Abstract
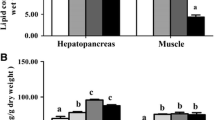
In environment, birds often fast in connection with breeding, migration or drastic climatic conditions and need to mobilize lipid reserves during these periods. The impairment of lipid metabolism by cadmium (Cd; 1 mg kg−1 added in diet) was investigated on palmiped Cairina moschata. Expression levels of genes involved in lipid metabolism, mitochondrial metabolism and detoxification were investigated in liver and muscle of ducks. Lipid content in muscle and liver were analysed and plasma triglycerides were quantified. After 20 days, ducks exposed to Cd displayed a lower body weight and lower lipid content in liver than controls. In muscle, the increase of lipid content was only significant for control ducks but not for exposed ducks. Exposed ducks appeared unable to sufficiently transport and store lipids into peripheral tissues. Cd impairs lipid metabolism by several ways. First, Cd triggered the down-regulation of fatty acids synthesis in liver even if the NADPH production and the mitochondrial metabolism are enhanced, suggesting a stronger energy needs. Secondly, the associated decrease of plasma triglycerides and lipoprotein lipase activity with Cd are consistent with impairment of lipids storage in peripheral tissues.


Similar content being viewed by others
References
André JM, Guy G, Gontier-Latonnelle K, Bernadet MD, Davail B, Hoo-Paris R, Davail S (2007) Influence of lipoprotein-lipase activity on plasma triacylglycerol concentration and lipid storage in three genotypes of ducks. Comp Biochem Physiol A Mol Integr Physiol 148:899–902. doi:10.1016/j.cbpa.2007.09.006
Auwerx J, Leroy P, Schoonjans K (1992) Lipoprotein lipase: recent contributions from molecular biology. Crit Rev Clin Lab Sci 29:243–268. doi:10.3109/10408369209114602
Baudrimont M, Schäfer J, Marie V, Maury-Brachet R, Bossy C, Boudou A, Blanc G (2005) Geochemical survey and metal bioaccumulation of three bivalve species (Crassostrea gigas, Cerastoderma edule and Ruditapes philippinarum) in the Nord Médoc salt marshes (Gironde estuary, France). Sci Total Environ 337:265–280. doi:10.1016/j.scitotenv.2004.07.009
Bertin G, Averbeck D (2006) Cadmium: cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie 88:1549–1559. doi:10.1016/j.biochi.2006.10.001
Berzina N, Markovs J, Isajevs S, Apsite M, Smirnova G (2007) Cadmium-induced enteropathy in domestic cocks: a biochemical and histological study after subchronic exposure. Basic Clin Pharmacol Toxicol 101:29–34. doi:10.1111/j.1742-7843.2007.00072.x
Cherel Y, Robin JP, Le Maho Y (1988) Physiology and biochemistry of long-term fasting in birds. Can J Zool 66:159–166. doi:10.1139/z88-022
Digel M, Ehehalt R, Stremmel W, Füllekrug J (2009) Acyl-CoA synthetases: fatty acid uptake and metabolic channeling. Mol Cell Biochem 326:23–28. doi:10.1007/s11010-008-0003-3
Erdogan Z, Erdogan S, Celik S, Unlu A (2005) Effects of ascorbic acid on cadmium-induced oxidative stress and performance of broilers. Biol Trace Elem Res 104:19–31. doi:10.1385/BTER:104:1:019
Folch J, Lees M, Stanley GHS (1957) A simple method fort he isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509
Gaskin F, Clayton RB (1972) An interstrain difference in cholesterol synthesis in vitro in mice, dependent upon a difference in endogenous NADPH-generating capacity. J Lipid Res 13:106–114
Goldberg IJ (1996) Lipoprotein lipase and lipolysis: central roles in lipoprotein metabolism and atherogenesis. J Lipid Res 37:693–707
Gonzalez P, Baudrimont M, Boudou A, Bourdineaud JP (2006) Comparative effects of direct cadmium contamination on gene expression in gills, liver, skeletal muscles and brain of the zebrafish (Danio rerio). Biometals 19:225–235. doi:10.1007/s10534-005-5670-x
Hermier D (1997) Lipoprotein metabolism and fattening in poultry. J Nutr 127:805–808
Herzberg GR, Brosnan JT, Hall B, Rogerson M (1988) Gluconeogenesis in liver and kidney of common murre (Uria aalge). Am J Physiol Regul Integr Comp Physiol 254:903–907
Iritani N, Ikeda Y, Fukuda H, Katsurada A (1984) Comparative study of lipogenic enzymes in several vertebrates. Lipids 19:828–835. doi:10.1007/BF02534511
Iynedjian P (1993) Mammalian glucokinase and its gene. Biochem J 293:1–13
Jordan SA, Bhatnagar MK, Bettger WJ (1990) Combined effects of methylmercury, lead, and cadmium on hepatic metallothionein and metal concentrations in the pekin duck. Arch Environ Contam Toxicol 19:886–891. doi:10.1007/BF01055055
Karmakar R, Banerjee A, Datta S, Chatterjee M (1999) Influence of cadmium intoxication on hepatic lipid peroxidation, glutathione level, and glutathione S-transferase and gamma-glutamyl transpeptidase activities: correlation with chromosome aberrations in bone marrow cells. J Environ Pathol Toxicol Oncol 18:277–287
Kim J, Shin JR, Koo TH (2009) Heavy metal distribution in some wild birds from Korea. Arch Environ Contam Toxicol 56:317–324. doi:10.1007/s00244-008-9180-z
Kumar P, Prasad Y, Patra A, Swarup D (2007) Levels of cadmium and lead in tissues of freshwater fish (Clarias batrachus L.) and chicken in western UP (India). Bull Environ Contam Toxicol 79:396–400. doi:10.1007/s00128-007-9263-y
Larregle EV, Varas SM, Oliveros LB, Martinez LD, Antón R, Marchevsky E, Giménez MS (2008) Lipid metabolism in liver of rat exposed to cadmium. Food Chem Toxicol 46:1786–1792. doi:10.1016/j.fct.2008.01.018
Le Maho Y, Vu Van Kha H, Koubi H, Dewasmes G, Girard J, Ferre P, Cagnard M (1981) Body composition, energy expenditure, and plasma metabolites in long-term fasting geese. Am J Physiol Endocrinol Metab 241:342–354
Lucia M, André JM, Bernadet MD, Gontier K, Guy G, Davail S (2008) Concentrations of metals (zinc, copper, cadmium, and mercury) in three domestic ducks in France: pekin, muscovy, and mule ducks. J Agric Food Chem 56:281–288. doi:10.1021/jf072523x
Mayack LA, Bush PB, Fletcher OJ, Page RK, Fendley TT (1981) Tissue residues of dietary cadmium in wood ducks. Arch Environ Contam Toxicol 10:637–645. doi:10.1007/BF01054886
Merkel M, Eckel RH, Goldberg IJ (2002) Lipoprotein lipase: genetics, lipid uptake, and regulation. J Lipid Res 43:1997–2006. doi:10.1194/jlr.R200015-JLR200
Pierron F, Baudrimont M, Bossy A, Bourdineaud JP, Brèthes D, Elie P, Massabuau JC (2007) Impairment of lipid storage by cadmium in the European eel (Anguilla anguilla). Aquat Toxicol 81:304–311. doi:10.1016/j.aquatox.2006.12.014
Pilo B, George JC (1983) Diurnal and seasonal variation in liver glycogen and fat in relation to metabolic status of liver and m. pectoralis in the migratory starling, Sturnus roseus, wintering in India. Comp Biochem Physiol A Comp Physiol 74:601–604
Revis NW, Major TC, Horton CY (1980) The effects of calcium, magnesium, lead, or cadmium on lipoprotein metabolism and atherosclerosis in the pigeon. J Environ Pathol Toxicol 4:293–303
Rho HK, Park J, Suh JH, Kim JB (2005) Transcriptional regulation of mouse 6-phosphogluconate dehydrogenase by ADD1/SREBP1c. Biochem Biophys Res Commun 332:288–296. doi:10.1016/j.bbrc.2005.04.120
Rodrigue J, Champoux L, Leclair D, Duchesne JF (2007) Cadmium concentrations in tissues of willow ptarmigan (Lagopus lagopus) and rock ptarmigan (Lagopus muta) in Nunavik, Northern Québec. Environ Pollut 147:642–647. doi:10.1016/j.envpol.2006.10.017
Rogalska J, Brzóska MM, Roszczenko A, Moniuszko-Jakoniuk J (2009) Enhanced zinc consumption prevents cadmium-induced alterations in lipid metabolism in male rats. Chem Biol Interact 177:142–152. doi:10.1016/j.cbi.2008.09.011
Rollin X, Médale F, Gutieres S, Blanc D, Kaushik SJ (2003) Short- and long-term nutritional modulation of acetyl-CoA carboxylase activity in selected tissues of rainbow trout (Oncorhynchus mykiss). Br J Nutr 89:803–810. doi:10.1079/BJN2003844
Rous S, Lüthi L (1968) Respective roles of NADH and NADPH in the synthesis of fatty acids. Helv Physiol Pharmacol Acta 26:243–246
Scheuhammer AM, Templeton DM (1990) Metallothionein production: similar responsiveness of avian liver and kidney to chronic cadmium administration. Toxicology 60:151–159. doi:10.1016/0300-483X(90)90169-H
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal ions. Free Radic Biol Med 18:321–336. doi:10.1016/0891-5849(94)00159-H
Wakil SJ, Stoops JK, Joshi VC (1983) Fatty acid synthesis and its regulation. Annu Rev Biochem 52:537–579. doi:10.1146/annurev.bi.52.070183.002541
Wang Y, Fang J, Leonard SS, Krishna Rao KM (2004) Cadmium inhibits the electron transfer chain and induces reactive oxygen species. Free Radic Biol Med 36:1434–1443. doi:10.1016/j.freeradbiomed.2004.03.010
Wayland M, Smits JE, Gilchrist HG, Marchant T, Keating J (2003) Biomarker responses in nesting, common eiders in the Canadian arctic in relation to tissue cadmium, mercury and selenium concentrations. Ecotoxicology 12:225–237. doi:10.1023/A:1022506927708
Weinstock PH, Levak-Frank S, Hudgins LC, Radner H, Friedman JM, Zechner R, Breslow JL (1997) Lipoprotein lipase controls fatty acid entry into adipose tissue, but fat mass is preserved by endogenous synthesis in mice deficient in adipose tissue lipoprotein lipase. Proc Natl Acad Sci USA 94:10261–10266
Zechner R (1997) The tissue-specific expression of lipoprotein lipase: implications for energy and lipoprotein metabolism. Curr Opin Lipidol 8:77–88
Acknowledgments
The authors would like to thank the Conseil Général des Landes (France) for their financial support and the INRA of Artiguères for technical help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lucia, M., André, JM., Gonzalez, P. et al. Effect of dietary cadmium on lipid metabolism and storage of aquatic bird Cairina moschata . Ecotoxicology 19, 163–170 (2010). https://doi.org/10.1007/s10646-009-0401-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-009-0401-7