Summary
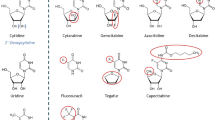
A novel cytidine analog fluorocyclopentenylcytosine (RX-3117; TV-1360) was characterized for its cytotoxicity in a 59-cell line panel and further characterized for cytotoxicity, metabolism and mechanism of action in 15 additional cancer cell lines, including gemcitabine-resistant variants. In both panels sensitivity varied 75-fold (IC50: 0.4- > 30 μM RX-3117). RX-3117 showed a different sensitivity profile compared to cyclopentenyl-cytosine (CPEC) and azacytidine, substrates for uridine-cytidine-kinase (UCK). Dipyridamole, an inhibitor of the equilibrative-nucleoside-transporter protected against RX-3117. Uridine and cytidine protected against RX-3117, but deoxycytidine (substrate for deoxycytidine-kinase [dCK]) not, although it protected against gemcitabine, demonstrating that RX-3117 is a substrate for UCK and not for dCK. UCK activity was abundant in all cell lines, including the gemcitabine-resistant variants. RX-3117 was a very poor substrate for cytidine deaminase (66,000-fold less than gemcitabine). RX-3117 was rapidly metabolised to its nucleotides predominantly the triphosphate, which was highest in the most sensitive cells (U937, A2780) and lowest in the least sensitive (CCRF-CEM). RX-3117 did not significantly affect cytidine and uridine nucleotide pools. Incorporation of RX-3117 into RNA and DNA was higher in sensitive A2780 and low in insensitive SW1573 cells. In sensitive U937 cells 1 μM RX-3117 resulted in 90 % inhibition of RNA synthesis but 100 μM RX-3117 was required in A2780 and CCRF-CEM cells. RX-3117 at IC50 values did not affect the integrity of RNA. DNA synthesis was completely inhibited in sensitive U937 cells at 1 μM, but in other cells even higher concentrations only resulted in a partial inhibition. At IC50 values RX-3117 downregulated the expression of DNA methyltransferase. In conclusion, RX-3117 showed a completely different sensitivity profile compared to gemcitabine and CPEC, its uptake is transporter dependent and is activated by UCK. RX-3117 is incorporated into RNA and DNA, did not affect RNA integrity, depleted DNA methyltransferase and inhibited RNA and DNA synthesis. Nucleotide formation is related with sensitivity.










Similar content being viewed by others
References
Hubeek I, Kaspers GJ, Ossenkoppele GJ, Peters GJ (2006) Cytosine arabinoside. Metabolism, mechanisms of resistance and clinical pharmacology. Cancer Drug Discov Dev: Deoxynucleoside Analogs Cancer Ther. 119–152. Ed.; Peters, GJ. Totowa, NJ, USA, Humana Press
Adema AD, Losekoot N, Smid K, Kathmann I, Myhren F, Sandvold ML, Peters GJ (2010) Induction of resistance to the lipophilic cytarabine prodrug elacytarabine (CP-4055) in CEM leukemic cells. Nucleosides Nucleotides Nucleic Acids 29:394–399
Andersson R, Aho U, Nilsson BI, Peters GJ, Pastor-Anglada M, Rasch W, Sandvold ML (2009) Gemcitabine chemoresistance in pancreatic cancer: molecular mechanisms and potential solutions. Scand J Gastroenterol 44:782–786
Mini E, Nobili S, Caciagli B, Landini I, Mazzei T (2006) Cellular pharmacology of gemcitabine. AnnOncol 17(Suppl 5):v7–v12
Kaminskas E, Farrell AT, Wang YC, Sridhara R, Pazdur R (2005) FDA drug approval summary: azacitidine (5-azacytidine, Vidaza) for injectable suspension. Oncologist 10:176–182
Santos FP, Kantarjian H, Garcia-Manero G, Issa JP, Ravandi F (2010) Decitabine in the treatment of myelodysplastic syndromes. Expert Rev Anticancer Ther 10:9–22
Plumb JA, Steele N, Finn PW, Brown R (2004) Epigenetic approaches to cancer therapy. Biochem Soc Trans 32:1095–1097
Yang X, Lay F, Han H, Jones PA (2010) Targeting DNA methylation for epigenetic therapy. Trends Pharmacol Sci 31:536–546
Grant S (1998) Ara-C: cellular and molecular pharmacology. Adv Cancer Res 72:197–233
Plunkett W, Iacoboni S, Keating MJ (1986) Cellular pharmacology and optimal therapeutic concentrations of 1-beta-D-arabinofuranosylcytosine 5′-triphosphate in leukemic blasts during treatment of refractory leukemia with high-dose 1-beta-D-arabinofuranosylcytosine. Scand J Haematol Suppl 44:51–59
Bergman AM, Pinedo HM, Peters GJ (2002) Determinants of resistance to 2′,2′-difluorodeoxycytidine (gemcitabine). Drug Resist Updat 5:19–33
Ruiz Van Haperen V, Veerman G, Vermorken JB, Peters GJ (1993) 2′,2′-Difluoro-deoxycytidine (gemcitabine) incorporation into RNA and DNA of tumour cell lines. Biochem Pharmacol 46:762–766
Huang P, Chubb S, Hertel LW, Grindey GB, Plunkett W (1991) Action of 2′,2′-difluorodeoxycytidine on DNA synthesis. Cancer Res 51:6110–6117
van Moorsel CJ, Smid K, Voorn DA, Bergman AM, Pinedo HM, Peters GJ (2003) Effect of gemcitabine and cis-platinum combinations on ribonucleotide and deoxyribonucleotide pools in ovarian cancer cell lines. Int J Oncol 22:201–207
Heinemann V, Xu YZ, Chubb S, Sen A, Hertel LW, Grindey GB, Plunkett W (1990) Inhibition of ribonucleotide reduction in CCRF-CEM cells by 2′,2′-difluorodeoxycytidine. Mol Pharmacol 38:567–572
Shewach DS, Reynolds KK, Hertel L (1992) Nucleotide specificity of human deoxycytidine kinase. Mol Pharmacol 42:518–524
Heinemann V, Xu YZ, Chubb S, Sen A, Hertel LW, Grindey GB, Plunkett W (1992) Cellular elimination of 2′,2′-difluorodeoxycytidine 5′-triphosphate: a mechanism of self-potentiation. Cancer Res 52:533–539
Heinemann V, Hertel LW, Grindey GB, Plunkett W (1988) Comparison of the cellular pharmacokinetics and toxicity of 2′,2′-difluorodeoxycytidine and 1-beta-D-arabinofuranosylcytosine. Cancer Res 48:4024–4031
Heinemann V, Schulz L, Issels RD, Plunkett W (1995) Gemcitabine: a modulator of intracellular nucleotide and deoxynucleotide metabolism. Semin Oncol 22:11–18
van Moorsel CJ, Bergman AM, Veerman G, Voorn DA, Ruiz van Haperen V, Kroep JR, Pinedo HM, Peters GJ (2000) Differential effects of gemcitabine on ribonucleotide pools of twenty-one solid tumour and leukaemia cell lines. Biochim Biophys Acta 1474:5–12
Momparler RL (2005) Pharmacology of 5-Aza-2′-deoxycytidine (decitabine). Semin Hematol 42:S9–S16
Bouffard DY, Momparler LF, Momparler RL (1994) Enhancement of the antileukemic activity of 5-aza-2′-deoxycytidine by cyclopentenyl cytosine in HL-60 leukemic cells. Anticancer Drugs 5:223–228
D'Incalci M, Covey JM, Zaharko DS, Kohn KW (1985) DNA alkali-labile sites induced by incorporation of 5-aza-2′-deoxycytidine into DNA of mouse leukemia L1210 cells. Cancer Res 45:3197–3202
Grant S, Bhalla K, Gleyzer M (1984) Effect of uridine on response of 5-azacytidine-resistant human leukemic cells to inhibitors of de novo pyrimidine synthesis. Cancer Res 44:5505–5510
Vesely J (1985) Mode of action and effects of 5-azacytidine and of its derivatives in eukaryotic cells. Pharmacol Ther 28:227–235
Lemaire M, Momparler LF, Raynal NJ, Bernstein ML, Momparler RL (2009) Inhibition of cytidine deaminase by zebularine enhances the antineoplastic action of 5-aza-2′-deoxycytidine. Cancer Chemother Pharmacol 63:411–416
Gourdeau H, Clarke ML, Ouellet F, Mowles D, Selner M, Richard A, Lee N, Mackey JR, Young JD, Jolivet J, Lafreniere RG, Cass CE (2001) Mechanisms of uptake and resistance to troxacitabine, a novel deoxycytidine nucleoside analogue, in human leukemic and solid tumor cell lines. Cancer Res 61:7217–7224
Parker WB, Shaddix SC, Rose LM, Waud WR, Shewach DS, Tiwari KN, Secrist JA III (2000) Metabolism of 4′-thio-beta-D-arabinofuranosylcytosine in CEM cells. Biochem Pharmacol 60:1925–1932
Azuma A, Huang P, Matsuda A, Plunkett W (2001) 2′-C-cyano-2′-deoxy-1-beta-D-arabino-pentofuranosylcytosine: a novel anticancer nucleoside analog that causes both DNA strand breaks and G(2) arrest. Mol Pharmacol 59:725–731
Schimmel KJ, Gelderblom H, Guchelaar HJ (2007) Cyclopentenyl cytosine (CPEC): an overview of its in vitro and in vivo activity. Curr Cancer Drug Targets 7:504–509
Kang GJ, Cooney DA, Moyer JD, Kelley JA, Kim HY, Marquez VE, Johns DG (1989) Cyclopentenylcytosine triphosphate. Formation and inhibition of CTP synthetase. J Biol Chem 264:713–718
Van Bree C, Van Kuilenburg AB (2008) Will cyclopentenyl cytosine (CPEC) ever have a future in the clinic? Leuk Res 32:201–202
Verschuur AC, van Gennip AH, Leen R, Voute PA, Van Kuilenburg AB (2000) Cyclopentenyl cytosine increases the phosphorylation and incorporation into DNA of arabinofuranosyl cytosine in a myeloid leukemic cell-line. Adv Exp Med Biol 486:311–317
Bierau J, van Gennip AH, Leen R, Meinsma R, Caron HN, Van Kuilenburg AB (2006) Cyclopentenyl cytosine-induced activation of deoxycytidine kinase increases gemcitabine anabolism and cytotoxicity in neuroblastoma. Cancer Chemother Pharmacol 57:105–113
Gharehbaghi K, Szekeres T, Yalowitz JA, Fritzer-Szekeres M, Pommier YG, Jayaram HN (2000) Sensitizing human colon carcinoma HT-29 cells to cisplatin by cyclopentenylcytosine, in vitro and in vivo. Life Sci 68:1–11
Choi WJ, Chung HJ, Chandra G, Alexander V, Zhao LX, Lee HW, Nayak A, Majik MS, Kim HO, Kim JH, Lee YB, Ahn CH, Lee SK, Jeong LS (2012) Fluorocyclopentenyl-cytosine with broad spectrum and potent antitumor activity. J Med Chem 55:4521–4525
van Bree C, Castro KN, Loves WJ, Franken NA, Peters GJ, Haveman J (2002) Sensitivity to ionizing radiation and chemotherapeutic agents in gemcitabine-resistant human tumor cell lines. Int J Radiat Oncol Biol Phys 54:237–244
van Triest B, Pinedo HM, van Hensbergen Y, Smid K, Telleman F, Schoenmakers PS, van der Wilt CL, van Laar JA, Noordhuis P, Jansen G, Peters GJ (1999) Thymidylate synthase level as the main predictive parameter for sensitivity to 5-fluorouracil, but not for folate-based thymidylate synthase inhibitors, in 13 nonselected colon cancer cell lines. Clin Cancer Res 5:643–654
Temmink OH, Bijnsdorp IV, Prins HJ, Losekoot N, Adema AD, Smid K, Honeywell RJ, Ylstra B, Eijk PP, Fukushima M, Peters GJ (2010) Trifluorothymidine resistance is associated with decreased thymidine kinase and equilibrative nucleoside transporter expression or increased secretory phospholipase A2. Mol Cancer Ther 9:1047–1057
Jansen G, Mauritz R, Drori S, Sprecher H, Kathmann I, Bunni M, Priest DG, Noordhuis P, Schornagel JH, Pinedo HM, Peters GJ, Assaraf YG (1998) A structurally altered human reduced folate carrier with increased folic acid transport mediates a novel mechanism of antifolate resistance. J Biol Chem 273:30189–30198
Bergman AM, Adema AD, Balzarini J, Bruheim S, Fichtner I, Noordhuis P, Fodstad O, Myhren F, Sandvold ML, Hendriks HR, Peters GJ (2011) Antiproliferative activity, mechanism of action and oral antitumor activity of CP-4126, a fatty acid derivative of gemcitabine, in in vitro and in vivo tumor models. Invest New Drugs 29:456–466
Ruiz van Haperen V, Veerman G, Eriksson S, Boven E, Stegmann AP, Hermsen M, Vermorken JB, Pinedo HM, Peters GJ (1994) Development and molecular characterization of a 2′,2′-difluorodeoxycytidine-resistant variant of the human ovarian carcinoma cell line A2780. Cancer Res 54:4138–4143
Keepers YP, Pizao PE, Peters GJ, van Ark-Otte J, Winograd B, Pinedo HM (1991) Comparison of the sulforhodamine B protein and tetrazolium (MTT) assays for in vitro chemosensitivity testing. Eur J Cancer 27:897–900
Ruiz van Haperen V, Veerman G, Braakhuis BJ, Vermorken JB, Boven E, Leyva A, Peters GJ (1993) Deoxycytidine kinase and deoxycytidine deaminase activities in human tumour xenografts. Eur J Cancer 29A:2132–2137
Peters GJ, Oosterhof A, Veerkamp JH (1983) Pyrimidine metabolism in peripheral and phytohemagglutinin-stimulated mammalian lymphocytes. Int J Biochem 15:51–55
Peters GJ, Laurensse E, Leyva A, Lankelma J, Pinedo HM (1986) Sensitivity of human, murine, and rat cells to 5-fluorouracil and 5′-deoxy-5-fluorouridine in relation to drug-metabolizing enzymes. Cancer Res 46:20–28
van Moorsel CJ, Pinedo HM, Veerman G, Bergman AM, Kuiper CM, Vermorken JB, van der Vijgh WJ, Peters GJ (1999) Mechanisms of synergism between cisplatin and gemcitabine in ovarian and non-small-cell lung cancer cell lines. Br J Cancer 80:981–990
de Wolf C, Jansen R, Yamaguchi H, de Haas M, van de Wetering K, Wijnholds J, Beijnen J, Borst P (2008) Contribution of the drug transporter ABCG2 (breast cancer resistance protein) to resistance against anticancer nucleosides. Mol Cancer Ther 7:3092–3102
Bergman AM, Pinedo HM, Jongsma AP, Brouwer M, Ruiz van Haperen V, Veerman G, Leyva A, Eriksson S, Peters GJ (1999) Decreased resistance to gemcitabine (2′,2′-difluorodeoxycitidine) of cytosine arabinoside-resistant myeloblastic murine and rat leukemia cell lines: role of altered activity and substrate specificity of deoxycytidine kinase. Biochem Pharmacol 57:397–406
Bergman AM, Giaccone G, van Moorsel CJ, Mauritz R, Noordhuis P, Pinedo HM, Peters GJ (2000) Cross-resistance in the 2′,2′-difluorodeoxycytidine (gemcitabine)-resistant human ovarian cancer cell line AG6000 to standard and investigational drugs. Eur J Cancer 36:1974–1983
Humeniuk R, Menon LG, Mishra PJ, Gorlick R, Sowers R, Rode W, Pizzorno G, Cheng YC, Kemeny N, Bertino JR, Banerjee D (2009) Decreased levels of UMP kinase as a mechanism of fluoropyrimidine resistance. Mol Cancer Ther 8:1037–1044
Eliopoulos N, Cournoyer D, Momparler RL (1998) Drug resistance to 5-aza-2′-deoxycytidine, 2′,2′-difluorodeoxycytidine, and cytosine arabinoside conferred by retroviral-mediated transfer of human cytidine deaminase cDNA into murine cells. Cancer Chemother Pharmacol 42:373–378
Eliopoulos N, Beausejour C, Momparler RL (1999) Chemoprotection against cytosine nucleoside analogs using the human cytidine deaminase gene. Prog Exp Tumor Res 36:124–142
Borst P, Balzarini J, Ono N, Reid G, de Vries H, Wielinga P, Wijnholds J, Zelcer N (2004) The potential impact of drug transporters on nucleoside-analog-based antiviral chemotherapy. Antiviral Res 62:1–7
Bergman AM, Munch-Petersen B, Jensen PB, Sehested M, Veerman G, Voorn DA, Smid K, Pinedo HM, Peters GJ (2001) Collateral sensitivity to gemcitabine (2′,2′-difluorodeoxycytidine) and cytosine arabinoside of daunorubicin- and VM-26-resistant variants of human small cell lung cancer cell lines. Biochem Pharmacol 61:1401–1408
Bergman AM, Pinedo HM, Talianidis I, Veerman G, Loves WJ, van der Wilt CL, Peters GJ (2003) Increased sensitivity to gemcitabine of P-glycoprotein and multidrug resistance-associated protein-overexpressing human cancer cell lines. Br J Cancer 88:1963–1970
Hasegawa T, Futagami M, Kim HS, Matsuda A, Wataya Y (2002) Analysis of single nucleotide polymorphisms in uridine/cytidine kinase gene encoding metabolic enzyme of 3'-ethynylcytidine. Nucleic Acids Res.Suppl 237–238
Wataya Y, Futagami M, Naito T, Uchikubo Y, Yokogawa T, Takenaka K, Kim HS, Matsuda A, Fukushima M, Kitade Y (2001) Anticancer molecular mechanism of 3′-ethynylcytidine (ECyd). Nucleic Acids Res.Suppl 233–234
Wataya Y, Naito T, Sato A, Hiramoto A, Kitade Y, Sasaki T, Matsuda A, Fukushima M, Kim HS (2009) Molecular mechanisms of apoptosis induced by 3′-ethynylcytidine. Nucleic Acids Symp.Ser.(Oxf) 291–292
Acknowledgments
This study was supported by Rexahn Pharmaceuticals. RX-3117 is licensed to TEVA Pharmaceuticals. Employees (the co-authors) of both companies participated in the design of the studies, discussed data and commented on the manuscript, and approved final submission.
Conflict of interest
OO, AO, EB are employees of TEVA Pharmaceuticals, YBL, C-HA, DJK are employees of Rexahn, Pharmaceuticals. GJP got a grant from Rexahn Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peters, G.J., Smid, K., Vecchi, L. et al. Metabolism, mechanism of action and sensitivity profile of fluorocyclopentenylcytosine (RX-3117; TV-1360). Invest New Drugs 31, 1444–1457 (2013). https://doi.org/10.1007/s10637-013-0025-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-013-0025-x