Summary
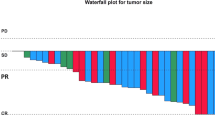
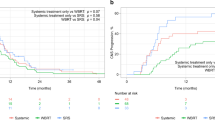
Background Central nervous system (CNS) metastases caused by small-cell lung cancer (SCLC) are incurable and therefore fatal. Although such metastases are usually treated with chemotherapy or radiotherapy, their sensitivity to these treatment measures is unclear. Amrubicin appears to be a promising agent for relapsed SCLC, but its effectiveness in CNS metastases originating from SCLC is unknown. Methods Between April 2002 and December 2009, 110 SCLC patients with CNS metastasis were treated at Shizuoka Cancer Center. Of these, we retrospectively reviewed 8 consecutive cases with CNS metastases originating from relapsed SCLC that were treated with amrubicin as a second-line therapy. Results We recorded three sensitive relapses and five refractory cases. Amrubicin yielded a CNS response rate of 50 % (2 partial responses and 2 complete response; 95 % CI, 21.5–78.5 %) and the disease control rate for CNS lesions was 87.5 % (95 % CI, 52.9–97.8 %). All of the sensitive relapse patients achieved a partial response. The median time to progression for CNS metastases was 150.5 days (95 % CI, 9–171 days), and the median survival time from the start of amrubicin administration was 230.5 days (95 % CI, 89–619 days). We also report a dramatic improvement in one patient’s radiological result of intramedullary spinal cord metastasis and alleviation of her symptoms following amrubicin monotherapy including this case series. Conclusions The results of this study suggest that amrubicin is active in patients with CNS metastases originating from SCLC.

Similar content being viewed by others
Abbreviations
- CNS:
-
Central nervous system
- SCLC:
-
Small-cell lung cancer
- CNS-TTP:
-
Time to progression of CNS metastases
- ISCM:
-
Intramedullary spinal cord metastasis
- CT:
-
Computed tomography
- MRI:
-
Magnetic resonance imaging
References
Patchell RA (1991) Brain metastases. Neurol Clin 9:817–824
Hirsch FR, Paulson OB, Hansen HH, Larsen SO (1983) Intracranial metastases in small cell carcinoma of the lung. Prognostic aspects. Cancer 51:529–533
Grossi F, Scolaro T, Tixi L, Loprevite M, Ardizzoni A (2001) The role of systemic chemotherapy in the treatment of brain metastases from small-cell lung cancer. Crit Rev Oncol Hematol 37:61–67
Onoda S, Masuda N, Seto T, Eguchi K, Takiguchi Y et al (2006) Phase II trial of amrubicin for treatment of refractory or relapsed small-cell lung cancer: Thoracic Oncology Research Group Study 0301. J Clin Oncol 24:5448–5453
Inoue A, Sugawara S, Yamazaki K, Maemondo M, Suzuki T et al (2008) Randomized phase II trial comparing amrubicin with topotecan in patients with previously treated small-cell lung cancer: North Japan Lung Cancer Study Group Trial 0402. J Clin Oncol 26:5401–5406
Murakami H, Yamamoto N, Shibata T, Takeda K, Ichinose Y et al (2014) A single-arm confirmatory study of amrubicin therapy in patients with refractory small-cell lung cancer: Japan Clinical Oncology Group Study (JCOG0901). Lung Cancer 84(1):67–72
O’Brien ME, Ciuleanu TE, Tsekov H, Shparyk Y, Cucevia B et al (2006) Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J Clin Oncol 24:5441–5447
Ettinger DS, Jotte R, Lorigan P, Gupta V, Garbo L et al (2010) Phase II study of amrubicin as second-line therapy in patients with platinum-refractory small-cell lung cancer. J Clin Oncol 28:2598–2603
Jotte R, Conkling P, Reynolds C, Galsky MD, Klein L et al (2010) Randomized phase II trial of single-agent amrubicin or topotecan as second-line treatment in patients with small-cell lung cancer sensitive to first-line platinum-based chemotherapy. J Clin Oncol 29(3):287–293
von Pawel J, Jotte R, Spigel DR, O’Brien ME, Socinski MA et al (2014) Randomized phase III trial of amrubicin versus topotecan as second-line treatment for patients with small-cell lung cancer. J Clin Oncol 32(35):4012–4019
Borgelt B, Gelber R, Kramer S, Brady LW, Chang CH et al (1980) The palliation of brain metastases: final results of the first two studies by the Radiation Therapy Oncology Group. Int J Radiat Oncol Biol Phys 6:1–9
Lagerwaard FJ, Levendag PC, Nowak PJ, Eijkenboom WM, Hanssens PE et al (1999) Identification of prognostic factors in patients with brain metastases: a review of 1292 patients. Int J Radiat Oncol Biol Phys 43:795–803
Saito EY, Viani GA, Ferrigno R, Nakamura RA, Novaes PE et al (2006) Whole brain radiation therapy in management of brain metastasis: results and prognostic factors. Radiat Oncol 1:20
Castrucci WA, Knisely JP (2008) An update on the treatment of CNS metastases in small cell lung cancer. Cancer J 14:138–146
Sung C, Blaney SM, Cole DE, Balis FM, Dedrick RL (1994) A pharmacokinetic model of topotecan clearance from plasma and cerebrospinal fluid. Cancer Res 54:5118–5122
Baker SD, Heideman RL, Crom WR, Kuttesch JF, Gajjar A et al (1996) Cerebrospinal fluid pharmacokinetics and penetration of continuous infusion topotecan in children with central nervous system tumors. Cancer Chemother Pharmacol 37:195–202
Ardizzoni A, Hansen H, Dombernowsky P, Gamucci T, Kaplan S et al (1997) Topotecan, a new active drug in the second-line treatment of small-cell lung cancer: a phase II study in patients with refractory and sensitive disease. The European Organization for Research and Treatment of Cancer Early Clinical Studies Group and New Drug Development Office, and the Lung Cancer Cooperative Group. J Clin Oncol 15:2090–2096
Korfel A, Oehm C, von Pawel J, Keppler U, Deppermann M et al (2002) Response to topotecan of symptomatic brain metastases of small-cell lung cancer also after whole-brain irradiation. a multicentre phase II study. Eur J Cancer 38:1724–1729
Mut M, Schiff D, Shaffrey ME (2005) Metastasis to nervous system: spinal epidural and intramedullary metastases. J Neurooncol 75:43–56
Wagner M, Besse B, Balleyguier C, Soria JC (2008) Leptomeningeal and medullary response to second-line erlotinib in lung adenocarcinoma. J Thorac Oncol 3:677–679
Acknowledgments
Sources of support: This work was supported in part by a Grant-in-Aid for Cancer Research from the Ministry of Health, Labor, and Welfare of Japan. We appreciate Ms. Yuka Matsui for her technical assistance of manuscript submission.
Competing interest
The authors declare that they have no competing interests.
Author’s contributions
SM and KK conceived of the study and contributed to writing and editing the report. RK, HA, AO, TS, AT and YN participated in data acquisition and interpretation. HK, TT and ME advised on clinical matters and the interpretation of the data and contributed to the writing of the report and helped to draft the manuscript. HM and TN helped to perform the statistical analysis. NY provided overall project management. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miura, S., Kaira, K., Kaira, R. et al. The efficacy of amrubicin on central nervous system metastases originating from small-cell lung cancer: a case series of eight patients. Invest New Drugs 33, 755–760 (2015). https://doi.org/10.1007/s10637-015-0233-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-015-0233-7