Summary
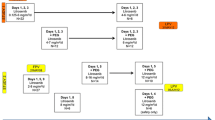
Objective Cobimetinib, a MEK1/2 inhibitor, was administered to patients with advanced solid tumors to assess safety, pharmacokinetics, pharmacodynamics, and anti-tumor activity. Methods For dose-escalation, a 3 + 3 design was used. Oral cobimetinib was administered once daily on a 21-day on/7-day off (21/7) or a 14-day on/14-day off (14/14) schedule. Serial plasma samples were collected for pharmacokinetic (PK) analysis on Day 1 and at steady state. In expansion stages, patients with RAS or RAF mutant tumors were treated at the maximum tolerated dose (MTD) of the 21/7 or 14/14 schedule. Results Ninety-seven patients received cobimetinib. In the 21/7 dose escalation, 36 patients enrolled in 8 cohorts (0.05 mg/kg–80 mg). Dose-limiting toxicities (DLTs) were Grade 4 hepatic encephalopathy, Grade 3 diarrhea, and Grade 3 rash. In the 14/14 dose escalation, 20 patients enrolled in 4 cohorts (60–125 mg). DLTs were Grade 3 rash and Grade 3 blurred vision associated with presence of reversible subretinal fluid. The MTD was 60 mg on 21/7 schedule and 100 mg on 14/14 schedule. Cobimetinib PK showed dose-proportional increases in exposure. The most frequent adverse events attributed to cobimetinib were diarrhea, rash, fatigue, edema, nausea, and vomiting. In patients treated at the 60-mg (21/7) or 100-mg (14/14) dose, one unconfirmed complete response and 6 confirmed partial responses were observed. All responses occurred in melanoma patients; 6 harbored the BRAFV600E mutation. Conclusions Cobimetinib is generally well tolerated and durable responses were observed in BRAFV600E mutant melanoma patients. Evaluation of cobimetinib in combination with other therapies is ongoing.



Similar content being viewed by others
References
Davies H, Bignell GR, Cox C, et al. (2002) Mutations of the BRAF gene in human cancer. Nature 417:949–954. doi:10.1111/ced.12015
Allen LF, Sebolt-Leopold J, Meyer MB (2003) CI-1040 (PD184352), a targeted signal transduction inhibitor of MEK (MAPKK). Semin Oncol 30(5 Suppl 16):105–116
Malumbres M, Barbacid M (2003) RAS oncogenes: the first 30 years. Nat Rev Cancer 3:459–65. Erratum in: Nat Rev Cancer 3:708
Bamford S, Dawson E, Forbes S, et al. (2004) The COSMIC (Catalogue of Somatic Mutations in Cancer) database and website. Br J Cancer 91:355–358
Karnoub A, Weinberg RA (2008) Ras oncogenes: split personalities. Nat Rev Mol Cell Biol 9:517–531. doi:10.1038/nrm2438
Hoeflich KP, Merchant M, Orr C, et al. (2012) Intermittent administration of MEK inhibitor GDC-0973 plus PI3K inhibitor GDC-0941 triggers robust apoptosis and tumor growth inhibition. Cancer Res 72:210–219. doi:10.1158/0008-5472.CAN-11-1515
McCubrey JA, Steelman LS, Chappell WH, et al. (2012) Advances in targeting signal transduction pathways. Oncotarget 3:1505–1521
Akinleye A, Furqan M, Mukhi N, Ravella P, Liu D (2013) MEK and the inhibitors: from bench to bedside. J Hematol Oncol 6:27. doi:10.1186/1756-8722-6-27
Wellbrock C, Karasarides M, Marais R (2004) The RAF proteins take centre stage. Nat Rev Mol Cell Biol 5:875–885
Solit DB, Garraway LA, Pratilas CA, et al. (2006) BRAF mutation predicts sensitivity to MEK inhibition. Nature 439:358–362
Roberts PJ, Der CJ (2007) Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 26:3291–3310
Hoeflich KP, O'Brien C, Boyd Z, et al. (2009) In vivo antitumor activity of MEK and phosphatidylinositol 3-kinase inhibitors in basal-like breast cancer models. Clin Cancer Res 15:4649–4664. doi:10.1158/1078-0432.CCR-09-0317
Hatzivassiliou G, Haling JR, Chen H, et al. (2013) Mechanism of MEK inhibition determines efficacy in mutant KRAS- versus BRAF-driven cancers. Nature 501:232–236. doi:10.1038/nature12441
Kim KB, Kefford R, Pavlick AC, et al. (2013) Phase II study of the MEK1/MEK2 inhibitor Trametinib in patients with metastatic BRAF-mutant cutaneous melanoma previously treated with or without a BRAF inhibitor. J Clin Oncol 31:482–489. doi:10.1200/JCO.2012.43.5966
Falchook GS, Lewis KD, Infante JR, et al. (2012) Activity of the oral MEK inhibitor trametinib in patients with advanced melanoma: a phase 1 dose-escalation trial. Lancet Oncol 13:782–789. doi:10.1016/S1470-2045(12)70269-3
Ascierto PA, Schadendorf D, Berking C, et al. (2013) MEK162 for patients with advanced melanoma harbouring NRAS or Val600 BRAF mutations: a non-randomised, open-label phase 2 study. Lancet Oncol 14:249–256. doi:10.1016/S1470-2045(13)70024-X
Farley J, Brady WE, Vathipadiekal V, et al. (2013) Selumetinib in women with recurrent low-grade serous carcinoma of the ovary or peritoneum: an open-label, single-arm, phase 2 study. Lancet Oncol 14:134–140. doi:10.1016/S1470-2045(12)70572-7
Bekaii-Saab T, Phelps MA, Li X, et al. (2011) Multi-institutional phase II study of selumetinib in patients with metastatic biliary cancers. J Clin Oncol 29:2357–2363. doi:10.1200/JCO.2010.33.9473
Ho AL, Grewal RK, Leboeuf R, et al. (2013) Selumetinib-enhanced radioiodine uptake in advanced thyroid cancer. N Engl J Med 368:623–632. doi:10.1056/NEJMoa1209288
Rosen L, LoRusso P, Ma WW et al (2011) A first-in-human Phase 1 study to evaluate the MEK1/2 inhibitor GDC-0973 administered daily in patients with advanced solid tumors [abstract]. In: Proceedings of the 102nd Annual Meeting of the American Association for Cancer Research; 2011 Apr 2–6; Orlando, FL. Philadelphia (PA): AACR; Cancer Res 71(8 Suppl):Abstract nr 4716. doi:10.1158/1538-7445.AM2011-4716
Wong H, Choo EF, Alicke B, et al. (2012) Antitumor activity of target and cytotoxic agents in murine subcutaneous tumor models correlates with clinical response. Clin Cancer Res 18:3846–3855. doi:10.1158/1078-0432.CCR-12-0738
Choo EF, Belvin M, Boggs J, et al. (2012) Preclinical disposition of gdc-0973 and prospective and retrospective analysis of human dose and efficacy predictions. Drug Metab Dispos 40:919–927. doi:10.1124/dmd.111.043778
Young H, Baum R, Cremerius U et al.; European Organization for Research and Treatment of cancer (EORTC) PET study group (1999) measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. Eur J Cancer 35:1773–82
Binns DS, Pirzkall A, Yu W et al.; OSI3926g Study Team (2011) Compliance with PET acquisition protocols for therapeutic monitoring of erlotinib therapy in an international trial for patients with non-small cell lung cancer. Eur J Nucl Med Mol Imaging 38:642. doi:10.1007/s00259-010-1665-0
Patel R, Tsan A, Tam R, et al. (2012) Mutation scanning using MUT-MAP, a high-throughput, microfluidic chip-based, multi-analyte panel. PLoS One 7:e51153. doi:10.1371/journal.pone.0051153
Therasse P, Arbuck SG, Eisenhauer EA et al.; European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada (2000) New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst 92:205–16
LoRusso PM, Adjei AA, Varterasian M, et al. (2005) Phase I and pharmacodynamic study of the oral MEK inhibitor CI-1040 in patients with advanced malignancies. J Clin Oncol 23:5281–5293
Martinez-Garcia M, Banerji U, Albanell J, et al. (2012) First-in-human, phase I dose-escalation study of the safety, pharmacokinetics, and pharmacodynamics of RO5126766, a first-in-class dual MEK/RAF inhibitor in patients with solid tumors. Clin Cancer Res 18:4806–4819
Infante JR, Fecher LA, Falchook GS, et al. (2012) Safety, pharmacokinetic, pharmacodynamic, and efficacy data for the oral MEK inhibitor trametinib: a phase 1 dose-escalation trial. Lancet Oncol 13:773–781. doi:10.1016/S1470-2045(12)70270-X
Honda K, Yamamoto N, Nokihara H, et al. (2013) Phase I and pharmacokinetic/pharmacodynamic study of RO5126766, a first-in-class dual Raf/MEK inhibitor, in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol 72:577–584. doi:10.1007/s00280-013-2228-4
Ribas A, Gonzalez R, Pavlick A, et al. (2014) Combination of vemurafenib and cobimetinib in patients with advanced BRAF(V600)-mutated melanoma: a phase 1b study. Lancet Oncol 15:954–965. doi:10.1016/S1470-2045(14)70301-8
Larkin J, Ascierto PA, Dréno B, et al. (2014) Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N Engl J Med 371:1867–1876. doi:10.1056/NEJMoa1408868
Flaherty KT, Robert C, Hersey P et al.; METRIC Study Group (2012) Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med 367:107–14. doi:10.1056/NEJMoa1203421
Catalanotti F, Solit DB, Pulitzer MP, et al. (2013) Phase II trial of MEK inhibitor selumetinib (AZD6244, ARRY-142886) in patients with BRAFV600E/K-mutated melanoma. Clin Cancer Res 19:2257–2264. doi:10.1158/1078-0432.CCR-12-3476
Weekes CD, Von Hoff DD, Adjei AA, et al. (2013) Multicenter phase I trial of the mitogen-activated protein kinase 1/2 inhibitor BAY 86–9766 in patients with advanced cancer. Clin Cancer Res 19:1232–1243. doi:10.1158/1078-0432.CCR-12-3529
Bennouna J, Lang I, Valladares-Ayerbes M, et al. (2011) A Phase II, open-label, randomised study to assess the efficacy and safety of the MEK1/2 inhibitor AZD6244 (ARRY-142886) versus capecitabine monotherapy in patients with colorectal cancer who have failed one or two prior chemotherapeutic regimens. Investig New Drugs 29:1021–1028. doi:10.1007/s10637-010-9392-8
Acknowledgments
The authors thank the patients and their families. This study was funded by Genentech, Inc. All authors participated in manuscript writing and approved the final version of the manuscript. We also acknowledge the contributions of Luna Musib, Steve Eppler, Alex de Crespigny, Jill Fredrickson, Mary Gates, and Yibing Yang. Editing and writing assistance was provided by Bryan Hains and Deborah Solymar (Genentech, Inc.) and was funded by Genentech, Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Lee S. Rosen, Patricia LoRusso, and Jonathan W. Goldman have received research funding from Genentech, Inc. Salim Yazji is a prior employee and current shareholder of Exelixis and is a current employee of Baxalta. Angela Shen is a prior employee of Exelixis and a current employee of Arvinas. Stuart Johnston is a shareholder and prior employee of Exelixis and a current employee of Nektar Therapeutics. Hsin-Ju Hsieh and Iris T. Chan are employees and shareholders of Roche. Branimir I. Sikic has received research funding from Exelixis, Genentech, Inc., Novartis, and Sanofi, and is a consultant for Novartis and Threshold Pharmaceuticals. Wen Wee Ma, Amy Weise, A. Dimitrios Colevas, and Alex Adjei declare that they have no potential conflicts of interest.
Research involving human participants
All procedures performed involving human participants in the protocol were approved by Institutional Review Boards prior to patient recruitment and conducted in accordance International Conference on Harmonization E6 Guidelines for Good Clinical Practice.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic Supplementary Material
ESM 1
(PDF 628 kb)
Rights and permissions
About this article
Cite this article
Rosen, L.S., LoRusso, P., Ma, W.W. et al. A first-in-human phase I study to evaluate the MEK1/2 inhibitor, cobimetinib, administered daily in patients with advanced solid tumors. Invest New Drugs 34, 604–613 (2016). https://doi.org/10.1007/s10637-016-0374-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-016-0374-3