Summary
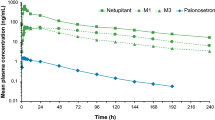
Rolapitant is a neurokinin-1 receptor antagonist that is approved in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting (CINV) associated with initial and repeat courses of emetogenic cancer chemotherapy, including but not limited to highly emetogenic chemotherapy. Here, we assessed the absorption, metabolism, and excretion of 14C-labeled rolapitant in healthy male subjects. Rolapitant was administered as a single 180-mg oral dose containing approximately 100 μCi of total radioactivity, with plasma, urine, and fecal samples collected at defined intervals after dosing. Rolapitant had a large apparent volume of distribution, indicating that it is widely distributed into body tissues. Rolapitant was slowly metabolized and eliminated with a mean half-life of 186 h. Exposure to the major metabolite of rolapitant, C4-pyrrolidinyl hydroxylated rolapitant or M19, was approximately 50% of rolapitant exposure in plasma. Renal clearance was not a significant elimination route for rolapitant-related entities. Total radioactivity recovered in urine accounted for 14.2% of the dose, compared to 72.7% recovery in feces. Adverse events (AEs) were generally mild; there were no serious AEs, and no clinically significant changes in laboratory or electrocardiogram parameters were observed. The combination of rolapitant safety, its long half-life, extensive tissue distribution, and slow elimination via the hepatobiliary route (rather than renal excretion) suggest suitability that a single dose of rolapitant may provide protection against CINV beyond the first 24 h after chemotherapy administration.



Similar content being viewed by others
References
Grunberg SM, Deuson RR, Mavros P, Geling O, Hansen M, Cruciani G, Daniele B, De Pouvourville G, Rubenstein EB, Daugaard G (2004) Incidence of chemotherapy-induced nausea and emesis after modern antiemetics. Cancer 100(10):2261–2268. https://doi.org/10.1002/cncr.20230
Berger MJ, Ettinger DS, Aston J, Barbour S, Bergsbaken J, Bierman PJ, Brandt D, Dolan DE, Ellis G, Kim EJ, Kirkegaard S, Kloth DD, Lagman R, Lim D, Loprinzi C, Ma CX, Maurer V, Michaud LB, Nabell LM, Noonan K, Roeland E, Rugo HS, Schwartzberg LS, Scullion B, Timoney J, Todaro B, Urba SG, Shead DA, Hughes M (2017) NCCN guidelines insights: antiemesis, version 2.2017. J Natl Compr Cancer Netw 15(7):883–893. https://doi.org/10.6004/jnccn.2017.0117
Cohen L, de Moor CA, Eisenberg P, Ming EE, Hu H (2007) Chemotherapy-induced nausea and vomiting—incidence and impact on patient quality of life at community oncology settings. Support Care Cancer 15(5):497–503. https://doi.org/10.1007/s00520-006-0173-z
Bloechl-Daum B, Deuson RR, Mavros P, Hansen M, Herrstedt J (2006) Delayed nausea and vomiting continue to reduce patients’ quality of life after highly and moderately emetogenic chemotherapy despite antiemetic treatment. J Clin Oncol 24(27):4472–4478. https://doi.org/10.1200/JCO.2006.05.6382
Hilarius DL, Kloeg PH, van der Wall E, van den Heuvel JJ, Gundy CM, Aaronson NK (2012) Chemotherapy-induced nausea and vomiting in daily clinical practice: a community hospital-based study. Support Care Cancer 20(1):107–117. https://doi.org/10.1007/s00520-010-1073-9
Hesketh PJ (2008) Chemotherapy-induced nausea and vomiting. N Engl J Med 358(23):2482–2494. https://doi.org/10.1056/NEJMra0706547
Duffy RA, Morgan C, Naylor R, Higgins GA, Varty GB, Lachowicz JE, Parker EM (2012) Rolapitant (SCH 619734): a potent, selective and orally active neurokinin NK1 receptor antagonist with centrally-mediated antiemetic effects in ferrets. Pharmacol Biochem Behav 102(1):95–100. https://doi.org/10.1016/j.pbb.2012.03.021
Garcia-Recio S, Gascon P (2015) Biological and pharmacological aspects of the NK1-receptor. Biomed Res Int 2015:495704. https://doi.org/10.1155/2015/495704
Rapoport B, Schwartzberg L, Chasen M, Powers D, Arora S, Navari R, Schnadig I (2016) Efficacy and safety of rolapitant for prevention of chemotherapy-induced nausea and vomiting over multiple cycles of moderately or highly emetogenic chemotherapy. Eur J Cancer 57:23–30. https://doi.org/10.1016/j.ejca.2015.12.023
Rapoport BL, Chasen MR, Gridelli C, Urban L, Modiano MR, Schnadig ID, Poma A, Arora S, Kansra V, Schwartzberg LS, Navari RM (2015) Safety and efficacy of rolapitant for prevention of chemotherapy-induced nausea and vomiting after administration of cisplatin-based highly emetogenic chemotherapy in patients with cancer: two randomised, active-controlled, double-blind, phase 3 trials. Lancet Oncol 16(9):1079–1089. https://doi.org/10.1016/S1470-2045(15)00035-2
Schwartzberg LS, Modiano MR, Rapoport BL, Chasen MR, Gridelli C, Urban L, Poma A, Arora S, Navari RM, Schnadig ID (2015) Safety and efficacy of rolapitant for prevention of chemotherapy-induced nausea and vomiting after administration of moderately emetogenic chemotherapy or anthracycline and cyclophosphamide regimens in patients with cancer: a randomised, active-controlled, double-blind, phase 3 trial. Lancet Oncol 16(9):1071–1078. https://doi.org/10.1016/S1470-2045(15)00034-0
Rapoport B, Chua D, Poma A, Arora S, Wang Y, Fein LE (2015) Study of rolapitant, a novel, long-acting, NK-1 receptor antagonist, for the prevention of chemotherapy-induced nausea and vomiting (CINV) due to highly emetogenic chemotherapy (HEC). Support Care Cancer 23(11):3281–3288. https://doi.org/10.1007/s00520-015-2738-1
Levade M, David E, Garcia C, Laurent PA, Cadot S, Michallet AS, Bordet JC, Tam C, Sie P, Ysebaert L, Payrastre B (2014) Ibrutinib treatment affects collagen and von Willebrand factor-dependent platelet functions. Blood 124(26):3991–3995. https://doi.org/10.1182/blood-2014-06-583294
Roila F, Molassiotis A, Herrstedt J, Aapro M, Gralla RJ, Bruera E, Clark-Snow RA, Dupuis LL, Einhorn LH, Feyer P, Hesketh PJ, Jordan K, Olver I, Rapoport BL, Roscoe J, Ruhlmann CH, Walsh D, Warr D, van der Wetering M, participants of the MECCC (2016) 2016 MASCC and ESMO guideline update for the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting and of nausea and vomiting in advanced cancer patients. Ann Oncol 27(suppl 5):v119–v133. https://doi.org/10.1093/annonc/mdw270
Hesketh PJ, Bohlke K, Lyman GH, Basch E, Chesney M, Clark-Snow RA, Danso MA, Jordan K, Somerfield MR, Kris MG, American Society of Clinical O (2016) Antiemetics: American Society of Clinical Oncology focused guideline update. J Clin Oncol 34(4):381–386. https://doi.org/10.1200/JCO.2015.64.3635
Rapoport BL (2017) Differential pharmacology and clinical utility of rolapitant in chemotherapy-induced nausea and vomiting. Cancer Manag Res 9:41–50. https://doi.org/10.2147/CMAR.S97543
Food and Drug Administration (Center for Drug Evaluation and Research) (2017) Guidance for industry: safety testing of drug metabolites. https://www.fda.gov/downloads/Drugs/.../Guidances/ucm079266.pdf
Committee for Medicinal Products for Human Use (2016) Guideline on evaluation of anticancer medicinal products in man. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2016/03/WC500203320.pdf
Nijenhuis CM, Schellens JH, Beijnen JH (2016) Regulatory aspects of human radiolabeled mass balance studies in oncology: concise review. Drug Metab Rev 48(2):266–280. https://doi.org/10.1080/03602532.2016.1181081
Wang X, Zhang ZY, Powers D, Wang J, Lu S, Kansra V (2017) Rolapitant absolute bioavailability and PET imaging studies in healthy adult volunteers. Clin Pharmacol Ther 102(2):332–339. https://doi.org/10.1002/cpt.637
Poma A, Christensen J, Pertikis H et al (2013) Rolapitant and its major metabolite do not affect the pharmacokinetics of midazolam, a sensitive cytochrome P450 3A4 substrate [abstract 441]. Support Care Cancer 21:S154
Hartlev LB, Boeje CR, Bluhme H, Palshof T, Rehling M (2012) Monitoring renal function during chemotherapy. Eur J Nucl Med Mol Imaging 39(9):1478–1482. https://doi.org/10.1007/s00259-012-2158-0
Tanabe N, Goto M, Morita H, Gotu T, Inagaki J, Yamanaki N, Kimura K (1991) Pharmacokinetics of cis-diammine-dichlor-platin in a hemodialysis patient. Cancer Investig 9(6):629–635. https://doi.org/10.3109/07357909109039874
Widemann BC, Adamson PC (2006) Understanding and managing methotrexate nephrotoxicity. Oncologist 11(6):694–703. https://doi.org/10.1634/theoncologist.11-6-694
Wang X, Zhang ZY, Powers D, Wang J, Lu S, Arora S, Hughes L, Christensen J, Kansra V (2017) Bioequivalence of intravenous and oral rolapitant: results from a randomized, open-label pivotal study. J Clin Pharmacol 57(12):1600–1606. https://doi.org/10.1002/jcph.966
Wang X, Zhang ZY, Arora S, Hughes L, Wang J, Powers D, Christensen J, Lu S, Kansra V (2018) Effects of rolapitant administered intravenously or orally on the pharmacokinetics of digoxin (P-glycoprotein substrate) and sulfasalazine (breast cancer resistance protein substrate) in healthy volunteers. J Clin Pharmacol 58(2):202–211. https://doi.org/10.1002/jcph.1005
Acknowledgments
The authors wish to acknowledge the contribution of the scientists at Schering-Plough Corporation in the execution of the study; to thank Jan Jaap van Lier, MD, who was the primary investigator for this study; and to thank Pearl Pathways, LLC for initial manuscript drafting. Writing and editorial support, funded by TESARO, Inc. (Waltham, MA, USA) and coordinated by Hemant Vyas, PhD of TESARO Inc., was provided by Tiffany Brake, PhD and Beverly Stanley, ELS of Team 9 Science, Vaniam Group (Chicago, IL, USA). All authors were involved in the collection, analysis, and interpretation of data, the writing of the manuscript, and the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Funding for this study and for the preparation of this manuscript was provided by TESARO, Inc. All authors are employees or former employees of the study sponsor. The authors have indicated that they have no other conflicts of interest regarding the content of this article. Professional medical writing support was used to develop this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
ESM 1
(DOCX 450 kb)
Rights and permissions
About this article
Cite this article
Zhang, ZY., Wang, J., Kansra, V. et al. Absorption, metabolism, and excretion of the antiemetic rolapitant, a selective neurokinin-1 receptor antagonist, in healthy male subjects. Invest New Drugs 37, 139–146 (2019). https://doi.org/10.1007/s10637-018-0638-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-018-0638-1