Summary
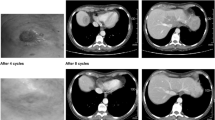
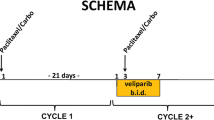
Purpose DFP-10917 is a novel deoxycytidine analog with a unique mechanism of action. Brief exposure to high concentrations of DFP-10917 inhibits DNA polymerase resulting in S-phase arrest, while prolonged exposure to DFP-10917 at low concentration causes DNA fragmentation, G2/M-phase arrest, and apoptosis. DFP-10917 demonstrated activity in tumor xenografts resistant to other deoxycytidine analogs. Experimental design Two phase I studies assessed the safety, pharmacokinetic, pharmacodynamic and preliminary efficacy of DFP-10917. Patients with refractory solid tumors received DFP-10917 continuous infusion 14-day on/7-day off and 7-day on/7-day off. Enrollment required age > 18 years, ECOG Performance Status 0–2 and adequate organ function. Results 29 patients were dosed in both studies. In 14-day infusion, dose-limiting toxicities (DLT) consisting of febrile neutropenia and thrombocytopenia occurred at 4.0 mg/m2/day. At 3.0 mg/m2/day, 3 patients experienced neutropenia in cycle 2. The dose of 2.0 mg/m2/day was well tolerated in 6 patients. In 7-day infusion, grade 4 neutropenia was DLT at 4.0 mg/m2/day. The maximum tolerated dose was 3 mg/m2/day. Other toxicities included nausea, vomiting, diarrhea, neutropenia, and alopecia. Eight patients had stable disease for >12 weeks. Paired comet assays performed for 7 patients showed an increase in DNA strand breaks at day 8. Pharmacokinetic data showed dose-proportionality for steady-state concentration and AUC of DFP-10917 and its primary metabolite. Conclusion Continuous infusion of DFP-10917 is feasible and well tolerated with myelosuppression as main DLT. The recommended doses are 2.0 mg/m2/day and 3.0 mg/m2/day on the 14-day and 7-day continuous infusion schedules, respectively. Preliminary activity was suggested. Pharmacodynamic data demonstrate biological activity at the tested doses.



Similar content being viewed by others
References
Galmarini CM, Mackey JR, Dumontet C (2002) Nucleoside analogues and nucleobases in cancer treatment. Lancet Oncol 3:415–424
Burris HA 3rd, Moore MJ, Andersen J et al (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15:2403–2413
Sandler AB, Nemunaitis J, Denham C et al (2000) Phase III trial of gemcitabine plus cisplatin versus cisplatin alone in patients with locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol 18:122–130
Hertel LW, Boder GB, Kroin JS et al (1990) Evaluation of the antitumor activity of gemcitabine (2′,2′-difluoro-2′-deoxycytidine). Cancer Res 50:4417–4422
Huang P, Chubb S, Hertel LW, Grindey GB, Plunkett W (1991) Action of 2′,2′-difluorodeoxycytidine on DNA synthesis. Clin Cancer Res 51:6110–6117
Matsuda A, Sasaki T (2004) Antitumor activity of sugar-modified cytosine nucleosides. Cancer Sci 95:105–111
Azuma A, Nakajima Y, Nishizono N et al (1993) Nucleosides and nucleotides. 122. 2′-C-cyano-2′-deoxy-1-beta-D-arabinofuranosylcytosine and its derivatives. A new class of nucleoside with a broad antitumor spectrum. J Med Chem 36:4183–4189
Azuma A, Hanaoka K, Kurihara A et al (1995) Nucleosides and nucleotides. 141. Chemical stability of a new antitumor nucleoside, 2′-C-cyano-2′-deoxy-1-beta-D-arabino- pentofuranosylcytosine in alkaline medium: formation of 2′-C-cyano-2′-deoxy-1-beta-D-ribo- pentofuranosylcytosine and its antitumor activity. J Med Chem 38:3391–3397
Azuma A, Huang P, Matsuda A, Plunkett W (2001) 2′-C-cyano-2′-deoxy-1-beta-D-arabino-pentofuranosylcytosine: a novel anticancer nucleoside analog that causes both DNA strand breaks and G(2) arrest. Mol Pharmacol 59:725–731
Matsuda A, Azuma A (1995) 2′-C-Cyano-2′-deoxy-1-β-D-arabinofuranosylcytosine (CNDAC): A mechanism-based DNA-strand-breaking antitumor nucleoside. Nucleosides Nucleotides 14:461–471
Hanaoka K, Suzuki M, Kobayashi T et al (1999) Antitumor activity and novel DNA-self-strand-breaking mechanism of CNDAC (1-(2-C-cyano-2-deoxy-beta-D-arabino-pentofuranosyl) cytosine) and its N4-palmitoyl derivative (CS-682). Int J Cancer 82:226–236
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Cancer Therapy Evaluation Program, Common Terminology Criteria for Adverse Events, Version 3.0, DCTD, NCI, NIH, DHHS. March 31, 2003 (http://ctep.cancer.gov), Publish Date: August 9, 2006
Yue L, Saikawa Y, Ota K et al (2003) A functional single-nucleotide polymorphism in the human cytidine deaminase gene contributing to ara-C sensitivity. Pharmacogenetics 13(1):29–38
Hartmann A, Agurell E, Beevers C et al (2003) Recommendations for conducting the in vivo alkaline Comet assay. 4th International Comet Assay Workshop. Mutagenesis 18:45–51
Olive PL, Banáth JP, Durand RE (1990) Heterogeneity in radiation-induced DNA damage and repair in tumor and normal cells measured using the "comet" assay. Radiat Res 122:86–94
Gilbert J, Carducci MA, Baker SD, Dees EC, Donehower R (2006) A Phase I study of the oral antimetabolite, CS-682, administered once daily 5 days per week in patients with refractory solid tumor. Investig New Drugs 24:499–508
Delaunoit T, Burch PA, Reid JM et al (2006) A phase I clinical and pharmacokinetic study of CS-682 administered orally in advanced malignant solid tumors. Investig New Drugs 24:327–333
Yonemori K, Ueno H, Okusaka T et al (2005) Severe drug toxicity associated with a single-nucleotide polymorphism of the cytidine deaminase gene in a Japanese cancer patient treated with gemcitabine plus cisplatin. Clin Cancer Res 11:2620–2624
Sugiyama E, Kaniwa N, Kim SR et al (2007) Pharmacokinetics of gemcitabine in Japanese cancer patients: the impact of a cytidine deaminase polymorphism. J Clin Oncol 25:32
Fukunaga AK, Marsh S, Murry DJ, Hurley TD, McLeod HL (2004) Identification and analysis of single-nucleotide polymorphisms in the gemcitabine pharmacologic pathway. Pharmacogenomics J 4(5):307–314
Ge Y, Jensen TL, Stout ML et al (2004) The role of cytidine deaminase and GATA1 mutations in the increased cytosine arabinoside sensitivity of Down syndrome myeloblasts and leukemia cell lines. Cancer Res 64(2):728–735
Schröder JK, Kirch C, Seeber S, Schütte J (1998) Structural and functional analysis of the cytidine deaminase gene in patients with acute myeloid leukemia. Br J Haematol 103(4):1096–1103
Ogawa M, Hori H, Ohta T, Onozato K, Miyahara M, Komada Y (2005) Sensitivity to gemcitabine and its metabolizing enzymes in neuroblastoma. Clin Cancer Res 11(9):3485–3493
Galmarini CM, Thomas X, Calvo F et al (2002) Potential mechanisms of resistance to cytarabine in AML patients. Leuk Res 26(7):621–629
Parker RJ, Gill I, Tarone R et al (1991) Platinum-DNA damage in leukocyte DNA of patients receiving carboplatin and cisplatin chemotherapy, measured by atomic absorption spectrometry. Carcinogenesis 12(7):1253–1258
Hartley JM, Spanswick VJ, Gander M et al (1999) Measurement of DNA cross-linking in patients on ifosfamide therapy using the single cell gel electrophoresis (comet) assay. Clin Cancer Res 5(3):507–512
Kopjar N, Garaj-Vrhovac V, Milas I (2002) Assessment of chemotherapy-induced DNA damage in peripheral blood leukocytes of cancer patients using the alkaline comet assay. Teratog Carcinog Mutagen 22(1):13–30
Plummer ER, Middleton MR, Jones C et al (2005) Temozolomide pharmacodynamics in patients with metastatic melanoma: dna damage and activity of repair enzymes O6-alkylguanine alkyltransferase and poly(ADP-ribose) polymerase-1. Clin Cancer Res 11(9):3402–3409
Acknowledgments
We thank all patients who participated in these studies. We thank Kazuhito Arakawa, Takashi Arima, Hideki Kawamata, Kazuaki Matsuoka, Hideki Nagase and Toshiyuki Toko for assistant in the clinical preparation and the data analysis.
Funding
The research was supported by Delta-Fly Pharma Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no potential conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent to participate in the study was obtained from each patient before study enrollment and the performance of any study-specific procedures.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sankhala, K., Takimoto, C.H., Mita, A.C. et al. Two phase I, pharmacokinetic, and pharmacodynamic studies of DFP-10917, a novel nucleoside analog with 14-day and 7-day continuous infusion schedules. Invest New Drugs 37, 76–86 (2019). https://doi.org/10.1007/s10637-018-0602-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-018-0602-0