Abstract
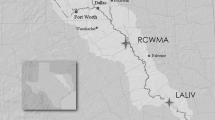
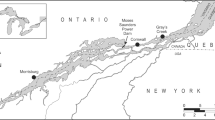
The concept of the Anthropocene, that humans are now re-engineering global ecosystems, is in part evidenced by the pervasive pollution by persistent organic pollutants (POPs). Certain POPs are hormone mimics and can disrupt endocrine and hence reproductive processes, shown mainly by laboratory studies with model species. There are, in contrast, fewer confirmations of such disruption from eco-epidemiological studies of wild mammals. Here we used the American mink (Neovison vison) as a sentinel species for such a study. Over the period 1998–2006, 161 mink carcasses were obtained from commercial trappers in the Canadian provinces of British Columbia and Ontario. Mink were aged, sexed, measured, and body condition assessed. Livers were analyzed either individually or pooled for organochlorine (OC) pesticides, polychlorinated biphenyls (PCBs), and subsets for polybrominated diphenyl ethers (PBDEs). We primarily addressed whether contaminants affected male reproductive development by measuring baculum size and assessing the influences of age and body condition. We also considered the influence of spatial variation on relative exposure and size of baculum. Statistical models separated by age class revealed that significant relationships between baculum length or mass and juvenile mink were mostly positive, whereas for adults and first year mink they were mostly negative. A significant negative relationship for adult mink was determined between DDE and both baculum length and mass. For juvenile mink we found significant positive relationships between ∑PCBs, DDE and ∑PBDEs with baculum length. Our results provide some indication of negative effects of halogenated contaminants on male reproductive development in wild mink, and the most likely candidate chemical is the confirmed anti-androgenic compound, DDE, rather than PCBs or other compounds.








Similar content being viewed by others
References
Aguilar A, Borrell A (1988) Age-and sex-related changes in organochlorine compound levels in fin whales (Balaenoptera physalus) from the eastern North Atlantic. Mar Environ Res 25:195–211
Anderson MJ, Clarke KR, Gorley RN (2008) PERMANOVA+ for Primer vs. 6: User Manual/tutorial. Primer-E, Plymouth, UK
Aulerich RJ, Bursian SJ, Napolitano AC, Oleas T (2000) Feeding growing mink (Mustela vison) PCB Aroclor® 1254 does not affect baculum (Os-penis) development. Bull Environ Contam Toxicol 64:443–447
Aulerich RJ, Ringer RK (1971) Effects of feeding coho salmon and other Great Lakes fish on mink reproduction. Can J Zool 49:611–616
Basu N, Head J, Scheuhammer AM, Bursian SJ, Rouvinen-Watt K, Chan HM (2009) The mink is still a reliable sentinel species in environmental health. Environ Res 109:940–941
Basu N, Scheuhammer AM, Bursian SJ, Elliott J, Rouvinen-Watt K, Chan HM (2007) Mink as a sentinel species in environmental health. Environ Res 103:130–144
Bettinetti R, Galassi S, Guilizzoni P, Quadroni S (2011) Sediment analysis to support the recent glacial origin of DDT pollution in Lake Iseo (Northern Italy). Chemosphere 85:163–169
Birks JD, Linn IJ (1982) Studies of home range of the feral mink, Mustela vison. Proc Zool Soc Lond 49:231–257
Bleavins MR, Aulerich RJ, Ringer RK (1982) Placental and mammary transfer of polychlorinated and polybrominated biphenyls in the mink and ferret. ASTM STP 757:121–131
Bouwman H, Polder A, Venter B, Skaare JU (2008) Organochlorine contaminants in cormorant, darter, egret, and ibis eggs from South Africa. Chemosphere 71(2):227–241
Bouwman H, Sereda B, Meinhardt HM (2006) Simultaneous presence of DDT and pyrethroid residues in human breast milk from a malaria endemic area in South Africa. Environ Pollut 144(3):902–917
Bowman J, Kidd AG, Martin PA, McDaniel TV, Nituch LA, Schulte-Hostedde AI (2012) Testing for bias in a sentinel species: contaminants in free-ranging domestic, wild, and hybrid mink. Environ Res 112:77–82
Bowman J, Schulte-Hostedde A (2009) The mink is not a reliable sentinel species. Environ Res 109:937–939
Brunström B, Lund BO, Bergman A, Asplund L, Athanassiadis I, Athanasiadou M, Jensen S, Örberg J (2001) Reproductive toxicity in mink (Mustela vison) chronically exposed to environmentally relevant polychlorinated biphenyl concentrations. Environ Toxicol Chem 20(10):2318–2327
Burnett LJ, Sorenson KJ, Brandt J, Sandhaus EA, Ciani D, Clark M, David C, Theule J, Kasielke S, Risebrough RW (2013) Eggshell thinning and depressed hatching success of California Condors reintroduced to central California. Condor 115(3):477–491
Bursian SJ, Kern J, Remington RE, Link JE, Fitzgerald SD (2013) Dietary exposure of mink (Mustela vison) to fish from the upper Hudson River, New York, USA: Effects on reproduction and offspring growth and mortality. Environ Toxicol Chem 32(4):780–793
Bursian SJ, Sharma C, Aulerich RJ, Yamini B, Mitchell RR, Orazio CE, Moore DR, Svirsky S, Tillitt DE (2006) Dietary exposure of mink (Mustela vison) to fish from the Housatonic River, Berkshire County, Massachusetts, USA: effects on reproduction, kit growth, and survival. Environ Toxicol Chem 25(6):1533–1540
Campbell LM, Schindler DW, Donald DB, Muir DCG (2000) Organochlorine transfer in the food web of subalpine Bow Lake, Banff National Park. Can J Fish Aquat Sci 57:1258–1269
Chen C, Hamm JT, Hass JR, Birnbaum LS (2001) Disposition of polychlorinated dibenzo-p-dioxins, dibenzofurans, and non-ortho polychlorinated biphenyls in pregnant Long Evans rats and the transfer to offspring. Toxicol Appl Pharmacol 173:65–88
Clarke KR, Gorley RN (2014) Primerv7: User manual/tutorial. Primer-E, Plymouth
Costa LG, Giordano G (2007) Developmental neurotoxicity of polybrominated diphenyl ether (PBDE) flame retardants. Neurotoxicology 28(6):1047–1067
Crofton KM, Kodavanti PR, Derr-Yellin EC, Casey AC, Kehn LS (2000) PCBs, thyroid hormones, and ototoxicity in rats: cross-fostering experiments demonstrate the impact of postnatal lactation exposure. Toxicol Sci 57(1):131–140
Diamanti-Kandarakis E, Bourguignon J-P, Giudice LC, Hauser R, Prins GS, Soto AM, Zoeller RT, Gore AC (2009) Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev 30(4):293–342
Elliott JE, Henny CJ, Harris ML, Wilson LK, Norstrom RJ (1999) Chlorinated hydrocarbons in livers of American mink (Mustela vison) and river otter (Lutra canadensis) from the Columbia and Fraser River basins, 1990–1992. Environ Monit Assess 57(3):229–252
Elliott JE, Levac J, Guigueno MF, Shaw DP, Wayland M, Morrissey CA, Muir DCG, Elliott KH (2012) Factors influencing legacy pollutant accumulation in Alpine Osprey: biology, topography, or melting glaciers? Environ Sci Technol 46(17):9681–9689
Elliott JE, Martin PA, Arnold TW, Sinclair PH (1994) Organochlorines and reproductive success of birds in orchard and non-orchard areas of central British Columbia, 1990−91. Arch Environ Contam Toxicol 26:435–443
Eskenazi B, Chevrier J, Rosas LG, Anderson HA, Bornman MS, Bouwman H, Chen A, Cohn BA, De Jager C, Henshel DS, Leipzig F (2009) The Pine River statement: human health consequences of DDT use. Environ Health Perspect 117(9):1359
Fuglei E, Bustnes JO, Hop H, Mørk T, Björnfoth H, van Bavel B (2007) Environmental contaminants in arctic foxes (Alopex lagopus) in Svalbard: relationships with feeding ecology and body condition. Environ Pollut 146:128–138
Ginsberg G, Hattis D, Sonawane B (2004) Incorporating pharmacokinetic differences between children and adults in assessing children’s risks to environmental toxicants. Toxicol Appl Pharmacol 198:164–183
Grove RA (2006) Environmental contaminants in male river otters collected from Oregon and Washington, 1994-99, with reproductive organ hypoplasia observed in otter males. PhD Dissertation, Oregon State University
Grove RA, Henny CJ (2008) Environmental contaminants in male river otters from Oregon and Washington, USA, 1994–1999. Environ Monit Assess 145:49–73
Guertin DA, Ben-David M, Harestad A, Drouillard KG, Elliott JE (2010) Non-invasive fecal samplinglinks individual river otters to chlorinated hydrocarbon contaminant exposure. Environ Toxicol Chem 29:275–284
Guillette LJ, Crain DA, Gunderson MP, Kools SAE, Milnes MR, Orlando EF, Rooney AA, Woodward AR (2000) Alligators and endocrine disrupting contaminants: a current perspective. Am Zool 40(3):438–452
Guillette Jr LJ, Gross TS, Masson GR, Matter JM, Percival HF, Woodward AR (1994) Developmental abnormalities of the gonad and abnormal sex hormone concentrations in juvenile alligators from contaminated and control lakes in Florida. Environ Health Perspect 102:680–688
Harding LE, Harris ML, Stephens CR, Elliott JE (1999) Reproductive and morphological condition of Wild Mink (Mustela vison) and River Otters (Lutra canadensis) in relation to chlorinated hydrocarbon contamination. Environ Health Perspect 107(2):141–147
Harris ML, Elliott JE, Butler RW, Wilson LK (2003a) Reproductive success and chlorinated hydrocarbon contamination of resident great blue herons (Ardea herodias) from coastal British Columbia, Canada, 1977 to 2000. Environ Pollut 121:207–227
Harris ML, Wilson LK, Elliott JE, Bishop CA, Tomlin AD, Henning KV (2000) Transfer of DDT and metabolites from fruit orchard soils to American robins (Turdus migratorius) twenty years after agricultural use of DDT in Canada. Arch Environ Contam Toxicol 39(2):205–220
Harris ML, Wilson LK, Norstrom RJ, Elliott JE (2003b) Egg concentrations of polychlorinated dibenzo-p-dioxins and dibenzofurans in double-crested (Phalacrocorax auritus) and Pelagic (P. pelagicus) cormorants from the Strait of Georgia, Canada, 1973− 1998. Environ Sci Technol 37:822–831
Haynes JM, Wellman ST, Beckett KJ, Pagano JJ, Fitzgerald SD, Bursian SJ (2009) Histological lesions in mink jaws are a highly sensitive biomarker of effect after exposure to TCDD-like chemicals: field and literature-based confirmations. Arch Environ Contam Toxicol 57(4):803–807
Hebert CE, Keenleyside KA (1995) To normalize or not to normalize? Fat is the question. Environ Toxicol Chem 14:801–807
Hebert CE, Norstrom RJ, Weseloh DC (2000) A quarter century of environmental surveillance: the Canadian Wildlife Service’s Great Lakes herring gull monitoring program. Environ Rev 7:147–166
Hebert CE, Weseloh DV, Kot L, Glooschenko V (1994) Organochlorine contaminants in a terrestrial foodweb on the Niagara Peninsula, Ontario, Canada 1987–89. Arch Environ Contam Toxicol 26(3):356–366
Henny CJ, Blus LJ, Gregory SV, Stafford CJ (1981) PCBs and organochlorine pesticides in wild mink and river otters from Oregon. In: Worldwide Furbearer Conference Proceedings. Patuxent Wildlife Research Center, Frostburg, Maryland, pp 1763–1780
Hickey JP, Batterman SA, Chernyak SM (2006) Trends of chlorinated organic contaminants in Great Lakes trout and walleye from 1970 to 1998. Arch Environ Contam Toxicol 50:97–110
Hinck JE, Norstrom RJ, Orazio CE, Schmitt CJ, Tillitt DE (2009) Persistence of organochlorine chemical residues in fish from the Tombigbee River (Alabama, USA): Continuing risk to wildlife from a former DDT manufacturing facility. Environ Pollut 157(2):582–591
Hochstein Jr MS, Render JA, Bursian SJ, Aulerich RJ (2001) Chronic toxicity of dietary 2,3,7,8-tetrachlorodibenzo-p-dioxin to mink. Vet Hum Tox 43:134–139
Hornshaw TC, Aulerich RI, Johnson HE (1983) Feeding Great Lakes fish to mink: Effects on mink and accumulation and elimination of PCBs by mink. J Toxicol Environ Health 11:933–946
Huang AC, Nelson C, Elliott JE, Guertin D, Ritland C, Douillard K, Cheng KM, Schwantje HM (2018) River Otters (Lontra canadensis) “trapped” in a coastal environment contaminated with persistent organic pollutants: demographic and physiological consequences. Environ Pollut 238:306–316. (in press)
Iwata H, Watanabe M, Okajima Y, Tanabe S, Amano M, Miyazaki N, Petrov EA (2004) Toxicokinetics of PCDD, PCDF, and coplanar PCB congeners in Baikal seals, Pusa sibirica: age-related accumulation, maternal transfer, and hepatic sequestration. Environ Sci Technol 38:3505–3513
Jürgens MD, Crosse J, Hamilton PB, Johnson AC, Jones KC (2016) The long shadow of our chemical past–high DDT concentrations in fish near a former agrochemicals factory in England. Chemosphere 162:333–344
Kean EF, Lyons G, Chadwick EA (2013) Persistent organic pollutants and indicators of otter health. A CHEM Trust Report. Cardiff University. UK. www.chemtrust.org.uk
Kelce WR, Stone CR, Laws SC, Gray LE, Kemppainen JA, Wilson EM (1995) Persistent DDT metabolite p,p-DDE is a potent androgen receptor antagonist. Nature 375:581–585
Kelly BC, Ikonomou MG, Blair JD, Morin AE, Gobas FA (2007) Food web–specific biomagnification of persistent organic pollutants. Science 317(5835):236–239
Lohmann R, Breivik K, Dachs J, Muir D (2007) Global fate of POPs: current and future research directions. Environ Pollut 150(1):150–165
Mably TA, Moore RW, Goy RW, Peterson RE (1992) In utero and lactational exposure of male rats to 2,3,7,8-tetrachlorodibenzo-p-dioxin: 2. Effects on sexual behavior and the regulation of luteinizing hormone secretion in adulthood. Toxicol Appl Pharmacol 114(1):108–117
Martin PA, McDaniel TV, Huges KD, Hunter B (2017) Organochlorine contaminants in wild mink from the lower Great Lakes Basin, Canada, 1998-2006. Environ Monit Assess 189:459–471. (in press)
Martin PA, McDaniel TV, Hunter B (2006) Temporal and spatial trends in chlorinated hydrocarbon concentrations of mink in Canadian Lakes Erie and St. Clair. Environ Mon Assess 113:245–263
Matson GM (1981) Workbook for Cementum analysis. Matson’s Laboratory, Milltown, Montana, USA
Miller AA, Elliott JE, Elliott KH, Guigueno MF, Wilson LK, Lee S, Idrissi A (2014) Spatial and Temporal trends in Brominated Flame Retardants in Seabirds from the Pacific Coast of Canada. Environ Pollut. 195:48-55. http://dx.doi.org/10.1016/j.envpol.2014.08.009
Moore R, Rudy T, Lin T, Ko K, Peterson R (2001) Abnormalities of sexual development in male rats with in utero and lactational exposure to the antiandrogenic plasticizer di(2-ethylhexyl) phthalate. Environ Health Perspect 109:229–237
Papp Z, Bortolotti GR, Smits JE (2005) Organochlorine contamination and physiological responses in nestling tree swallows in Point Pelee National Park, Canada. Arch Environ Contam Toxicol 49(4):563–568
Paul JR (1968) Baculum development in mink. Trans Ill State Acad Sci 61:308–309
Persson S, Bäcklin B-M, Kindahl H, Brunström B, Magnusson U (2011) Influence of age, nutritional status and season on the reproductive system in wild male mink (Neovison vison). Eur J Wildl Res 57:1057–1063
Persson S, Brunström B, Bäcklin BM, Kindahl H, Magnusson U (2012) Wild mink (Neovison vison) as sentinels in environmental monitoring. Acta Vet Scand 54:1–3
Persson S, Magnusson U (2015) Environmental pollutants and alterations in the reproductive system in wild male mink (Neovison vison) from Sweden. Chemosphere 120:237–245
Persson S, Rotander A, van Bavel B, Brunström B, Bäcklin B-M, Magnusson U (2013) Influence of age, season, body condition and geographical area on concentrations of chlorinated and brominated contaminants in Wild Mink (Neovison vison) in Sweden. Chemosphere 90:1664–1671
Peterson RE, Theobald HM, Kimmel GL (1993) Developmental and reproductive toxicity of dioxins and related compounds: cross-species comparisons. Crit Rev Toxicol 23:283–335
Proulx G, Weseloh DVC, Elliott JE, Teeple S, Anghern PAM, Mineau P (1987) Organochlorine and PCB residues in Lake Erie mink populations. Bull Environ Contam Toxicol 39:939–944
Roos A, Greyerz E, Olsson M, Sandegren F (2001) The otter (Lutra lutra) in Sweden - population trends in relation to ∑DDT and total PCB concentrations during 1968-99. Environ Pollut 111(3):457–469
SAS Instit (2000) SAS/STAT Users Guide: Statistics. Release 8. SAS Institute, Cary, North Carolina
Sherburne JA, Dimond JB (1969) DDT persistence in wild hares and mink. J Wildl Manage 33:944–948
Schulte-Hostedde AI, Zinner B, Millar JS, Hickling GJ (2005) Restitution of mass-size residuals: Validating body condition indices. Ecology 86(1):155–163
Sonne C, Leifsson PS, Dietz R, Born EW, Letcher RJ, Hyldstrup L, Riget FF, Kirkegaard M, Muir DCG (2006) Xenoendocrine pollutants may reduce size of sexual organs in east Greenland Polar Bears (Ursus maritimus). Environ Sci Technol 40(18):5668–5674
Sonne C, Gustavson K, Letcher RJ, Diezt R (2015) Physiologically-based pharmacokinetic modelling of distribution, bioaccumulation and excretion of POPs in Greenland sledge dogs (Canis familiaris). Environ Res 142:380–386
Steffen W, Persson Å, Deutsch L, Zalasiewicz J, Williams M, Richardson K, CCrumley, Crutzen P, Folke C, Gordon L, Molina M (2011) The Anthropocene: From global change to planetary stewardship Ambio 40(7):739–761
Syed JH, Malik RN, Muhammad A (2014) Organochlorine pesticides in surface soils and sediments from obsolete pesticides dumping site near Lahore city, Pakistan: contamination status and their distribution. J Chem Ecol 30(1):87–96
Vandenberg LN, Colborn T, Hayes TB, Heindel JJ, Jacobs Jr DR, Lee DH, Shioda T, Soto AM, vom Saal FS, Welshons WV, Zoeller RT (2012) Hormones and endocrine-disrupting chemicals: low-dose effects and nonmonotonic dose responses. Endocr Rev 33(3):378–455
You L, Casanova M, Archibeque-Engle S, Sar M, Fan LQ, Heck HD (1998) Impaired male sexual development in perinatal Sprague–Dawley and Long–Evans Hooded Rats exposed in utero and lactationally to, p′-DDE. Toxicol Sci 45:162–173
You L, Gazi E, Archibeque-Engle S, Casanova M, Conolly RB, Heck H (1999) Transplacental and lactational transfer of p, p′-DDE in Sprague–Dawley rats. Toxicol Appl Pharmacol 157:134–144
Wania F, Dugani CB (2003) Assessing the long‐range transport potential of polybrominated diphenyl ethers: a comparison of four multimedia models. Environ Toxicol Chem 22(6):1252–1261
Woodward AR, Percival HF, Rauschenberger RH, Gross TS, Rice KG, Conrow R (2011) Abnormal alligators and organochlorine pesticides in Lake Apopka, Florida. In: Elliott JE, Bishop CA, Morrissey CA (eds) Wildlife ecotoxicology: forensic approaches. Springer, New York, pp 153–187
Wong LIL, Labrecque M, Ibuki N, Cox ME, Elliott JE, Beischlag TV (2015) P,p′-Dichlorodiphenyltrichloroethane (p,p′-DDT) and p,p′-dichlorodiphenyldichloroethylene (p,p′-DDE) repress prostate specific antigen levels in human prostate cancer cell lines. Chem Biol Interact 230:40–49
Wren CD (1991) Cause‐effect linkages between chemicals and populations of mink (Mustela vison) and otter (Lutra canadensis) in the great lakes basin. J Toxicol Environ Health 33(4):549–585
Xu LC, Sun H, Chen JF, Bian Q, Song L, Wang XR (2006) Androgen receptor activities of p,p′-DDE, fenvalerate and phoxim detected by androgen receptor reporter gene assay. Toxicol Lett 160:151–157
Zhang S, Bursian S, Martin PA, Chan HM, Martin JW (2008) Dietary accumulation, disposition, and metabolism of technical pentabrominated diphenyl ether (DE-71) in pregnant mink (Mustela vison) and their offspring. Environ Toxicol Chem 27:1184–1193
Zhang S, Bursian SJ, Martin PA, Chan HM, Tomy G, Palace VP, Mayne GJ, Martin JW (2009) Reproductive and developmental toxicity of a pentabrominated diphenyl ether mixture, DE-71, to ranch mink (Mustela vison) and hazard assessment for wild mink in the Great Lakes region. Toxicol Sci 110:107–116
Zwiernik MJ, Kay DP, Moore J, Beckett KJ, Seong Khim J, Newsted JL, Roark SA, Giesy JP (2008) Exposure and effects assessment of resident mink (Mustela vison) exposed to polychlorinated dibenzofurans and other dioxin-like compounds in the Tittabawassee River Basin, Midland, Michigan, USA. Environ Toxicol Chem 27(10):2076–2087
Acknowledgements
We thank the many trappers throughout the provinces of British Columbia and Ontario for providing carcasses. Dr. M. McAdie, H. Gill, M. Fronteddu for tissue collection, cleaning and measurements; G. Savard coordinated the tissue preparation of liver samples for chemistry. H. Won, P. Dunlop, B. Wakeford, K. Drouillard and N. Ismali are thanked for their work on the chemical analyses. B. Hunter provided advice on veterinary pathology. M. Anderson provided R statistical advice for multivariate modelling in PERMANOVA+, and R. Brook who assisted with GLMM models.
Funding
Funding was provided mainly by the Georgia Basin and Great Lakes Action Plans of Environment and Climate Change Canada to J. Elliott and P. Martin, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The animals used in this study were obtained solely from trappers licensed under the governments of British Columbia and Ontario, and the principle investigators had the required carcass possession permits from those governments.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Elliott, J.E., Kirk, D.A., Martin, P.A. et al. Effects of halogenated contaminants on reproductive development in wild mink (Neovison vison) from locations in Canada. Ecotoxicology 27, 539–555 (2018). https://doi.org/10.1007/s10646-018-1926-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-018-1926-4