Summary
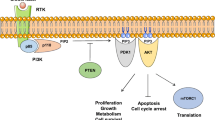
Breast cancer has been emerging as a most common threat among women, thus many efforts were made to find drugs for fighting breast cancer. So far, PI3K (Phosphatidylinositol-4,5-bisphosphate 3-kinase) inhibitors have been believed to be effective drugs until frequent resistance emerged. Recently, PI3K H1047R mutation has been reported to sensitize breast cancer cells to PI3K inhibition by aspirin. Considering aspirin activates AMPK (AMP-activated protein kinase) simultaneously, it is possible that AMPK activators and PI3K inhibitors can synergistically inhibit breast cancers. Here we clearly observed synergistic suppression of cell growth in all three breast cancer cell lines (MCF-7, MDA-MB-361 and HCC38) when co-treating cells with PI3K inhibitor GDC-0941 and AMPK activator AICAR (5-Aminoimidazole-4-carboxamide ribonucleotide). What is more, it is rather remarkable that the synergistic effect was much more dramatic in PIK3CA (PI3K catalytic subunit alpha) mutated (E545K) cells (MCF-7 and MDA-MB-361) than in PIK3CA wild-type cells (HCC38), which implied there is a relationship between PI3K genetic status and the efficacy of combination therapy. By using PIK3CA wild-type isogenic MCF-7 cell line, which exhibited attenuated cell proliferation compared with the parental MCF-7 cell line, we found endogenous reverse mutation of PIK3CA E545K alleles to wild-type sequence in MCF-7 cells dramatically impaired the synergy of PI3Ki&Ka (combinatorial PI3K inhibition and AMPK activation). Furthermore, PI3Ki&Ka significantly attenuated tumorigenesis of parental MCF-7 cells but not PIK3CA wild-type isogenic MCF-7 cells in tumor xenograft models. Taken together, our results suggest a promising precision therapy of PI3Ki&Ka in PIK3CA mutant breast cancers.




Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65(1):5–29. https://doi.org/10.3322/caac.21254
Lawrence MS, Stojanov P, Mermel CH, Robinson JT, Garraway LA, Golub TR, Meyerson M, Gabriel SB, Lander ES, Getz G (2014) Discovery and saturation analysis of cancer genes across 21 tumour types. Nature 505(7484):495–501. https://doi.org/10.1038/nature12912
Massacesi C, Di Tomaso E, Urban P, Germa C, Quadt C, Trandafir L, Aimone P, Fretault N, Dharan B, Tavorath R, Hirawat S (2016) PI3K inhibitors as new cancer therapeutics: implications for clinical trial design. Oncotargets Ther 9:203–210. https://doi.org/10.2147/Ott.S89967
Dienstmann R, Rodon J, Serra V, Tabernero J (2014) Picking the point of inhibition: a comparative review of PI3K/AKT/mTOR pathway inhibitors. Mol Cancer Ther 13(5):1021–1031. https://doi.org/10.1158/1535-7163.Mct-13-0639
Muranen T, Selfors LM, Worster DT, Iwanicki MP, Song L, Morales FC, Gao S, Mills GB, Brugge JS (2012) Inhibition of PI3K/mTOR leads to adaptive resistance in matrix-attached cancer cells. Cancer Cell 21(2):227–239. https://doi.org/10.1016/j.ccr.2011.12.024
Edgar KA, Wallin JJ, Berry M, Lee LB, Prior WW, Sampath D, Friedman LS, Belvin M (2010) Isoform-specific phosphoinositide 3-kinase inhibitors exert distinct effects in solid tumors. Cancer Res 70(3):1164–1172. https://doi.org/10.1158/0008-5472.CAN-09-2525
Folkes AJ, Ahmadi K, Alderton WK, Alix S, Baker SJ, Box G, Chuckowree IS, Clarke PA, Depledge P, Eccles SA, Friedman LS, Hayes A, Hancox TC, Kugendradas A, Lensun L, Moore P, Olivero AG, Pang J, Patel S, Pergl-Wilson GH, Raynaud FI, Robson A, Saghir N, Salphati L, Sohal S, Ultsch MH, Valenti M, Wallweber HJA, Wan NC, Wiesmann C, Workman P, Zhyvoloup A, Zvelebil MJ, Shuttleworth SJ (2008) The identification of 2-(1H-indazol-4-yl)-6-(4-methanesulfonyl-piperazin-1-ylmethyl)-4-morpholin-4-yl-thieno[3,2-d]pyrimidine (GDC-0941) as a potent, selective, orally bioavailable inhibitor of class I PI3 kinase for the treatment of cancer. J Med Chem 51(18):5522–5532. https://doi.org/10.1021/jm800295d
Schmid P, Pinder SE, Wheatley D, Macaskill J, Zammit C, Hu J, Price R, Bundred N, Hadad S, Shia A, Sarker SJ, Lim L, Gazinska P, Woodman N, Korbie D, Trau M, Mainwaring P, Gendreau S, Lackner MR, Derynck M, Wilson TR, Butler H, Earl G, Parker P, Purushotham A, Thompson A (2016) Phase II randomized preoperative window-of-opportunity study of the PI3K inhibitor Pictilisib plus Anastrozole compared with Anastrozole alone in patients with estrogen receptor-positive breast cancer. J Clin Oncol 34(17):1987–1994. https://doi.org/10.1200/JCO.2015.63.9179
McConnell JR, McAlpine SR (2013) Heat shock proteins 27, 40, and 70 as combinational and dual therapeutic cancer targets. Bioorg Med Chem Lett 23(7):1923–1928. https://doi.org/10.1016/j.bmcl.2013.02.014
Shi S, Yao W, Xu J, Long J, Liu C, Yu X (2012) Combinational therapy: new hope for pancreatic cancer? Cancer Lett 317(2):127–135. https://doi.org/10.1016/j.canlet.2011.11.029
Jeon SM (2016) Regulation and function of AMPK in physiology and diseases. Exp Mol Med 48(7):e245. https://doi.org/10.1038/emm.2016.81
El-Masry OS, Brown BL, Dobson PR (2012) Effects of activation of AMPK on human breast cancer cell lines with different genetic backgrounds. Oncol Lett 3(1):224–228. https://doi.org/10.3892/ol.2011.458
Theodoropoulou S, Brodowska K, Kayama M, Morizane Y, Miller JW, Gragoudas ES, Vavvas DG (2013) Aminoimidazole carboxamide ribonucleotide (AICAR) inhibits the growth of retinoblastoma in vivo by decreasing angiogenesis and inducing apoptosis. PLoS One 8(1):e52852. https://doi.org/10.1371/journal.pone.0052852
Xiong L, Liu Z, Ouyang G, Lin L, Huang H, Kang H, Chen W, Miao X, Wen Y (2017) Autophagy inhibition enhances photocytotoxicity of Photosan-II in human colorectal cancer cells. Oncotarget 8(4):6419–6432. https://doi.org/10.18632/oncotarget.14117
Pawalowska M, Markowska A (2012) The influence of metformin in the etiology of selected cancers. Contemp Oncol (Pozn) 16(3):223–229. https://doi.org/10.5114/wo.2012.29289
Hallstrom TC, Mori S, Nevins JR (2008) An E2F1-dependent gene expression program that determines the balance between proliferation and cell death. Cancer Cell 13(1):11–22. https://doi.org/10.1016/j.ccr.2007.11.031
Jose C, Hebert-Chatelain E, Bellance N, Larendra A, Su M, Nouette-Gaulain K, Rossignol R (2011) AICAR inhibits cancer cell growth and triggers cell-type distinct effects on OXPHOS biogenesis, oxidative stress and Akt activation. Biochim Biophys Acta 1807(6):707–718. https://doi.org/10.1016/j.bbabio.2010.12.002
El-Masry OS, Al-Sakkaf K, Brown BL, Dobson PR (2015) Differential crosstalk between the AMPK and PI3K/Akt pathways in breast cancer cells of differing genotypes: leptin inhibits the effectiveness of AMPK activation. Oncol Rep 34(4):1675–1680. https://doi.org/10.3892/or.2015.4198
Suzuki T, Bridges D, Nakada D, Skiniotis G, Morrison SJ, Lin JD, Saltiel AR, Inoki K (2013) Inhibition of AMPK catabolic action by GSK3. Mol Cell 50(3):407–419. https://doi.org/10.1016/j.molcel.2013.03.022
Henry WS, Laszewski T, Tsang T, Beca F, Beck AH, McAllister SS, Toker A (2017) Aspirin suppresses growth in PI3K-mutant breast cancer by activating AMPK and inhibiting mTORC1 signaling. Cancer Res 77(3):790–801. https://doi.org/10.1158/0008-5472.CAN-16-2400
Bitto A, Sell C, Crowe E, Lorenzini A, Malaguti M, Hrelia S, Torres C (2010) Stress-induced senescence in human and rodent astrocytes. Exp Cell Res 316(17):2961–2968. https://doi.org/10.1016/j.yexcr.2010.06.021
Wallin JJ, Guan J, Prior WW, Lee LB, Berry L, Belmont LD, Koeppen H, Belvin M, Friedman LS, Sampath D (2012) GDC-0941, a novel class I selective PI3K inhibitor, enhances the efficacy of docetaxel in human breast cancer models by increasing cell death in vitro and in vivo. Clin Cancer Res 18(14):3901–3911. https://doi.org/10.1158/1078-0432.CCR-11-2088
O'Brien C, Wallin JJ, Sampath D, GuhaThakurta D, Savage H, Punnoose EA, Guan J, Berry L, Prior WW, Amler LC, Belvin M, Friedman LS, Lackner MR (2010) Predictive biomarkers of sensitivity to the phosphatidylinositol 3′ kinase inhibitor GDC-0941 in breast cancer preclinical models. Clin Cancer Res 16(14):3670–3683. https://doi.org/10.1158/1078-0432.CCR-09-2828
Beaver JA, Gustin JP, Yi KH, Rajpurohit A, Thomas M, Gilbert SF, Rosen DM, Ho Park B, Lauring J (2013) PIK3CA and AKT1 mutations have distinct effects on sensitivity to targeted pathway inhibitors in an isogenic luminal breast cancer model system. Clin Cancer Res 19(19):5413–5422. https://doi.org/10.1158/1078-0432.CCR-13-0884
Yang XL, Lin FJ, Guo YJ, Shao ZM, Ou ZL (2014) Gemcitabine resistance in breast cancer cells regulated by PI3K/AKT-mediated cellular proliferation exerts negative feedback via the MEK/MAPK and mTOR pathways. Onco Targets Ther 7:1033–1042. https://doi.org/10.2147/OTT.S63145
Samuels Y, Diaz LA, Jr., Schmidt-Kittler O, Cummins JM, Delong L, Cheong I, Rago C, Huso DL, Lengauer C, Kinzler KW, Vogelstein B, Velculescu VE (2005) Mutant PIK3CA promotes cell growth and invasion of human cancer cells. Cancer Cell 7 (6):561–573. doi:https://doi.org/10.1016/j.ccr.2005.05.014
Knudsen ES, Knudsen KE (2008) Tailoring to RB: tumour suppressor status and therapeutic response. Nat Rev Cancer 8(9):714–724. https://doi.org/10.1038/nrc2401
Weinberg RA (1995) The retinoblastoma protein and cell cycle control. Cell 81(3):323–330
Boulares AH, Yakovlev AG, Ivanova V, Stoica BA, Wang G, Iyer S, Smulson M (1999) Role of poly(ADP-ribose) polymerase (PARP) cleavage in apoptosis. Caspase 3-resistant PARP mutant increases rates of apoptosis in transfected cells. J Biol Chem 274(33):22932–22940
Will M, Qin AC, Toy W, Yao Z, Rodrik-Outmezguine V, Schneider C, Huang X, Monian P, Jiang X, de Stanchina E, Baselga J, Liu N, Chandarlapaty S, Rosen N (2014) Rapid induction of apoptosis by PI3K inhibitors is dependent upon their transient inhibition of RAS-ERK signaling. Cancer Discov 4(3):334–347. https://doi.org/10.1158/2159-8290.CD-13-0611
Queiroz EA, Puukila S, Eichler R, Sampaio SC, Forsyth HL, Lees SJ, Barbosa AM, Dekker RF, Fortes ZB, Khaper N (2014) Metformin induces apoptosis and cell cycle arrest mediated by oxidative stress, AMPK and FOXO3a in MCF-7 breast cancer cells. PLoS One 9(5):e98207. https://doi.org/10.1371/journal.pone.0098207
Vora SR, Juric D, Kim N, Mino-Kenudson M, Huynh T, Costa C, Lockerman EL, Pollack SF, Liu M, Li XY, Lehar J, Wiesmann M, Wartmann M, Chen Y, Cao ZA, Pinzon-Ortiz M, Kim S, Schlegel R, Huang A, Engelman JA (2014) CDK 4/6 inhibitors sensitize PIK3CA mutant breast cancer to PI3K inhibitors. Cancer Cell 26(1):136–149. https://doi.org/10.1016/j.ccr.2014.05.020
Abraham J (2015) PI3K/AKT/mTOR pathway inhibitors: the ideal combination partners for breast cancer therapies? Expert Rev Anticancer Ther 15(1):51–68. https://doi.org/10.1586/14737140.2015.961429
Liang X, Wang P, Gao Q, Tao X (2014) Exogenous activation of LKB1/AMPK signaling induces G(1) arrest in cells with endogenous LKB1 expression. Mol Med Rep 9(3):1019–1024. https://doi.org/10.3892/mmr.2014.1916
Eroles P, Bosch A, Perez-Fidalgo JA, Lluch A (2012) Molecular biology in breast cancer: intrinsic subtypes and signaling pathways. Cancer Treat Rev 38(6):698–707. https://doi.org/10.1016/j.ctrv.2011.11.005
Cancer Genome Atlas N (2012) Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70. https://doi.org/10.1038/nature11412
Stephens PJ, Tarpey PS, Davies H, Van Loo P, Greenman C, Wedge DC, Nik-Zainal S, Martin S, Varela I, Bignell GR, Yates LR, Papaemmanuil E, Beare D, Butler A, Cheverton A, Gamble J, Hinton J, Jia MM, Jayakumar A, Jones D, Latimer C, Lau KW, McLaren S, McBride DJ, Menzies A, Mudie L, Raine K, Rad R, Chapman MS, Teague J, Easton D, Langerod A, Lee MTM, Shen CY, Tee BTK, Huimin BW, Broeks A, Vargas AC, Turashvili G, Martens J, Fatima A, Miron P, Chin SF, Thomas G, Boyault S, Mariani O, Lakhani SR, van de Vijver M, Van Tveer L, Foekens J, Desmedt C, Sotiriou C, Tutt A, Caldas C, Reis JS, SAJR A, Salomon AV, Borresen-Dale AL, Richardson AL, Campbell PJ, Futreal PA, Stratton MR, Osbreac (2012) The landscape of cancer genes and mutational processes in breast cancer. Nature 486(7403):400–404. https://doi.org/10.1038/nature11017
Zheng D, Zhu G, Liao S, Yi W, Luo G, He J, Pei Z, Li G, Zhou Y (2015) Dysregulation of the PI3K/Akt signaling pathway affects cell cycle and apoptosis of side population cells in nasopharyngeal carcinoma. Oncol Lett 10(1):182–188. https://doi.org/10.3892/ol.2015.3218
Isakoff SJ, Engelman JA, Irie HY, Luo J, Brachmann SM, Pearline RV, Cantley LC, Brugge JS (2005) Breast cancer-associated PIK3CA mutations are oncogenic in mammary epithelial cells. Cancer Res 65(23):10992–11000. https://doi.org/10.1158/0008-5472.CAN-05-2612
Eichhorn PJ, Gili M, Scaltriti M, Serra V, Guzman M, Nijkamp W, Beijersbergen RL, Valero V, Seoane J, Bernards R, Baselga J (2008) Phosphatidylinositol 3-kinase hyperactivation results in lapatinib resistance that is reversed by the mTOR/phosphatidylinositol 3-kinase inhibitor NVP-BEZ235. Cancer Res 68(22):9221–9230. https://doi.org/10.1158/0008-5472.CAN-08-1740
Acknowledgements
We thank Dr. Haian Fu (Emory University, Atlanta, USA) for kindly giving us the parental MCF-7 and PIK3CA wild-type isogenic MCF-7 cell lines. This work is funded by the Research Foundation of Education Bureau of Hunan Province, China (Grant No. 2050205).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, S., Tang, Y., Yan, M. et al. PIK3CA mutation sensitizes breast cancer cells to synergistic therapy of PI3K inhibition and AMPK activation. Invest New Drugs 36, 763–772 (2018). https://doi.org/10.1007/s10637-018-0563-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-018-0563-3