Abstract
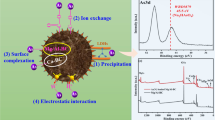
To develop a novel granular adsorbent to remove arsenic and antimony from water, calcined Mg/Al-layered double-hydroxide (CLDH)-incorporated polyethersulfone (PES) granular adsorbents (PES-LDH) were prepared using a core-shell method having 25% PES in an N,N-dimethylformamide solution. The PES-LDH displayed a spherical hollow shape having a rough surface and the average particle size of 1–2 mm. On the PES-LDH surface, nanosized CLDH (100–150 nm) was successfully immobilized by consolidation between PES and CLDH. The adsorption of Sb(V) by PES-LDH was found to be more favorable than for As(V), with the maximum adsorption capacity of As(V) and Sb(V) being 7.44 and 22.8 mg/g, respectively. The regeneration results indicated that a 0.5 M NaOH and 5 M NaCl mixed solution achieved an 80% regeneration efficiency in As(V) adsorption and desorption. However, the regeneration efficiency of Sb(V) gradually decreased due to its strong binding affinity, even though the PES-LDH showed much higher Sb(V) adsorption efficiency than As(V). This study suggested that PES-LDH could be a promising granular adsorbent for the remediation of As(V) and Sb(V) contained in wastewater.







Similar content being viewed by others
References
Anawar, H. M., Akai, J., Mostofa, K. M. G., Safiullah, S., & Tareq, S. M. (2001). Arsenic poisoning in groundwater: Health risk and geochemical sources in Bangladesh. Environment International, 27, 597–604. https://doi.org/10.1016/S0160-4120(01)00116-7.
Banerjee, K., Amy, G. L., Prevost, M., Nour, S., Jekel, M., Gallagher, P. M., et al. (2008). Kinetic and thermodynamic aspects of adsorption of arsenic onto granular ferric hydroxide (GFH). Water Research, 42(13), 3371–3378.
Belzile, N., Chen, Y., Filella, M., Belzile, N., & Chen, Y. (2002). Antimony in the environment: A review focused on natural waters II. Relevant solution chemistry. Earth-Science Reviews, 59(1–2), 265–285. https://doi.org/10.1016/S0012-8252(01)00070-8.
Carey, B. J., Daeneke, T., Nguyen, E. P., & Wang, Y. (2015). Two solvent grinding sonication method for the synthesis of two-dimensional tungsten disulphide flakes. Chemical Communications, 51, 3770–3773. https://doi.org/10.1039/C4CC08399G.
Chang, Q., Lin, W., & Ying, W. (2010). Preparation of iron-impregnated granular activated carbon for arsenic removal from drinking water. Journal of Hazardous Materials, 184(1–3), 515–522. https://doi.org/10.1016/j.jhazmat.2010.08.066.
Chanpiwat, P., Sthiannopkao, S., Cho, K. H., Kim, K.-W., San, V., Suvanthong, B., et al. (2011). Contamination by arsenic and other trace elements of tube-well water along the Mekong River in Lao PDR. Environmental pollution (Barking, Essex: 1987), 159(2), 567–576.
Chen, N., Zhang, Z., Feng, C., Sugiura, N., Li, M., & Chen, R. (2010). Fluoride removal from water by granular ceramic adsorption. Journal of colloid and interface science, 348(2), 579–584. https://doi.org/10.1016/j.jcis.2010.04.048
Chen, N., Zhang, Z., Feng, C., Li, M., Zhu, D., & Sugiura, N. (2011). Studies on fluoride adsorption of iron-impregnated granular ceramics from aqueous solution. Materials Chemistry and Physics, 125(1–2), 293–298. https://doi.org/10.1016/j.matchemphys.2010.09.037.
Cheng, X., Huang, X., Wang, X., & Sun, D. (2010). Influence of calcination on the adsorptive removal of phosphate by Zn–Al layered double hydroxides from excess sludge liquor. Journal of Hazardous Materials, 177(1–3), 516–523. https://doi.org/10.1016/j.jhazmat.2009.12.063.
Cui, H., Li, Q., Gao, S., & Shang, J. K. (2012). Strong adsorption of arsenic species by amorphous zirconium oxide nanoparticles. Journal of Industrial and Engineering Chemistry, 18(4), 1418–1427. https://doi.org/10.1016/j.jiec.2012.01.045.
Das, N. N., Konar, J., Mohanta, M. K., & Srivastava, S. C. (2004). Adsorption of Cr(VI) and Se(IV) from their aqueous solutions onto Zr4 + -substituted ZnAl/MgAl-layered double hydroxides: Effect of Zr 4+ substitution in the layer. Journal of Colloid and Interface Science, 270, 1–8. https://doi.org/10.1016/S0021-9797(03)00400-4.
Dong, L., Zinin, P. V., Cowen, J. P., & Ming, L. C. (2009). Iron coated pottery granules for arsenic removal from drinking water. Journal of Hazardous Materials, 168, 626–632. https://doi.org/10.1016/j.jhazmat.2009.02.168.
Dou, X., Mohan, D., & Pittman, C. U. (2013). Arsenate adsorption on three types of granular schwertmannite. Water Research, 47(9), 2938–2948. https://doi.org/10.1016/j.watres.2013.01.035.
Fan, H.-L., Shangguan, J., Liang, L.-T., Li, C.-H., & Lin, J.-Y. (2013). A comparative study of the effect of clay binders on iron oxide sorbent in the high-temperature removal of hydrogen sulfide. Process Safety and Environmental Protection, 91(3), 235–243. https://doi.org/10.1016/j.psep.2012.04.001.
Flores, R. G., Andersen, S. L. F., Maia, L. K. K., José, H. J., & Moreira, R. D. F. P. M. (2012). Recovery of iron oxides from acid mine drainage and their application as adsorbent or catalyst. Journal of Environmental Management, 111, 53–60. https://doi.org/10.1016/j.jenvman.2012.06.017.
Gebel, T. (1997). Arsenic and antimony: Comparative approach on mechanistic toxicology. Chemico-Biological Interactions, 107(3), 131–144. https://doi.org/10.1016/S0009-2797(97)00087-2.
Goh, K.-H., Lim, T.-T., & Dong, Z. (2008). Application of layered double hydroxides for removal of oxyanions: A review. Water Research, 42, 1343–1368.
Goh, K.-H., Lim, T.-T., & Dong, Z. (2009). Enhanced arsenic removal by hydrothermally treated nanocrystalline Mg/Al layered double hydroxide with nitrate intercalation. Environmental Science and Technology, 43, 2537–2543. https://doi.org/10.1021/es802811n.
Hanh, H. T., Kim, K.-W., Bang, S., & Hoa, N. M. (2011). Community exposure to arsenic in the Mekong river delta, Southern Vietnam. Journal of Environmental Monitoring, 13(7), 2025–2032. https://doi.org/10.1039/c1em10037h.
He, J., Bardelli, F., Gehin, A., Silvester, E., & Charlet, L. (2016). Novel chitosan goethite bionanocomposite beads for arsenic remediation. Water Research, 101, 1–9. https://doi.org/10.1016/j.watres.2016.05.032.
Kameda, T., Honda, M., & Yoshioka, T. (2011). Removal of antimonate ions and simultaneous formation of a brandholzite-like compound from magnesium–aluminum oxide. Separation and Purification Technology, 80(2), 235–239. https://doi.org/10.1016/j.seppur.2011.04.032.
Kanel, S. R., Manning, B., Charlet, L., & Choi, H. (2005). Removal of arsenic(III) from groundwater by nanoscale zero-valent iron. Environmental Science and Technology, 39(5), 1291–1298. https://doi.org/10.1021/es048991u.
Lalhmunsiama, Lalchhingpuii, Nautiyal, B. P., Tiwari, D., Choi, S. I., Kong, S.-H., & Lee, S.-M. (2016). Silane grafted chitosan for the efficient remediation of aquatic environment contaminated with arsenic(V). Journal of Colloid and Interface Science, 467, 203–212. https://doi.org/10.1016/j.jcis.2016.01.019.
Lee, H., Kim, D., Kim, J., Ji, M.-K., Han, Y.-S., Park, Y.-T., et al. (2015a). As(III) and As(V) removal from the aqueous phase via adsorption onto acid mine drainage sludge (AMDS) alginate beads and goethite alginate beads. Journal of Hazardous Materials, 292, 146–154. https://doi.org/10.1016/j.jhazmat.2015.03.026.
Lee, S.-H., Kim, K.-W., Choi, H., & Takahashi, Y. (2015b). Simultaneous photooxidation and sorptive removal of As(III) by TiO2 supported layered double hydroxide. Journal of Environmental Management, 161, 228–236. https://doi.org/10.1016/j.jenvman.2015.06.049.
Lin, T.-F., & Wu, J.-K. (2001). Adsorption of arsenite and arsenate within activated alumina grains: Equilibrium and kinetics. Water Research, 35(8), 2049–2057.
Liu, F., Le, X. C., McKnight-Whitford, A., Xia, Y., Wu, F., Elswick, E., et al. (2010). Antimony speciation and contamination of waters in the Xikuangshan antimony mining and smelting area, China. Environmental Geochemistry and Health, 32(5), 401–413. https://doi.org/10.1007/s10653-010-9284-z.
Liu, L., Shen, F., Zhang, B., Jiang, H., Li, J., Luo, J., et al. (2016). Fabrication of PES-based membranes with a high and stable desalination performance for membrane distillation. RSC Advances, 6(109), 107840–107850. https://doi.org/10.1039/c6ra22193a.
Lu, H., Zhu, Z., Zhang, H., Zhu, J., & Qiu, Y. (2015). Simultaneous removal of arsenate and antimonate in simulated and practical water samples by adsorption onto Zn/Fe layered double hydroxide. Chemical Engineering Journal, 276, 365–375. https://doi.org/10.1016/j.cej.2015.04.095.
Majzlan, J., Lalinská, B., Chovan, M., Bläß, U., Brecht, B., Göttlicher, J., et al. (2011). A mineralogical, geochemical, and microbiogical assessment of the antimony- and arsenic-rich neutral mine drainage tailings near Pezinok, Slovakia. American Mineralogist, 96(1), 1–13. https://doi.org/10.2138/am.2011.3556.
Mandal, B. K., & Suzuki, K. T. (2002). Arsenic round the world: A review. Talanta, 58(1), 201–235. https://doi.org/10.1016/S0039-9140(02)00268-0.
Mohan, D., & Pittman, C. U. (2007). Arsenic removal from water/wastewater using adsorbents—A critical review. Journal of Hazardous Materials, 142(1–2), 1–53.
Pan, Y. F., Chiou, C. T., & Lin, T. F. (2010). Adsorption of arsenic(V) by iron-oxide-coated diatomite (IOCD). Environmental Science and Pollution Research, 17(8), 1401–1410. https://doi.org/10.1007/s11356-010-0325-z.
Pan, B., Wu, J., Pan, B., Lv, L., Zhang, W., Xiao, L., et al. (2009). Development of polymer-based nanosized hydrated ferric oxides (HFOs) for enhanced phosphate removal from waste effluents. Water Research, 43(17), 4421–4429. https://doi.org/10.1016/j.watres.2009.06.055.
Parida, K., & Mohapatra, L. (2012). Recent progress in the development of carbonate-intercalated Zn/Cr LDH as a novel photocatalyst for hydrogen evolution aimed at the utilization of solar light. Dalton Transactions, 41(4), 1173–1178. https://doi.org/10.1039/C1DT10957J.
Pena, M. E., Korfiatis, G. P., Patel, M., Lippincott, L., & Meng, X. (2005). Adsorption of As(V) and As(III) by nanocrystalline titanium dioxide. Water Research, 39(11), 2327–2337. https://doi.org/10.1016/j.watres.2005.04.006.
Phan, K., Phan, S., Huoy, L., Suy, B., Wong, M. H., Hashim, J. H., et al. (2013). Assessing mixed trace elements in groundwater and their health risk of residents living in the Mekong River basin of Cambodia. Environmental Pollution, 182, 111–119. https://doi.org/10.1016/j.envpol.2013.07.002.
Rahimpour, A., & Madaeni, S. S. (2007). Polyethersulfone (PES)/cellulose acetate phthalate (CAP) blend ultrafiltration membranes: Preparation, morphology, performance and antifouling properties. Journal of Membrane Science, 305(1–2), 299–312. https://doi.org/10.1016/j.memsci.2007.08.030.
Ray, P. Z., & Shipley, H. J. (2015). Inorganic nano-adsorbents for the removal of heavy metals and arsenic: A review. RSC Advances, 5(38), 29885–29907. https://doi.org/10.1039/C5RA02714D.
Sadeghi, I., Aroujalian, A., Raisi, A., Dabir, B., & Fathizadeh, M. (2013). Surface modification of polyethersulfone ultrafiltration membranes by corona air plasma for separation of oil/water emulsions. Journal of Membrane Science, 430, 24–36. https://doi.org/10.1016/j.memsci.2012.11.051.
Sundar, S., & Chakravarty, J. (2010). Antimony toxicity. International Journal of Environmental Research and Public Health, 7(12), 4267–4277. https://doi.org/10.3390/ijerph7124267.
Thanawatpoontawee, S., Imyim, A., & Praphairaksit, N. (2016). Iron-loaded zein beads as a biocompatible adsorbent for arsenic(V) removal. Journal of Industrial and Engineering Chemistry, 43, 127–132. https://doi.org/10.1016/j.jiec.2016.07.058.
US Environmental Protection Agency (EPA). (2001). National primary drinking water regulation: Arsenic and clarifications to compliance and new source contaminants monitoring: Final rule. Federal Register, 66, 6976–7066.
US Environmental Protection Agency (EPA). (2009). National Primary Drinking Water Regulations. Drinking Water Contaminants, EPA 816-F-09-004.
Yu, T., Wang, X., & Li, C. (2014). Removal of antimony by FeCl3-modified granular-activated carbon in aqueous solution. Journal of Environmental Engineering (United States), 140(9), 3–8. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000736.
Zhang, G., Qu, J., Liu, H., Liu, R., & Wu, R. (2007). Preparation and evaluation of a novel Fe–Mn binary oxide adsorbent for effective arsenite removal. Water Research, 41(9), 1921–1928.
Zhao, X., Dou, X., Mohan, D., Pittman, C. U., Ok, Y. S., & Jin, X. (2014). Antimonate and antimonite adsorption by a polyvinyl alcohol-stabilized granular adsorbent containing nanoscale zero-valent iron. Chemical Engineering Journal, 247, 250–257. https://doi.org/10.1016/j.cej.2014.02.096.
Acknowledgements
This research was financially supported by Brain Korea 21 Plus Project (BK21 Plus) of School of Environmental Science and Engineering at Gwangju Institute of Science and Technology (GIST), Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, SH., Choi, H. & Kim, KW. Removal of As(V) and Sb(V) in aqueous solution by Mg/Al-layered double hydroxide-incorporated polyethersulfone polymer beads (PES-LDH). Environ Geochem Health 40, 2119–2129 (2018). https://doi.org/10.1007/s10653-018-0087-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-018-0087-y