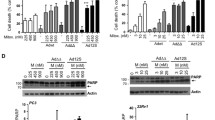
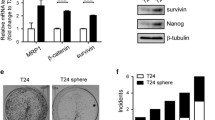
Summary
As a potential cancer therapy, we developed a recombinant adenovirus named Ad-VT, which was designed to express the apoptosis-inducing gene (apoptin) and selectively replicate in cancer cells via E1a manipulation. However, how it performs in bladder cancer remains unclear. We examined the antitumor efficacy of Ad-VT in bladder cancers using CCK-8 assays and xenograft models. Autophagy levels were evaluated by western blotting, MDC staining, and RFP-GFP-LC3 aggregates’ analyses. Here, we report the selective replication and antitumor efficacy (viability inhibition and apoptosis induction) of Ad-VT in bladder cancer cells. Using xenograft tumor models, we demonstrate that its effects are tumor specific resulting in the inhibition of tumor growth and improvement of the survival of mice models. Most Importantly, Ad-VT induced a complete autophagy flux leading to autophagic cancer cell death through a signaling pathway involving AMPK, raptor and mTOR. Finally, we suggest that treatment combination of Ad-VT and rapamycin results in a synergistic improvement of tumor control and survival compared to monotherapy. This study suggests that Ad-VT can induce selective autophagic antitumor activities in bladder cancer through the AMPK-Raptor-mTOR pathway, which can be further improved by rapamycin.








Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Chen W, He J, Sun K, Zheng R, Zeng H, Zhang S, Xia C, Yang Z, Li H, Zou X (2018) Cancer incidence and mortality in China, 2014. Chin J Cancer Res 030(001):1–12
Hu C, Liu Y, Lin Y, Liang JK, Zhong WW, Li K, Huang WT, Wang DJ, Yan GM, Zhu WB (2018) Intravenous injections of the oncolytic virus M1 as a novel therapy for muscle-invasive bladder cancer. Cell Death Dis 9(3):274
Loehrer PJ, Einhorn LH, Elson PJ, Crawford ED, Kuebler P, Tannock I, Raghavan D, Stuart-Harris R, Sarosdy MF, Lowe BA (1992) A randomized comparison of cisplatin alone or in combination with methotrexate, vinblastine, and doxorubicin in patients with metastatic urothelial carcinoma: a cooperative group study. J Clin Oncol 10(7):1066–1073
Choi JW, Lee JS, Kim SW, Yun CO (2011) Evolution of oncolytic adenovirus for cancer treatment. Adv Drug Deliv Rev 64(8):720–729
Apoptin (2008) Therapeutic Potential of an Early Sensor of Carcinogenic Transformation. Annu Rev Pharmacol Toxicol 48(1):143–169
Li X, Liu Y, Wen Z, Li C, Lu H, Tian M, Jin K, Sun L, Gao P, Yang E, Xu X, Kan S, Wang Z, Wang Y, Jin N (2010) Potent anti-tumor effects of a dual specific oncolytic adenovirus expressing apoptin in vitro and in vivo. Mol Cancer 9:10. https://doi.org/10.1186/1476-4598-9-10
Li X (2012) Therapeutic efficacy of an hTERT promoter-driven oncolytic adenovirus that expresses apoptin in gastric carcinoma. Int J Mol Med 30(4):747–54
Yang G, Meng X, Sun L, Hu N, Jiang S, Sheng Y, Chen Z, Zhou Y, Chen D, Li X, Jin N (2015) Antitumor effects of a dual cancer-specific oncolytic adenovirus on colorectal cancer in vitro and in vivo. Exp Ther Med 9(2):327–334
Yanxin Q, Huanhuan Guo N, Dongyun Hu, He Shi Z (2014) Preclinical pharmacology and toxicology study of Ad-hTERT-E1a-Apoptin, a novel dual cancer-specific oncolytic adenovirus. Toxicol Appl Pharmacol 280(2):362–9
Sun Y, Li C, Shu Y, Ju X, Zou Z, Wang H, Rao S, Guo F, Liu H, Nan W (2012) Inhibition of autophagy ameliorates acute lung injury caused by avian influenza A H5N1 infection. Sci Signal 5(212):ra16–ra16
Qin AP, Liu CF, Qin YY, Hong LZ, Xu M, Yang L, Liu J, Qin ZH, Zhang HL (2010) Autophagy was activated in injured astrocytes and mildly decreased cell survival following glucose and oxygen deprivation and focal cerebral ischemia. Autophagy 6(6):738–753
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140(3):0–326
Manuela Antonioli, Martina Di, Rienzo Mauro, Piacentini Gian, Maria Fimia (2017) Emerging mechanisms in initiating and terminating autophagy. Trends Biochem Sci 42(1):28–4
Kimura S, Noda T, Yoshimori T (2007) Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3(5):452–460
Levy JMM, Towers CG, Thorburn A (2017) Targeting autophagy in cancer. Nat Rev Cancer 17(9):528–542
B R, JE CVZB, LG O DSL, CJ FSDFERD (2004) Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet 36(6):585–595. https://doi.org/10.1038/ng1362
Jaeschke A (2002) Tuberous sclerosis complex tumor suppressor-mediated S6 kinase inhibition by phosphatidylinositide-3-OH kinase is mTOR independent. J Cell Biol 159(2):217–224
Egan DF, Shackelford DB, Mihaylova MM, Gelino S, Kohnz RA, Mair W, Vasquez DS, Joshi A, Gwinn DM, Taylor R (2011) Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science 331(6016):456–461
Abdulrahman BA, Khweek AA, Akhter A, Caution K, Amer AO (2011) Autophagy stimulation by rapamycin suppresses lung inflammation and infection by Burkholderia cenocepacia in a model of cystic fibrosis. Autophagy 7(11):1359–1370
Ramesh N (2006) CG0070, a conditionally replicating granulocyte macrophage colony-stimulating factor-armed oncolytic adenovirus for the treatment of bladder cancer. Clin Cancer Res 12(1):305–313
Jiang H, Rivera-Molina Y, Clise-Dwyer K, Bover L, Vence L, Yuan Y, Lang FF, Toniatti C, Hossain MB, Gomez-Manzano C (2017) Oncolytic adenovirus and tumor-targeting immune modulatory therapy improve autologous cancer vaccination. Cancer Res 77(14):3894–3907
Wu CL, Shieh GS, Chang CC, Yo YT, Su CH, Chang MY, Huang YH, Wu P, Shiau AL (2008) Tumor-selective replication of an oncolytic adenovirus carrying Oct-3/4 response elements in murine metastatic bladder cancer models. Clin Cancer Res 14(4):1228–1238
Rodriguez-Rocha H, Gomez-Gutierrez JG, Garcia-Garcia A, Rao XM, Lan C, Mcmasters KM, Zhou HS (2011) Adenoviruses induce autophagy to promote virus replication and oncolysis. Virology 416(1–2):9–15
Paglin S, Yahalom J (2006) Pathways that regulate autophagy and their role in mediating tumor response to treatment. Autophagy 2(4):291–293
Annovazzi L, Mellai M, Caldera V, Valente G, Schiffer D (2009) mTOR, S6 and AKT expression in relation to proliferation and apoptosis/autophagy in Glioma. Anticancer Res 29(8):3087–3094
Funding
This work was supported by the Key projects of science and technology boosting economy in 2020, (grant number SQ2020YFF0417940).
Author information
Authors and Affiliations
Contributions
Chao Shang conceived and performed the experiments and analysed the data. Chen-Chen Ge, Jing Lu, Gao-Jie Song, Yi-Long Zhu, Yi-Quan Li, Zhi-Ru Xiu, Wen-Jie Li, Shan-Zhi Li, Jia-Nan Cong, Zi-Rui Liu conducted the mouse experiments. Li Xiao, Li-Li Sun and Ning-Yi Jin overall supervised the experiments and revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The animal experimental protocols were approved by the Institutional Animal Care and Use Committee of the Academy of Military Medical Science (AMMS) and all efforts were made to minimize animal suffering and reduce the number of animals used for the experiments.
Consent for publication
Not applicable.
Research involving human participants and/or animals
The animal experimental protocols were approved by the Institutional Animal Care and Use Committee of the Academy of Military Medical Science (AMMS). The manuscript does not contain clinical studies or patient data.
Informed consent
For this type of study, informed consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shang, C., Zhu, YL., Li, YQ. et al. Autophagy promotes oncolysis of an adenovirus expressing apoptin in human bladder cancer models. Invest New Drugs 39, 949–960 (2021). https://doi.org/10.1007/s10637-021-01073-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-021-01073-x