Abstract
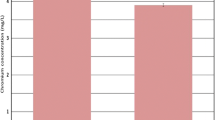
The removal of hexavalent and trivalent chromium from hydroponic solution by plants to changes in temperature was investigated. Pre-rooted hybrid willows (Salix matsudana Koidz × alba L.) were exposed to a nutrient solution spiked with potassium chromate (K2CrO4) or chromium chloride (CrCl3) for 4 days. Ten different temperatures were tested ranging from 11 to 32°C. Total Cr in solutions and in plant materials were all analyzed quantitatively. The results revealed that large amounts of the applied Cr were removed from the hydroponic solution in the presence of the plants. Significantly faster removal of Cr(III) than Cr(VI) was achieved by hybrid willows from the hydroponic solutions at all temperatures (P < 0.01). The removal rates of both chemical forms of Cr by plants increased linearly with the increase of temperatures. The highest removal rate of Cr(VI) was found at 32°C with a value of 1.99 μg Cr/g day, whereas the highest value of Cr(III) was 3.55 μg Cr/g day at the same temperature. Roots were the main sink for Cr accumulation in plants at all temperatures. Translocation of both chemical forms of Cr from roots to lower stems was only found at temperatures ≥24°C. The temperature coefficient values (Q 10) were 2.41 and 1.42 for Cr(VI) and Cr(III), respectively, indicating that the removal of Cr(VI) by hybrid willows was much more susceptible to changes in temperature than that of Cr(III). This information suggests that changes in temperature have a substantial influence on the uptake and accumulation of both chemical forms of Cr by plants.




Similar content being viewed by others
References
Atkin OK, Evans J, Ball M, Lambers H, Pons T (2000) Leaf respiration of snow gum in the light and dark: interactions between temperature and irradiance. Plant Physiol 122:915–923
Atkin OK, Zhang Q, Wiskich J (2002) Effect of temperature on rates of alternative and cytochrome pathway respiration and their relationship with the redox poise of the Quinone pool. Plant Physiol 128:212–222
Azcón-Bieto J (1992) Relationships between photosynthesis and respiration in the dark in plants. In: Barber J, Guerrero MG, Medrano H (eds) Trends in photosynthesis research. Intercept Ltd, Andover, pp 241–253
Baghour M, Moreno DA, Herna′ndez J, Castilla N, Romero L (2001) Influence of root temperature on phytoaccumulation of As, Ag, Cr and Sb in potato plants (Solanum tuberosum L. var. spunta). J Environ Sci Health A 36:1389–1401
Banks MK, Schwab AP, Henderson C (2006) Leaching and reduction of chromium in soil as affected by soil organic content and plants. Chemosphere 62:255–264
Becquer T, Quantin C, Sicot M, Boudot JP (2003) Chromium availability in ultramafic soils from New Caledonia. Sci Total Environ 301:251–261
Chatterjee J, Chatterjee C (2000) Phytotoxicity of cobalt, chromium and copper in cauliflower. Environ Pollut 109:69–74
Davis FT, Puryear JD, Newton RJ, Egilla JN, Grossi JAS (2002) Mycorrhizal fungi increase chromium uptake by sunflower plants: influence on tissue mineral concentration, growth, and gas exchange. J Plant Nutr 25:2389–2407
Dixit V, Pandey V, Shyam R (2002) Chromium ions inactivate electron transport and enhance superoxide generation in vivo in pea (Pisum sativum L.cv. Azad) root mitochondria. Plant Cell Environ 25:687–693
Fitter AH, Graves JD, Self GK, Brown TK, Bogie DS, Taylor K (1998) Root production, turnover and respiration under two grassland types along with an altitudinal gradient-influence of temperature and solar radiation. Oecologia 114:20–30
Fritioff A, Kautsky L, Greger M (2005) Influence of temperature and salinity on heavy metal uptake by submersed plants. Environ Pollut 133:265–274
Greger M (1999) Metal availability, uptake, transport and accumulation in plants. In: Prasad MNV, Hagemeyer J (eds) Heavy metal stress in plants: from molecules to ecosystems. Springer-Verlag, Heidelberg, pp 51–57
Han FX, Banin A, Su Y, Monts DL, Plodinec MJ, Kingery WL, Triplett GB (2002) Industrial age anthropogenic inputs of heavy metals into the pedosphere. Naturwissenschaften 89:497–504
Hunter JG, Vergnano O (1953) Trace element toxicities in oat plants. Ann Appl Biol 4:761–777
Katz SA, Salem H (1994) The biological and environmental chemistry of chromium. VCH Publishers, New York
Kimbrough DE, Cohen Y, Winer AM, Creelam L, Mabuni C (1999) A critical assessment of chromium in the environment. Crit Rev Environ Sci Technol 29:1–46
Kumar PBAN, Dushenkov V, Motto H, Raskin I (1995) Phytoextraction: the use of plants to remove heavy metals from soils. Environ Sci Technol 29:1232–1238
Larcher W (1995) Physiological plant ecology, 3rd edn. Springer, Berlin
McGrath SP (1982) The uptake and translocation of tri- and hexavalent chromium and effects on the growth of oat in flowing nutrient solution and in soil. New Phytol 92:381–390
Mei BJ, Puryear JD, Newton RJ (2002) Assessment of Cr tolerance and accumulation in selected plant species. Plant Soil 247:223–231
Pulford ID, Watson C, McGregor SD (2001) Uptake of chromium by trees: prospects for phytoremediation. Environ Geochem Health 23:307–311
Ramachandran V, D’Souza TJ, Mistry KB (1980) Uptake and transport of chromium in plants. J Nucl Agric Biol 9:126–129
Sachs L (1992) Angewandte Statistik. Springer, Berlin
Scoccianti V, Crinelli R, Tirillini B, Mancinelli V, Speranza A (2006) Uptake and toxicity of Cr (Cr3+) in celery seedlings. Chemosphere 64:1695–1703
Shahandeh H, Hossner LR (2000) Plant screening for chromium phytoremediation. Int J Phytorem 2:31–51
Shanker AK, Cervantes C, Loza-Tavera H, Avudainayagam S (2005) Chromium toxicity in plants. Environ Int 31:739–753
Sharma DC, Sharma CP, Tripathi RD (2003) Phytotoxic lesions of chromium in maize. Chemosphere 51:63–68
Skeffington RA, Shewry PR, Peterson PJ (1976) Chromium uptake and transport in barley seedlings (Hordeum vulgare L.). Planta 132:209–214
Tjoelker MG, Oleksyn J, Reich PB (1999) Acclimation of respiration to temperature and CO2 in seedlings of boreal tree species in relation to plant size and relative growth rate. Glob Chang Biol 5:679–691
Trapp S, Zambrano KC, Kusk KO, Karlson U (2000) A phytotoxicity test using transpiration of willows. Arch Environ Contam Toxicol 39:154–160
Yu XZ, Gu JD (2007) Accumulation and distribution of trivalent chromium and effects on hybrid willow (Salix matsudana Koidz × alba L.) metabolism. Arch Environ Contam Toxicol 52:503–511
Yu XZ, Gu JD (2008) The role of EDTA in phytoextraction of hexavalent and trivalent chromium by two willows tress. Ecotoxicol 17:143–152
Yu XZ, Trapp S, Zhou PH, Hu H (2005) The effect of temperature on the rates of cyanide metabolism of two woody plants. Chemosphere 59:1099–1104
Yu XZ, Gu JD, Huang SZ (2007a) Hexavalent chromium induced stress and metabolic responses of in hybrid willows. Ecotoxicol 16:299–309
Yu XZ, Trapp S, Zhou PH, Chen L (2007b) Effect of temperature on the uptake and metabolism of cyanide by weeping willows. Int J Phytorem 9:243–255
Yu XZ, Gu JD, Xin LQ (2008) Differences in uptake and translocation of hexavalent and trivalent chromium by two species of willows. Ecotoxicol 17:747–755
Zayed AM, Terry N (2003) Chromium in environment: factors affecting biological remediation. Plant Soil 249:135–156
Zayed A, Lytle CM, Terry N, Qian JH (1998) Chromium accumulation, translocation and chemical speciation in vegetable crops. Planta 206:293–299
Acknowledgments
This work was financially supported by The National Science Foundation of China (NSFC: 30770389).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, XZ., Peng, XY. & Xing, LQ. Effect of temperature on phytoextraction of hexavalent and trivalent chromium by hybrid willows. Ecotoxicology 19, 61–68 (2010). https://doi.org/10.1007/s10646-009-0386-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-009-0386-2