., Al-Saady, N. A., Khan, A. J., & Deadman, M. L. (2011). First Report of a Group 16SrII Phytoplasma Associated with Witches'-Broom of Eggplant in Oman. Plant Disease, 95, 360.
Altschul, S. F., Madden, T. L., AA, Schaffer, Zhang, J., Zhang, Z., Miller, W., & Lipman, D. J. (1997). Gapped BLAST (Basic Local Alignment Search Tool) and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Research, 25, 3389–3402.
Amaral-Mello, A.P.O., Eckstein, B., Flôres, D., Kreyci, P.F., and Bedendo, I.P. (2010) Identification by computer-simulated RFLP of phytoplasmas associated with eggplant giant calyx representative of two subgroups, a lineage of 16SrIII-J and the new subgroup 16SrIII-U International Journal of Systematic and Evolutionary Microbiology In press.
Anfoka, G. H. A., Khalil, A. B., & Fattash, I. (2003). Detection and molecular characterization of a phytoplasma associated with big bud disease of tomatoes in Jordan. Journal of Phytopathology, 151, 223–227.
Arocha, Y., Antesana, O., Montellano, E., Franco, P., Plata, G., & Jones, P. (2007). 'Candidatus Phytoplasma lycopersici', a phytoplasma associated with 'hoja de perejil' disease in Bolivia. International Journal of Systematic and Evolutionary Microbiology, 57, 1704–1710.
Blancard, D. (2009) Les maladies de la tomate: Quae Editions, France.
Boiteux, L. S., Lima, M. I., & Kitajima, E. W. (1994). Giant calyx - a disease of eggplant (Solanum melongena) associated with a mycoplasma-like organism in Brazil. Plant Pathology, 43, 751–754.
Bonfield, J. K., Smith, K. F., & Staden, R. (1995). A new DNA sequence assembly program. Nucleic Acids Research, 24, 4992–4999.
Choueiri, E., Salar, P., Jreijiri, F., El-Zammar, S., Massaad, R., Abdul-Nour, H., et al. (2007). Occurrence and distribution of 'Candidatus Phytoplasma trifolii' associated with diseases of solanaceous crops in Lebanon. European Journal of Plant Pathology, 118, 411–416.
Cousin, M. T., & Abadie, M. (1982). Action des mycoplasmes sur l’anthère. Etude en microscopies photonique et électronique. Revue de Cytologie, de Biologie Végétale et de Botanique, 5, 41–57.
Cousin, M. T., Dafalla, G., Demazeau, E., Theveu, E., & Grosclaude, J. (1989). In situ detection of MLOs for Solanaceae stolbur and faba bean phyllody by indirect immunofluorescence. Journal of Phytopathology, 124, 71–79.
Davis, R. I., Schneider, B., & Gibb, K. (1997). Detection and differentiation of phytoplasmas in Australia. Australian Journal of Agricultural Research, 48, 535–544.
Deng, S. J., & Hiruki, C. (1991). Amplification of 16S ribosomal-RNA genes from culturable and nonculturable mollicutes. Journal of Microbiological Methods, 14, 53–61.
El-Banna, M. Om-Hashem & El-Deep, S.H. (2007). Phytoplasma associated with Mango malformation disease in Egypt. Egyptian Journal of Phytopathology, 35, 141–153.
Firrao, G., Andersen, M., Bertaccini, A., Boudon Padieu, E., Bové, J. M., Daire, X., et al. (2004). 'Candidatus Phytoplasma', a taxon for the wall-less, non-helical prokaryotes that colonize plant phloem and insects. International Journal of Systematic and Evolutionary Microbiology, 54, 1243–1255.
Garnier, M. (2000). The stolbur phytoplasma: an ubiquitous agent. Comptes Rendus de l'Academie d'Agriculture de France, 86, 27–33.
Gundersen, D. E., & Lee, I.-M. (1996). Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopathologia Mediterranea, 35, 114–151.
Gundersen, D. E., Lee, I. M., Rehner, S. A., Davis, R. E., & Kingsbury, D. T. (1994). Phylogeny of mycoplasmalike organisms (phytoplasmas): a basis for their classification. Journal of Bacteriology, 176, 5244–5254.
Hiruki, C., & Wang, K. (2004). Clover proliferation phytoplasma: ‘Candidatus Phytoplasma trifolii’. International Journal of Systematic and Evolutionary Microbiology, 54, 1349–1353.
Jarausch, W., Jarausch-Wehrheim, B., Danet, J. L., Broquaire, J. M., Dosba, F., Saillard, C., & Garnier, M. (2001). Detection and identification of European stone fruit yellows and other phytoplasmas in wild plants in the surroundings of apricot chlorotic leaf roll-affected orchards in southern France. European Journal of Plant Pathology, 107, 209–217.
Kelly, P.L., Arocha, Y., and Dider, S.Z. (2009) First report of a 16SrI, ‘Candidatus Phytoplasma asteris’ isolate affecting eggplant and Mikania sp. in Bangladesh. New Disease Reports 18: 52.
Khan, A. J., Botti, S., Al-Subhi, A. M., Gundersen-Rindal, D. E., & Bertaccini, A. F. (2002). Molecular identification of a new phytoplasma associated with alfalfa witches'-broom in Oman. Phytopathology, 92, 1038–1047.
Lee, I. M., & Davis, R. E. (1992). Mycoplasmas which infect plants and insects. In J. Maniloff, R.N. McElhaney, L.R. Finch & J.B. Baseman (Eds.), Mycoplasmas: Molecular Biology and Pathogenesis (pp. 379–390). American Society of Microbiology: Washington, D.C.
Lee, L. M., Hammond, R. W., Davis, R. E., & Gundersen, D. E. (1993). Universal amplification and analysis of pathogen 16S rDNA for classification and identification of mycoplasmalike organisms. Phytopathology, 83, 834–842.
Lee, I. M., Davis, R. E., & Gundersen-Rindal, D. E. (2000). Phytoplasma: phytopathogenic mollicutes. Annual Review of Microbiology, 54, 221–255.
Lee, I. M., Zhao, Y., Davis, R. E., Wei, W., & Martini, M. (2007). Prospects of DNA-based systems for differenciation and classification of phytoplasmas. Bulletin of Insectology, 60, 239–244.
Maixner, M., Ahrens, U., & Seemuller, E. (1995). Detection of the German grapevine yellows (Vergilbungskrankheit) MLO in grapevine, alternative hosts and a vector by a specific PCR procedure. European Journal of Plant Pathology, 101, 241–250.
McCoy, R. E., Caudwell, A., Chang, C. J., Chen, T. A., Chiykowski, L. N., Cousin, M. T., et al. (1989). Plant diseases associated with mycoplasma-like organisms. In R. F. Whitcomb & J. G. Tully (Eds.), The Mycoplasmas (pp. 545–640). New York: Academic.
Mitra, D. K. (1988). Little leaf disease of eggplant. In K. Maramorosch & S. P. Raychaudhuri (Eds.), Mycoplasma disease of crops (pp. 343–348). New York: Springer.
Montano, H. G., Davis, R. E., Dally, E. L., Pimentel, J. P., & Brioso, P. S. T. (2000). Identification and phylogenetic analysis of a new phytoplasma from diseased chayote in Brazil. Plant Disease, 84, 429–436.
Montano, H. G., Davis, R. E., Dally, E. L., Hogenhout, S., Pimentel, J. P., & Brioso, P. S. (2001). 'Candidatus Phytoplasma brasiliense', a new phytoplasma taxon associated with hibiscus witches' broom disease. International Journal of Systematic & Evolutionary Microbiology, 51, 1109–1118.
Montano, H. G., Brioso, P. S. T., Pimentel, J. P., Figueiredo, D. V., & Cunha, J. O. (2006). Cucurbita moschata, new phytoplasma host in Brazil. Journal of Plant Pathology, 88, 226–226.
Montano, H. G., Paulo, S. T. B., Roberta, C. P., & Joao, P. P. (2007). Sicana odorifera (Cucurbitaceae) a new phytoplasma host. Bulletin of Insectology, 60, 287–288.
Omar, A. F., Emeran, A. A., & Abass, J. M. (2008). Detection of Phytoplasma associated with periwinkle virescence in Egypt. Plant Pathology Journal, 7, 92–97.
Pearce, T., Scott, J., and Pethybridge, S.J. (2011) First Report of a 16SrII-D Subgroup Phytoplasma Associated with Pale Purple Coneflower Witchs’-Broom Disease in Australia. Plant Disease in press.
Pracros, P., Renaudin, J., Eveillard, S., Mouras, A., & Hernould, M. (2006). Tomato flower abnormalities induced by stolbur phytoplasma infection are associated with changes of expression of floral development genes. Molecular Plant-Microbe Interactions, 19, 62–68.
Saeed, E. M., Roux, J., & Cousin, M. T. (1993). Studies of polyclonal antibodies for the detection of MLOs associated with faba bean (Vicia faba L) using different Elisa methods and dot-blot. Journal of Phytopathology-Phytopathologische Zeitschrift, 137, 33–43.
Saeed, E., Seemüller, E., Schneider, B., Saillard, C., Blanchard, B., Bertheau, Y., & Cousin, M. T. (1994). Molecular-cloning, detection of chromosomal DNA of the mycoplasmalike organism (MLO) associated with faba bean (Vicia-faba L) phyllody by southern blot hybridization and the polymerase chain-reaction (PCR). Journal of Phytopathology-Phytopathologische Zeitschrift, 142, 97–106.
Schneider, B., Seemüller, E., Smart, C. D., & Kirkpatrick, B. C. (1995a). Phylogenetic classification of plant pathogenic mycoplasmalike organisms or phytoplasmas. In S. Razin & J. G. Tully (Eds.), Molecular and diagnostic procedures in mycoplasmology (pp. 369–380). San Diego, CA: Academic.
Schneider, B., Cousins, M. T., Klinkong, S., & Seemüller, E. (1995b). Taxonomic Relatedness and Phylogenetic Positions of Phytoplasmas Associated with Diseases of Faba Bean, Sunnhemp, Sesame, Soybean, and Eggplant. Journal of Plant Diseases and Protection, 102, 225–232.
Schneider, B., Padovan, A., De la Rue, S., Eichner, R., Davis, R., Bernuetz, A., & Gibb, K. (1999). Detection and differentiation of phytoplasmas in Australia: an update. Australian Journal of Agricultural Research, 50, 333–342.
Seemüller, E., Schneider, B., Mäurer, R., Ahrens, U., Daire, X., Kison, H., et al. (1994). Phylogenetic classification of phytopathogenic mollicutes by sequence analysis of 16S ribosomal DNA. International Journal of Systematic Bacteriology, 44, 440–446.
Sertkaya, G., Martini, M., Musetti, R., & Osler, R. (2007). Detection and molecular characterization of phytoplasmas infecting sesame and solanaceous crops in Turkey. Bulletin of Insectology, 60, 141–142.
Shaw, M. E., Kirkpatrick, B. C., & Golino, D. A. (1993). The beet leafhopper-transmitted virescence agent causes tomato big bud disease in California. Plant Disease, 77, 290–295.
Siddique, A. B. M., Agrawal, G. K., Alam, N., & Krishna Reddy, M. (2001). Electron microscopy and molecular characterization of phytoplasmas associated with little leaf disease of brinjal (Solanum melongena L.) and periwinkle (Catharanthus roseus) in Bangladesh. Journal of Phytopathology, 149, 237–244.
Streten, C., Conde, B., Herrington, M., Moulden, J., & Gibb, K. (2005). “Candidatus Phytoplasma australiense” is associated with pumpkin yellow leaf curl disease in Queensland, Western Australia and the Northern territory. Australian Plant Pathology, 34, 103–105.
Tamura, K., Dudley, J., Nei, M., & Kumar, S. (2007). MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Molecular Biology and Evolution, 24, 1596–1599.
Thompson, J. D., Higgins, D. G., & Gibson, T. J. (1994). CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Research, 22, 4673–4680.
Wei, W., Davis, R. E., Lee, I. M., & Zhao, Y. (2007). Computer simulated RFLP analysis of 16S rRNA genes: identification of ten new phytoplasma groups. International Journal of Systematic & Evolutionary Microbiology, 57, 1855–1867.
Weintraub, P. G., & Beanland, L. (2006). Insect vectors of phytoplasmas. Annual Review of Entomology, 51, 91–111.
White, D. T., Billington, S. J., Walsh, K. B., & Scott, P. T. (1997). DNA sequence analysis supports the association of phytoplasmas with papaya (Carica papaya) dieback, yellow crinkle and mosaic. Australasian Plant Pathology, 26, 28–36.
White, D. T., Blackall, L. L., Scott, P. T., & Walsh, K. B. (1998). Phylogenetic positions of phytoplasmas associated with dieback, yellow crinkle and mosaic diseases of papaya, and their proposed inclusion in 'Candidatus Phytoplasma australiense' and a new taxon', Candidatus Phytoplasma australasia'. International Journal of Systematic Bacteriology, 48, 941–951.
Zreik, L., Carle, P., Bové, J. M., & Garnier, M. (1995). Characterization of the mycoplasmalike organism associated with witches-broom disease of lime and proposition of a Candidatus taxon for the organism, Candidatus Phytoplasma aurantifolia. International Journal of Systematic Bacteriology, 45, 449–453.
Acknowledgements
This work was supported by the bilateral grant IMHOTEP number 20735PA from the Egyptian and French ministries of foreign affairs. We gratefully acknowledge Prof. Dr. Al-Shafie Ibrahim Al-shafie and Mr. Salem Hamdem for helping during surveys and Miss Shimaa A. Khalaf for assistance with DNA extraction.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Omar, A.F., Foissac, X. Occurrence and incidence of phytoplasmas of the 16SrII-D subgroup on solanaceous and cucurbit crops in Egypt. Eur J Plant Pathol 133, 353–360 (2012). https://doi.org/10.1007/s10658-011-9908-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-011-9908-x