Summary
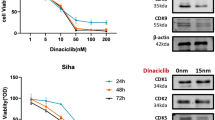
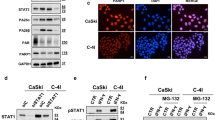
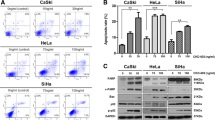
Objectives Our goal was to investigate the effects of rucaparib on the proliferation of cervical cancer cells and sensitivity to radiotherapy. Methods We used the human cervical cancer cell lines Hela and Siha and evaluated their viability and activity using various methods. Cellular proliferation was assessed by CCK-8 and clonogenic assays after treatment with rucaparib. Cell cycle analysis was performed using propidium iodide staining. Western immunoblotting analysis was used to detect the expression of cyclin D1 and CDK4. Immunofluorescence staining assay was performed to detect the expression of the DNA injury marker ץ-H2AX after treatment with rucaparib and radiotherapy. Animal experiments were also performed to evaluate tumor size after treatment with rucaparib. Immunohistochemistry was performed to analyze the expression of Ki-67. Results Rucaparib suppressed proliferation, induced G2/M phase arrest, and reduced the expression of cyclin D1 and CDK4 in cervical cancer cells. When rucaparib was combined with radiotherapy in cervical cancer cells, clone formation decreased significantly and G2/M phase arrest was accentuated. The expression of the DNA-damage marker ץ-H2AX was increased significantly, and rucaparib suppressed tumor growth in vivo. Conclusions Rucaparib exerts significant anti-proliferative effects and can serve as an effective radiosensitizer in cervical cancer, suggesting its candidacy in cervical cancer treatment and worthiness for further investigation.





Similar content being viewed by others
References
Jemal A, Bray F, Center MM et al (2011) Global cancer statistics [J]. CA Cancer J Clin 61(2):69–90
Chen W, Zheng R, Baade PD et al (2016) Cancer statistics in China, 2015 [J]. CA Cancer J Clin 66(2):115–132
Shi JF, Canfell K, Lew JB et al (2011) The burden of cervical cancer in China: synthesis of the evidence [J]. Int J Cancer. 130:641–652
Barker HE, Paget JT, Khan AA et al (2015) The tumor microenvironment after radiotherapy: mechanisms of resistance and recurrence [J]. Nat Rev Cancer 15(7):409–425
Schaue D, McBride WH (2015) Opportunities and challenges of radiotherapy for treating cancer [J]. Nat Rev Clin Oncol 12(9):527–540
Anwar M, Aslam HM, Anwar S (2015) PARP inhibitors [J]. Hered Cancer ClinPract 13(1):4
Rouleau M, Patel A, Hendzel MJ et al (2010) PARP inhibition: PARP1 and beyond [J]. Nat Rev Cancer 10(4):293–301
Underhill C, Toulmonde M, Bonnefoi H (2011) A review of PARP inhibitors:from bench to bedside [J]. Ann Oncol 22(2):268–279
Ceccaldi R, Rondinelli B (2016) D’ Andrea AD. Repair pathway choices and consequences at the double-strand break [J]. Trends Cell Biol 26(1):52–64
Guo GS, Zhang FM, Gao RJ et al (2011) DNA repair and synthetic lethality [J]. Int J Oral Sci 3(4):176–179
Powell ME (2010) Modern radiotherapy and cervical cancer. Int J Gynecol Cancer. 20:S49–S51
Hong JH, Tsal CS, Lai CH (2004) et a1. Recurrent squamous cell carcinoma of cervix after definitive radiotherapy [J]. Int J Radiat Oncol Biol Phys 60(1):249–257
Chambon P, Weill JD, Mandel P (1963) Nicotinamide mononucleotide activation of new DNA-dependent polyadenylicacid synthesizing nuclear enzyme [J]. Biochem Biophys Res Commun 11:39–43
Nirmal S, Amteshwar SJ (2010) Poly(ADP-ribose) polymerase-1(PARP-1) and its therapeutic implications [J]. Vascul Pharmacol 53(3-4):77–78
Plummer ER (2006) Inhibition of poly(ADP-ribose) polymerase in cancer [J]. Curr Opin Pharmacol 6(4):364–368
Powell C, Mikropoulos C, Kaye SB et al (2010) Pre-clinical and clinical evaluation of PARP inhibitors as tumor-specific radiosensitisers [J]. Cancer Treat Rev 36(7):566–575
Peralta LA, Rodriguez MI, Linares JL et al (2009) PARP inhibitors: new partners in the therapy of cancer and inflammatory diseases [J]. Free Radic Biol Med 47(1):13–26
Chalmers AJ (2009) The potential role and application of PARP inhibitors in cancer treatment [J]. Br Med Bull 89:23–40
Curtin NJ (2005) PARP inhibitors for cancer therapy [J]. Expert Rev Mol Med 7(4):1–20
Cepeda V, Fuertes MA, Castilla J et al (2006) Poly(ADP-ribose) polymerase-1 (PARP-1) inhibitors in cancer chemotherapy [J]. Recent Pat Anticancer Drug Discov 1(1):39–53
Fauzee NJ, Pan J, Wang YL (2010) PARP and PARG Inhibitors-New Therapeutic Targets in Cancer Treatment [J]. Pathol Oncol Res 16(4):469–478
Bryant HE, Schultz N, Thomas HD et al (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase [J]. Nature 434(7035):913–917
Gien LT, Mackay HJ (2009) The Emerging Role of PARP Inhibitors in the Treatment of Epithelial Ovarian Cancer [J]. Journal of Oncology 2010(1687-8450):151750
Odell ID, Wallace SS, Pederson DS (2013) Rules of engagement for base excision repair in chromatin [J]. J Cell Physiol 228(2):258–266
Farmer H, Mc Cabe N, Lord CJ et al (2005) Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy [J]. Nature 434(7035):917–921
Tentori L, Lacal PM, Muzi A et al (2007) Poly(ADP-ribose) polymerase(PARP) inhibition or PARP-1 gene deletion reduces angiogenesis [J]. Eur J Cancer 43(14):2124–2133
Tan DS, Rothermundt C, Thomas K et al (2008) “BRCAness” syndrome in ovarian cancer: a case-control study describing the clinical features and outcome of patients with epithelial ovarian cancer associated with BRCA1and BRCA2 mutations [J]. J Clin Oncol 26(34):5530–5536
Fong PC, Boss DS, Yap TA et al (2009) Inhibition of poly(ADP-ribose)polymerase in tumors from BRCA mutation carriers [J]. N Engl J Med 361(2):123–134
Drew Y, Plummer R (2009) PARP inhibitors in cancer therapy: two modes of attack on the cancer cell widening the clinical applications [J]. Drug Resist Updat 12(6):153–156
FDA. LYNPARZA[EB/OL]. [2014-12-19](2015-02-02) http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/206162lbl.pdf.
Acknowledgments
We would like to thank LetPub (www.letpub.com) for providing linguistic assistance during the preparation of this manuscript.
Funding
The work was supported by the Department of Health and Planning Commission in Wuxi, the China. The work was also supported by the Department of Affilited Hospital of Jiangnan University in Wuxi, the China.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
Author Mei Tang declares that he has no conflict of interest.
Author Qiuli Liu declares that he has no conflict of interest.
Author Leyuan Zhou declares that he has no conflict of interest.
Author Ling Chen declares that he has no conflict of interest.
Author Xueqing Yang declares that he has no conflict of interest.
Author Jinjin Yu declares that he has no conflict of interest.
Author Yuan Wang declares that he has no conflict of interest.
Author Haifeng Qiu declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Tang, M., Liu, Q., Zhou, L. et al. The poly (ADP-ribose) polymerase inhibitor rucaparib suppresses proliferation and serves as an effective radiosensitizer in cervical cancer. Invest New Drugs 37, 65–75 (2019). https://doi.org/10.1007/s10637-018-0616-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-018-0616-7