Summary
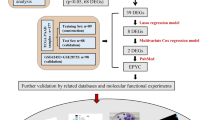
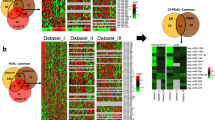
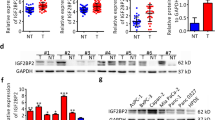
Despite great improvements in surgical procedures and chemotherapy, pancreatic cancer remains one of the most aggressive and fatal human malignancies, with a low 5-year survival rate. Therefore, novel therapeutic strategies for the prevention and treatment of pancreatic cancer are urgently needed. The present study aimed to investigate the mechanisms by which metformin exerts its anticancer effects on the microRNA-mRNA interactions in human pancreatic cancer. Microarray and systematic analyses revealed that the anti-pancreatic cancer effects of metformin were correlated with 3 up-regulated microRNAs and 4 of their target mRNAs. In addition, the microarray and systematic analyses ultimately demonstrated that 3 microRNAs regulated 4 key mRNAs in a sub-pathway of pancreatic cancer and then affected growth, angiogenesis, and apoptosis. This finding may provide a deeper understanding of the mechanisms by which metformin suppresses proliferation and angiogenesis and promotes apoptosis in pancreatic cancer cells. Collectively, this experiment improves the understanding of the mechanisms by which metformin suppresses pancreatic cancer and indicates that metformin, the most commonly used drug for the treatment of diabetes mellitus, may be a promising candidate agent for the treatment of pancreatic cancer.




Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics. CA Cancer J Clin 66:7–30
Ryan DP, Hong TS, Bardeesy N (2014) Pancreatic adenocarcinoma. N Engl J Med 371:2140–2141
Hartwig W, Werner J, Jager D, Debus J, Buchler MW (2013) Improvement of surgical results for pancreatic cancer. Lancet Oncol 14:e476–e485
Yue W, Zheng X, Lin Y, Yang CS, Xu Q, Carpizo D, Huang H, DiPaola RS, Tan XL (2015) Metformin combined with aspirin significantly inhibit pancreatic cancer cell growth in vitro and in vivo by suppressing anti-apoptotic proteins mcl-1 and bcl-2. Oncotarget 6:21208–21224
Cheng G, Lanza-Jacoby S (2015) Metformin decreases growth of pancreatic cancer cells by decreasing reactive oxygen species: role of nox4. Biochem Biophys Res Commun 465:41–46
Incio J, Suboj P, Chin SM, Vardam-Kaur T, Liu H, Hato T, Babykutty S, Chen I, Deshpande V, Jain RK, Fukumura D (2015) Metformin reduces desmoplasia in pancreatic cancer by reprogramming stellate cells and tumor-associated macrophages. PLoS One 10:e0141392
Wang J, Gao Q, Wang D, Wang Z, Hu C (2015) Metformin inhibits growth of lung adenocarcinoma cells by inducing apoptosis via the mitochondria-mediated pathway. Oncol Lett 10:1343–1349
Bao B, Azmi AS, Ali S, Zaiem F, Sarkar FH (2014) Metformin may function as anti-cancer agent via targeting cancer stem cells: the potential biological significance of tumor-associated mirnas in breast and pancreatic cancers. Ann Transl Med 2:59
Liu Z, Qi S, Zhao X, Li M, Ding S, Lu J, Zhang H (2016) Metformin inhibits 17beta-estradiol-induced epithelial-to-mesenchymal transition via betaklotho-related erk1/2 signaling and ampkalpha signaling in endometrial adenocarcinoma cells. Oncotarget 7:21315–21331
Fujimori T, Kato K, Fujihara S, Iwama H, Yamashita T, Kobayashi K, Kamada H, Morishita A, Kobara H, Mori H, Okano K, Suzuki Y, Masaki T (2015) Antitumor effect of metformin on cholangiocarcinoma: in vitro and in vivo studies. Oncol Rep 34:2987–2996
Wang J, Li G, Wang Y, Tang S, Sun X, Feng X, Li Y, Bao G, Li P, Mao X, Wang M, Liu P (2015) Suppression of tumor angiogenesis by metformin treatment via a mechanism linked to targeting of her2/hif-1alpha/vegf secretion axis. Oncotarget 6:44579–44592
Ren P, Gong F, Zhang Y, Jiang J, Zhang H (2015) Microrna-92a promotes growth, metastasis, and chemoresistance in non-small cell lung cancer cells by targeting pten. Tumour Biol 37:3215–3225
Liu Y, Wang Y, Sun X, Mei C, Wang L, Li Z, Zha X (2015) Mir-449a promotes liver cancer cell apoptosis by down-regulation of calpain6 and pou2f1. Oncotarget 7:13491–13501
Wang N, Wei H, Yin D, Lu Y, Zhang Y, Zhang Q, Ma X, Zhang S (2015) Microrna-195 inhibits proliferation of cervical cancer cells by targeting cyclin d1a. Tumour Biol 37:4711–4720
Li Y, Wang Y, Li L, Kong R, Pan S, Ji L, Liu H, Chen H, Sun B (2016) Hyperoside induces apoptosis and inhibits growth in pancreatic cancer via bcl-2 family and nf-kappab signaling pathway both in vitro and in vivo. Tumour Biol 37:7345–7355
Wang Y, Zhou Y, Jia G, Han B, Liu J, Teng Y, Lv J, Song Z, Li Y, Ji L, Pan S, Jiang H, Sun B (2014) Shikonin suppresses tumor growth and synergizes with gemcitabine in a pancreatic cancer xenograft model involvement of nf-kappab signaling pathway. Biochem Pharmacol 88:322–333
Wang Y, Zhou Y, Zhou H, Jia G, Liu J, Han B, Cheng Z, Jiang H, Pan S, Sun B (2012) Pristimerin causes g1 arrest, induces apoptosis, and enhances the chemosensitivity to gemcitabine in pancreatic cancer cells. PLoS One 7:e43826
Wang YW, Wang SJ, Zhou YN, Pan SH, Sun B (2012) Escin augments the efficacy of gemcitabine through down-regulation of nuclear factor-kappab and nuclear factor-kappab-regulated gene products in pancreatic cancer both in vitro and in vivo. J Cancer Res Clin Oncol 138:785–797
Shen Y, Pan Y, Xu L, Chen L, Liu L, Chen H, Chen Z, Meng Z (2015) Identifying microrna-mrna regulatory network in gemcitabine-resistant cells derived from human pancreatic cancer cells. Tumour Biol 36:4525–4534
Saif MW (2010) Options for the treatment of gemcitabine-resistant advanced pancreatic cancer: are we there yet? JOP 11:288–289
Neumann M, Naumann M (2007) Beyond ikappabs: alternative regulation of nf-kappab activity. FASEB J 21:2642–2654
Gilmore TD (2006) Introduction to nf-kappab: players, pathways, perspectives. Oncogene 25:6680–6684
Zhang JJ, Zhu Y, Xie KL, Peng YP, Tao JQ, Tang J, Li Z, ZK X, Dai CC, Qian ZY, Jiang KR, JL W, Gao WT, Du Q, Miao Y (2014) Yin yang-1 suppresses invasion and metastasis of pancreatic ductal adenocarcinoma by downregulating mmp10 in a muc4/erbb2/p38/mef2c- dependent mechanism. Mol Cancer 13:130
Qiu X, Cheng JC, Klausen C, Fan Q, Chang HM, So WK, Leung PC (2015) Transforming growth factor-alpha induces human ovarian cancer cell invasion by down-regulating e-cadherin in a snail-independent manner. Biochem Biophys Res Commun 461:128–135
Chen H, Li L, Wang S, Lei Y, Ge Q, Lv N, Zhou X, Chen C (2014) Reduced mir-126 expression facilitates angiogenesis of gastric cancer through its regulation on vegf-a. Oncotarget 5:11873–11885
Yang G, Zhang P, Lv A, Liu Y, Wang G (2016) Mir-205 functions as a tumor suppressor via targeting tgf-alpha in osteosarcoma. Exp Mol Pathol 100:160–166
Lawrence DA (1985) Transforming growth factors--an overview. Biol Cell 53:93–98
Salomon DS, Kim N, Saeki T, Ciardiello F (1990) Transforming growth factor-alpha: an oncodevelopmental growth factor. Cancer Cells 2:389–397
Funding
This work was supported by the National Nature Scientific Foundation of China (No. 81372613 and 81,302,057), The National High Technology Research and Development Program of China (No. 2014AA020609), and the Youth Science Foundation of Heilongjiang Province (No. QC2012C042).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yilong Li declares that he has no conflicts of interest. Le Li declares that he has no conflicts of interest. Guangquan Zhang declares that he has no conflicts of interest. Yongwei Wang declares that he has no conflicts of interest. Hua Chen declares that he has no conflicts of interest. Rui Kong declares that he has no conflicts of interest. Shangha Pan declares that he has no conflicts of interest. Bei Sun declares that he has no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in this study.
Rights and permissions
About this article
Cite this article
Li, Y., Li, L., Zhang, G. et al. Crucial microRNAs and genes in metformin’s anti-pancreatic cancer effect explored by microRNA-mRNA integrated analysis. Invest New Drugs 36, 20–27 (2018). https://doi.org/10.1007/s10637-017-0508-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-017-0508-2