Abstract
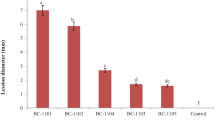
From 2003 to 2006, a total of 426 single-conidial isolates of B. cinerea collected from greenhouse vegetables in China were characterized for resistance to benzimidazole fungicides and diethofencarb according to inhibition of mycelial growth. Rapid development of double-resistance to benzimidazoles and diethofencarb was observed. Three types of benzimidazole-resistant isolates, Ben R1, Ben R2 and Ben R3 were detected. A new phenotype, Ben R3, which showed low level of resistance to benzimidazole fungicides and resistance to diethofencarb, was detected with frequencies of 6.8%, 10.0%, 13.2% and 12.4% from 2003 to 2006, respectively. Further studies indicated that Ben R3 was caused by a point mutation from GAG in sensitive(S) isolates to GTG at codon 198 in the β-tubulin gene, predicted to cause a change from glutamic acid to valine. Ben R3 isolates had comparable growth, sporulation and pathogenicity ability as isolates of other phenotypes but were more sensitive at lower temperatures.

Similar content being viewed by others
References
Albertini, C., Gredt, M., & Leroux, P. (1999). Mutations of the β-tubulin gene associated with different phenotypes of benzimidazole resistance in the cereal eyespot fungi Tapesia yallundae and Tapesia acuformis. Pesticide Biochemistry Physiology, 64, 17–31.
Beever, R. E., Laracy, E. P., & Park, H. A. (1989). Strain of Botrytis cinerea resistant to dicarboximide and benzimidazole fungicides in New Zealand vineyards. Plant Pathology, 38, 427–437.
Chen, C. J., Li, J., Qi, Z. Q., Wang, J. X., & Zhou, M. G. (2005). Cloning of α-tubulin gene from Fusarium graminearum and analyzing its relationship with carbendazim-resistance. Acta Microbiology Sinica, 45, 288–291.
Dianez, F., Santos, M., & Blanco, R. (2002). Fungicide resistance in Botrytis cinerea isolates from strawberry crops in Huelva (South-western Spain). Phytoparasitic, 30, 529–534.
Davidse, L. C. (1986). Benzimidazole fungicides: mechanism of action and biological impact. Annual Review of Phytopathology, 24, 43–65.
Davidson, R. M., Hanson, L. E., Franc, G. D., & Panella, L. (2006). Analysis of β-tubulin gene fragments from benzimidazole-sensitive and -tolerant Cercospora beticola. Journal of Phytopathology, 154, 321–328.
Elad, Y., Shabi, E., & Katan, T. (1988). Negative cross-resistance between benzimidazole and N-phenylcarbamate fungicides and control of Botrytis cinerea on grapes. Plant Pathology, 37, 141–147.
Elad, Y. (1992). Reduced sensitivity of Botrytis cinerea to two sterol biosynthesis-inhibiting fungicides: fenetrazole and fenethanil. Plant Pathology, 41, 47–54.
Kawchuk, L. M., Hutchison, L. J., Verhaeghe, C. A., Lynch, D. R., Bains, P. S., & Holly, J. D. (2002). Isolation of the β-tubulin gene and characterization of thiabendazole resistance in Gibberella pulicaris. Canada Journal of Plant Pathology, 24, 233–238.
Kachroo, T., Leong, S. A., & Chattoo, B. B. (1995). A rapid method of isolation of genomic DNA from filamentous fungi. International Rice Research Notes, 21, 2–3. 47.
Koenraadt, H., Somerville, S. C., & Jones, A. L. (1992). Characterization of mutations in the beta-tubulin gene of benomyl-resistant field strains of Venturia inaequalis and other plant pathogenic fungi. Phytopathology, 82, 1348–1354.
Leroux, P., Fritz, R., Debieu, D., Albertini, C., Lanen, C., Bach, J., et al. (2002). Mechanisms of resistance to fungicides in field strains of Botrytis cinerea. Pest Management Science, 58, 876–888.
Leroux, P. (1992). Negative cross-resistance in fungicides: from the laboratory to the field. In I. Denholm, A. L. Devonshire & D. W. Hollomon (Eds.), Resistance 9 : achievements and developments in combating pesticide resistance (pp. 179–189). London: Elsevier.
Ma, Z. H., Yoshimura, M. A., & Michailides, T. J. (2003). Identificantion and characterization of benzimidazole resistance in Monilinia fructicola from stone fruit orchards in California. Applied and Environmental Microbiology, 69, 7145–7152.
Ma, Z. H., Yoshimura, M. A., Holtz, B. A., & Michailides, T. J. (2005). Characterization and PCR-based detection of benzimidazole-resistant isolates of Monilinia laxa in California. Pest Management Science, 61, 449–457.
Maymon, M., Zveibil, A., Pivonia, S., Minz, D., & Freeman, S. (2006). Identification and characterization of benomyl-resistant and -sensitive populations of Colletotrichum gloeosporioides from Statice (Limonium spp.). Phytopathology, 96, 542–548.
McKay, G., Egan, J. D., Morris, E., & Brown, A. E. (1998). Identification of benzimidazole resistance in Cladobotryum dendroides using a PCR-based method. Mycological Research, 102, 671–676.
Noethover, J., & Matteoni, J. A. (1986). Resistance of Botrytis cinerea to benomyl and iprodione in vineyards and green houses after exposure to the fungicides along or mixed with captan. Plant Disease, 70, 398–402.
Pappas, A. C. (1997). Evolution of fungicide resistance in Botrytis cinerea in protected crops in Greece. Crop Protection, 16, 257–263.
Prins, T. W., Tudzynski, P., & Tiedemann, A. V. (2000). Infection strategies of Botrytis cinerea and related necrotrophic pathogens. In J. W. Kronstad (Ed.), Fungi pathology (pp. 33–64). Dordrecht: Kluwer Academic.
Rosslenbroich, H. J. & Stuebler, D. (2000). Botrytis cinerea-history of chemical control and novel fungicides for its management. Crop Protection, 19, 557–561.
Sholberg, P. L., Harlton, C., Haag, P., Lëvseque, C. A., Gorman, D. O., & Seifert, K. (2005). Benzimidazole and diphenylamine sensitivity and identity of Penicillium spp. that cause postharvest blue mold of apples using β-tubulin gene sequences. Postharvest Biology Technology, 36, 41–49.
Yarden, O., & Katan, T. (1993). Mutations leading to substitutions at amino acids 198 and 200 of beta tubulin that correlate with benomyl-resistance phenotypes of field strains of Botrytis cinerea. Phytopathology, 83, 1478–1483.
Zhang, C. Q., Yuan, S. K., Sun, H. Y., Qi, Z. Q., Zhou, M. G., & Zhu, G. N. (2007). Sensitivity of Botrytis cinerea from vegetable greenhouses to boscalid. Plant Pathology, 56, 646–653.
Zhang, C. Q., Zhang, Y., Wei, F. L., Liu, S. Y., & Zhu, G. N. (2006). Detection of resistance of Botryotinia fuckeliana from protected vegetables to different classes of fungicides. Chinese Journal of Pesticide Science, 8, 245–249.
Zhang, Y. J., Yan, X. Q., Han, J. C., & Liu, H. P. (2003). Detection of resistance to diethofencarb (NPC) in Botrytis cinerea Pers. Journal of Shanxi Agricultural University, 23, 308–311.
Zhou, M. G., Ye, Z. Y., & Liu, J. F. (1994). Advance in fungicide resistance in China. Journal of Nanjing Agricultural University, 17, 33–41.
Ziogas, B. N., & Girgis, S. M. (1993). Cross-resistance relationships between benzimidazole fungicides and diethofencarb in Botrytis cinerea and their genetical basis in Ustilago maydis. Pesticide Science, 39, 199–205.
Acknowledgements
This research was partially supported by grant no. Y3080042 of Zhejiang Natural Science Foundation and grant no. 2351000971 of Zhejiang Forest College funding.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, C.Q., Liu, Y.H. & Zhu, G.N. Detection and characterization of benzimidazole resistance of Botrytis cinerea in greenhouse vegetables. Eur J Plant Pathol 126, 509–515 (2010). https://doi.org/10.1007/s10658-009-9557-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-009-9557-5