Summary
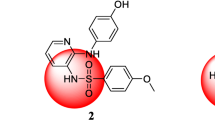
Novel quinoline-dithiocarbamate hybrids were synthesized and designed by the molecular hybridization strategy. All these derivatives were evaluated for their antiproliferative activity against three selected cancer cell lines (MGC-803, HepG-2 and PC-3). Among them, compound 10c displayed the best antiproliferative activity against PC-3 cells with an IC50 value of 0.43 μM. Celluar mechanisms investigated that compound 10c could inhibit the migration against PC-3 cells by regulation the expression levels of E-cadherin and N-cadherin. Compound 10c induced morphological changes of PC-3 cells and regulated apoptosis-related proteins (Bcl-2, Bax and Cleaved-Parp). In addition, compound 10c inhibited tubulin polymerization in vitro with an IC50 value of 4.02 μM. Importantly, compound 10c inhibited the growth of PC-3 cells in vivo with the low toxicity toward mice. These results suggested that compound 10c might be an antitumor agent with potential for treating prostate cancer.








Similar content being viewed by others
References
Nainwal LM, Tasneem S, Akhtar W, Verma G, Khan MF, Parvez S, Shaquiquzzaman M, Akhter M, Alam MM (2019) Green recipes to quinoline: a review. Eur J Med Chem 164:121–170
Afzal O, Kumar S, Haider MR, Ali MR, Kumar R, Jaggi M, Bawa S (2015) A review on anticancer potential of bioactive heterocycle quinoline. Eur J Med Chem 97:871–910
Li S, Hu L, Li J, Zhu J, Zeng F, Huang Q, Qiu L, Du R, Cao R (2019) Design, synthesis, structure-activity relationships and mechanism of action of new quinoline derivatives as potential antitumor agents. Eur J Med Chem 162:666–678
Lee HY, Chang CY, Su CJ, Huang HL, Mehndiratta S, Chao YH, Hsu CM, Kumar S, Sung TY, Huang YZ, Li YH, Yang CR, Liou JP (2016) 2-(Phenylsulfonyl)quinoline N-hydroxyacrylamides as potent anticancer agents inhibiting histone deacetylase. Eur J Med Chem 122:92–101
Li W, Shuai W, Sun H, Xu F, Bi Y, Xu J, Ma C, Yao H, Zhu Z, Xu S (2019) Design, synthesis and biological evaluation of quinoline-indole derivatives as anti-tubulin agents targeting the colchicine binding site. Eur J Med Chem 163:428–442
Buac D, Schmitt S, Ventro G, Kona FR, Dou QP (2012) Dithiocarbamate-based coordination compounds as potent proteasome inhibitors in human cancer cells. Mini Rev Med Chem 12:1193–1201
Laskar S, Sánchez-Sánchez L, Flores SM, López-Muñoz H, Escobar-Sánchez ML, López-Ortiz M, Hernández-Rodríguez M, Regla I (2018) Identification of (1S,4S)-2,5-diazabicyclo[2.2.1]heptane-dithiocarbamate-nitrostyrene hybrid as potent antiproliferative and apoptotic inducing agent against cervical cancer cell lines. Eur J Med Chem 146:621–635
Yang CR, Peng B, Cao SL, Ren TT, Jiang W, Wang FC, Li YS, Wang G, Li Z, Xu S, Liao J, Wang H, Li J, Xu X (2018) Synthesis, cytotoxic evaluation and target identification of thieno[2,3-d]pyrimidine derivatives with a dithiocarbamate side chain at C2 position. Eur J Med Chem 154:324–340
Li RD, Wang HL, Li YB, Wang ZQ, Wang X, Wang YT, Ge ZM, Li RT (2015) Discovery and optimization of novel dual dithiocarbamates as potent anticancer agents. Eur J Med Chem 93:381–391
Kerru N, Singh P, Koorbanally N, Raj R, Kumar V (2017) Recent advances (2015–2016) in anticancer hybrids. Eur J Med Chem 142:179–212
Guilford P, Hopkins J, Harraway J, McLeod M, McLeod N, Harawira P, Taite H, Scoular R, Miller A, Reeve AE (1998) E-cadherin germline mutations in familial gastric cancer. Nature 392:402
Karen MH, David YSC, Eric RF (2002) The SLUG zinc-finger protein represses E-cadherin in breast cancer. Cancer Res 62:1613–1618
Hassan M, Watari H, AbuAlmaaty A, Ohba Y, Sakuragi N (2014) Apoptosis and molecular targeting therapy in cancer. Biomed Res Int 2014:150845–150845
Matsuura K, Canfield K, Feng W, Kurokawa M (2016) Metabolic regulation of apoptosis in Cancer. Int Rev Cell Mol Bio 327:43–87
La Regina G, Bai R, Rensen WM, Di CE, Coluccia A, Piscitelli F, Famiglini V, Reggio A, Nalli M, Pelliccia S, Da PE, Costa B, Granata I, Porta A, Maresca B, Soriani A, Iannitto ML, Santoni A, Li J, Miranda CM, Chen F, Ni Y, Brancale A, Dondio G, Vultaggio S, Varasi M, Mercurio C, Martini C, Hamel E, Lavia P, Novellino E, Silvestri R (2013) Toward highly potent cancer agents by modulating the C-2 group of the arylthioindole class of tubulin polymerization inhibitors. J Med Chem 56:123–149
Zhao W, Bai JK, Li HM, Chen T, Tang YJ (2015) Tubulin structure-based drug design for the development of novel 4β-sulfur-substituted podophyllum tubulin inhibitors with anti-tumor activity. Sci Rep 5:10172–10172
Reddy MVR, Akula B, Cosenza SC, Lee CM, Mallireddigari MR, Pallela VR, Subbaiah DRCV, Udofa A, Reddy EP (2012) (Z)-1-aryl-3-arylamino-2-propen-1-ones, highly active stimulators of tubulin polymerization: synthesis, structure-activity relationship (SAR), tubulin polymerization, and cell growth inhibition studies. J Med Chem 55:5174–5187
Thomas E, Gopalakrishnan V, Hegde M, Kumar S, Karki SS, Raghavan SC, Choudhary BA (2016) Novel resveratrol based tubulin inhibitor induces mitotic arrest and activates apoptosis in Cancer cells. Sci Rep 6:34653–34653
Yang F, Song L, Wang H, Wang J, Xu Z, Xing N (2015) Combination of quercetin and 2-Methoxyestradiol enhances inhibition of human prostate Cancer LNCaP and PC-3 cells xenograft tumor growth. PLoS One 10:e0128277–e0128277
Friedemann M, Nacke B, Hagelgans A, Jandeck C, Bechmann N, Ullrich M, Belter B, Neuber C, Sukocheva O, Pietzsch J, Menschikowski M (2018) Diverse effects of phospholipase A2 receptor expression on LNCaP and PC-3 prostate cancer cell growth in vitro and in vivo. Oncotarget 9:35983–35996
Lee ST, Wong PF, He H, Hooper JD, Mustafa MR (2013) Alpha-tomatine attenuation of in vivo growth of subcutaneous and orthotopic xenograft tumors of human prostate carcinoma PC-3 cells is accompanied by inactivation of nuclear factor-kappa B signaling. PLoS One 8:e57708–e57708
George TG, Endeshaw MM, Morgan RE, Mahasenan KV, Delfín DA, Mukherjee MS, Yakovich AJ, Fotie J, Li C, Werbovetz KA (2007) Synthesis, biological evaluation, and molecular modeling of 3,5-substituted-N1-phenyl-N4,N4-di-n-butylsulfanilamides as antikinetoplastid antimicrotubule agents. Bioorg Med Chem 15:6071–6079
Acknowledgments
So thanks the supports from The 4th affiliated hospital of CMU.
Funding
This work was supported by The 4th affiliated hospital of CMU.
Author information
Authors and Affiliations
Contributions
Jia Liu., Dongwei Xue., Xingwang Zhu., Liu Yu., Minghuan Mao and Yili Liu performed the design work and experiments. Jia Liu and Yili Liu witten the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. All applicable guidelines for the care and use of animals were followed by the 4th affiliated hospital of CMU (Shenyang, China).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, J., Xue, D., Zhu, X. et al. Anticancer evaluation of a novel dithiocarbamate hybrid as the tubulin polymerization inhibitor. Invest New Drugs 38, 525–532 (2020). https://doi.org/10.1007/s10637-019-00799-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-019-00799-z