Summary
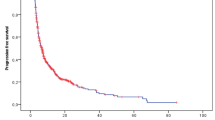
Background The study was aim to assess the efficacy and safety of sunitinib on 14 Chinese patients with locally unresectable or metastatic Alveolar Soft Part Sarcoma (ASPS) at two institutions retrospectively. Methods Patients were treated with 37.5 mg of sunitinib once daily continuously without a scheduled off-treatment period. Dose holds or reductions were recommended for grade 3 AEs but were required for grade 4 AEs. Restarting treatment of sunitinib was allowed when AEs returned back to grade 1 or disappeared. The treatment was continued until progression disease (PD), unacceptable toxicity or death. Results From January 2011 to December 2015, 14 patients with unresectable or metastatic ASPS received sunitinib treatment. Among them, 4 patients achieved partial remission (PR), and 10 patients achieved stable disease (SD). Median progression free survival (PFS) was 41.0 months (95 % CI: 7.7 to 74.4 month). Median overall survival (OS) was not reached. The 1- and 4-year OS rates were 90.0 % and 60.0 % respectively. Two patients with primarily unresectable ASPS received complete surgical resection after neoadjuvant treatment of sunitinib. The majority of toxicities encountered were grade 1 or 2 and manageable. The most common adverse events (AEs) were bleeding (35.7 %), hair & skin color change (37.5 %) and mucositis (28.6 %). Conclusions Sunitinib is effective in locally unresectable or metastatic ASPS with a good safety profile. Neoadjuvant treatment with sunitinib improves the chance of resection for patients with locally advanced ASPS.


Similar content being viewed by others
References
Portera CA Jr., Ho V, Patel SR, Hunt KK, Feig BW, Respondek PM, Yasko AW, Benjamin RS, Pollock RE, Pisters PW (2001) Alveolar soft part sarcoma: clinical course and patterns of metastasis in 70 patients treated at a single institution. Cancer 91(3):585–591
Ladanyi M, Lui MY, Antonescu CR, Krause-Boehm A, Meindl A, Argani P, Healey JH, Ueda T, Yoshikawa H, Meloni-Ehrig A, Sorensen PH, Mertens F, Mandahl N, van den Berghe H, Sciot R, Dal Cin P, Bridge J (2001) The der(17)t(X;17)(p11;q25) of human alveolar soft part sarcoma fuses the TFE3 transcription factor gene to ASPL, a novel gene at 17q25. Oncogene 20(1):48–57
Ogose A, Yazawa Y, Ueda T, Hotta T, Kawashima H, Hatano H, Morita T (2003) Alveolar soft part sarcoma in Japan: multi-institutional study of 57 patients from the Japanese Musculoskeletal Oncology Group. Oncology 65(1):7–13
Reichardt P, Lindner T, Pink D, Thuss-Patience PC, Kretzschmar A, Dorken B (2003) Chemotherapy in alveolar soft part sarcomas. What do we know? Eur J Cancer 39(11):1511–1516
Chow LQ, Eckhardt SG (2007) Sunitinib: from rational design to clinical efficacy. J Clin Oncol 25(7):884–896
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Oudard S, Negrier S, Szczylik C, Pili R, Bjarnason GA, Garcia-del-Muro X, Sosman JA, Solska E, Wilding G, Thompson JA, Kim ST, Chen I, Huang X, Figlin RA (2009) Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol 27(22):3584–3590
Demetri GD, van Oosterom AT, Garrett CR, Blackstein ME, Shah MH, Verweij J, McArthur G, Judson IR, Heinrich MC, Morgan JA, Desai J, Fletcher CD, George S, Bello CL, Huang X, Baum CM, Casali PG (2006) Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet 368(9544):1329–1338
Demetri GD, Garrett CR, Schoffski P, Shah MH, Verweij J, Leyvraz S, Hurwitz HI, Pousa AL, Le Cesne A, Goldstein D, Paz-Ares L, Blay JY, McArthur GA, Xu QC, Huang X, Harmon CS, Tassell V, Cohen DP, Casali PG (2012) Complete longitudinal analyses of the randomized, placebo-controlled, phase III trial of sunitinib in patients with gastrointestinal stromal tumor following imatinib failure. Clin Cancer Res 18(11):3170–3179
Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas C, Valle J, Metrakos P, Smith D, Vinik A, Chen JS, Horsch D, Hammel P, Wiedenmann B, Van Cutsem E, Patyna S, Lu DR, Blanckmeister C, Chao R, Ruszniewski P (2011) Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N Engl J Med 364(6):501–513
George S, Merriam P, Maki RG, Van den Abbeele AD, Yap JT, Akhurst T, Harmon DC, Bhuchar G, O'Mara MM, D'Adamo DR, Morgan J, Schwartz GK, Wagner AJ, Butrynski JE, Demetri GD, Keohan ML (2009) Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol 27(19):3154–3160
Stacchiotti S, Negri T, Zaffaroni N, Palassini E, Morosi C, Brich S, Conca E, Bozzi F, Cassinelli G, Gronchi A, Casali PG, Pilotti S (2011) Sunitinib in advanced alveolar soft part sarcoma: evidence of a direct antitumor effect. Ann Oncol 22(7):1682–1690
Stroobants S, Goeminne J, Seegers M, Dimitrijevic S, Dupont P, Nuyts J, Martens M, van den Borne B, Cole P, Sciot R, Dumez H, Silberman S, Mortelmans L, van Oosterom A (2003) 18FDG-Positron emission tomography for the early prediction of response in advanced soft tissue sarcoma treated with imatinib mesylate (Glivec). Eur J Cancer 39(14):2012–2020
Choi H (2005) Critical issues in response evaluation on computed tomography: lessons from the gastrointestinal stromal tumor model. Curr Oncol Rep 7(4):307–311
Lazar AJ, Das P, Tuvin D, Korchin B, Zhu Q, Jin Z, Warneke CL, Zhang PS, Hernandez V, Lopez-Terrada D, Pisters PW, Pollock RE, Lev D (2007) Angiogenesis-promoting gene patterns in alveolar soft part sarcoma. Clin Cancer Res 13(24):7314–7321
Kummar S, Allen D, Monks A, Polley EC, Hose CD, Ivy SP, Turkbey IB, Lawrence S, Kinders RJ, Choyke P, Simon R, Steinberg SM, Doroshow JH, Helman L (2013) Cediranib for metastatic alveolar soft part sarcoma. J Clin Oncol 31(18):2296–2302
Wagner AJ, Goldberg JM, Dubois SG, Choy E, Rosen L, Pappo A, Geller J, Judson I, Hogg D, Senzer N, Davis IJ, Chai F, Waghorne C, Schwartz B, Demetri GD (2012) Tivantinib (ARQ 197), a selective inhibitor of MET, in patients with microphthalmia transcription factor-associated tumors: results of a multicenter phase 2 trial. Cancer 118(23):5894–5902
Kollmannsberger C, Soulieres D, Wong R, Scalera A, Gaspo R, Bjarnason G (2007) Sunitinib therapy for metastatic renal cell carcinoma: recommendations for management of side effects. Can Urol Assoc J 1(2 Suppl):S41–S54
Chu TF, Rupnick MA, Kerkela R, Dallabrida SM, Zurakowski D, Nguyen L, Woulfe K, Pravda E, Cassiola F, Desai J, George S, Morgan JA, Harris DM, Ismail NS, Chen JH, Schoen FJ, Van den Abbeele AD, Demetri GD, Force T, Chen MH (2007) Cardiotoxicity associated with tyrosine kinase inhibitor sunitinib. Lancet 370(9604):2011–2019
Folpe AL, Deyrup AT (2006) Alveolar soft-part sarcoma: a review and update. J Clin Pathol 59(11):1127–1132
Lieberman PH, Foote FW Jr., Stewart FW, Berg JW (1966) Alveolar soft-part sarcoma. JAMA 198(10):1047–1051
Acknowledgments
We thank the patients who shared their experiences with our oncologists, as well as the network of investigators. In particular, we would like to acknowledge Yunhua Lu for helping us with data collection.
Authors’s contribution
LZG and WCM made contributions to the conception, design and analysis of data. LT and LW carried out most of the data collection and drafted the main manuscript. LZG, WCM, WHJ and ZSJ revised the manuscript. AK and ZXW discussed the data. All authors have read and approved the manuscript for publication.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict interests
The authors declare that they have no competing interests.
Funding
None.
Ethical approval
For this type of study formal consent is not required.
Additional information
Ting Li and Lei Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, T., Wang, L., Wang, H. et al. A retrospective analysis of 14 consecutive Chinese patients with unresectable or metastatic alveolar soft part sarcoma treated with sunitinib. Invest New Drugs 34, 701–706 (2016). https://doi.org/10.1007/s10637-016-0390-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-016-0390-3