Summary
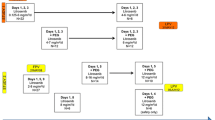
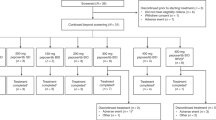
Purpose To determine the maximum tolerated dose and safety of the epothilone, KOS-862, in patients with advanced solid tumors or lymphoma. Patients and Methods Patients were treated weekly for 3 out of 4 weeks (Schedule A) or 2 out of 3 weeks (Schedule B) with KOS-862 (16–120 mg/m2). Pharmacokinetic (PK) sampling was performed during cycles 1 and 2; pharmacodynamic (PD) assessment for microtubule bundle formation (MTBF) was performed after the 1st dose, only at or above 100 mg/m2. Results Thirty-two patients were enrolled, and twenty-nine completed ≥1 cycle of therapy. Dose limiting toxicity [DLT] was observed at 120 mg/m2. PK data were linear from 16 to 100 mg/m2, with proportional increases in mean Cmax and AUCtot as a function of dose. Full PK analysis (mean ± SD) at 100 mg/m2 revealed the following: half-life (t ½) = 9.1 ± 2.2 h; volume of distribution (Vz) = 119 ± 41 L/m2; clearance (CL) = 9.3 ± 3.2 L/h/m2. MTBF (n = 9) was seen in 40% of PBMCs within 1 h and in 15% of PBMC at 24-hours post infusion at 100 mg/m2. Tumor shrinkage (n = 2, lymphoma), stable disease >3 months (n = 5, renal, prostate, oropharynx, cholangiocarcinoma, and Hodgkin lymphoma), and tumor marker reductions (n = 1, colorectal cancer/CEA) were observed. Conclusion KOS-862 was well tolerated with manageable toxicity, favorable PK profile, and the suggestion of clinical activity. The maximum tolerated dose was determined to be 100 mg/m2 weekly 3-on/1-off. MTBF can be demonstrated in PBMCs of patients exposed to KOS-862.




Similar content being viewed by others
References
Schiff PB, Fant J, Horwitz SB (1979) Promotion of microtubule assembly in vitro by taxol. Nature 277(5698):665–667
Yvon AM, Wadsworth P, Jordan MA (1999) Taxol suppresses dynamics of individual microtubules in living human tumor cells. Mol Biol Cell 10(4):947–959
Altmann KH, Wartmann M, O'Reilly T (2000) Epothilones and related structures–a new class of microtubule inhibitors with potent in vivo antitumor activity. Biochim Biophys Acta 1470(3):M79–M91
Horwitz SB et al (1993) Taxol: mechanisms of action and resistance. J Natl Cancer Inst Monogr 15:55–61
Jachez B, Nordmann R, Loor F (1993) Restoration of taxol sensitivity of multidrug-resistant cells by the cyclosporine SDZ PSC 833 and the cyclopeptolide SDZ 280–446. J Natl Cancer Inst 85(6):478–483
Nogales E et al (1995) Structure of tubulin at 6.5 A and location of the taxol-binding site. Nature 375(6530):424–427
Rowinsky EK et al (1993) Clinical toxicities encountered with paclitaxel (Taxol). Semin Oncol 20(4 Suppl 3):1–15
Rose WC (1992) Taxol: a review of its preclinical in vivo antitumor activity. Anticancer Drugs 3(4):311–321
Kowalski RJ, Giannakakou P, Hamel E (1997) Activities of the microtubule-stabilizing agents epothilones A and B with purified tubulin and in cells resistant to paclitaxel (Taxol(R)). J Biol Chem 272(4):2534–2541
Nettles JH et al (2004) The binding mode of epothilone A on alpha, beta-tubulin by electron crystallography. Science 305(5685):866–869
Gerth K et al (1996) Epothilons A and B: antifungal and cytotoxic compounds from Sorangium cellulosum (Myxobacteria). Production, physico-chemical and biological properties. J Antibiot (Tokyo) 49(6):560–563
Lee SH et al (2007) Epothilones induce human colon cancer SW620 cell apoptosis via the tubulin polymerization independent activation of the nuclear factor-kappaB/IkappaB kinase signal pathway. Mol Cancer Ther 6(10):2786–2797
Cheng KL, Bradley T, Budman DR (2008) Novel microtubule-targeting agents—the epothilones. Biologics 2(4):789–811
Trivedi M et al (2008) Epothilones: a novel class of microtubule-stabilizing drugs for the treatment of cancer. Future Oncol 4(4):483–500
Harrison M, Swanton C (2008) Epothilones and new analogues of the microtubule modulators in taxane-resistant disease. Expert Opin Investig Drugs 17(4):523–546
Goodin S, Kane MP, Rubin EH (2004) Epothilones: mechanism of action and biologic activity. J Clin Oncol 22(10):2015–2025
Nicolaou KC et al (1997) Synthesis of epothilones A and B in solid and solution phase. Nature 387(6630):268–272
Chou TC et al (1998) Desoxyepothilone B is curative against human tumor xenografts that are refractory to paclitaxel. Proc Natl Acad Sci U S A 95(26):15798–15802
Skehan P et al (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82(13):1107–1112
Chou TC et al (2001) The synthesis, discovery, and development of a highly promising class of microtubule stabilization agents: curative effects of desoxyepothilones B and F against human tumor xenografts in nude mice. Proc Natl Acad Sci U S A 98(14):8113–8118
Chou TC et al (1998) Desoxyepothilone B: an efficacious microtubule-targeted antitumor agent with a promising in vivo profile relative to epothilone B. Proc Natl Acad Sci U S A 95(16):9642–9647
Holen K (2004) Phase I study using continuous intravenous (CI) KOS-862 (Epothilone D) in patients with solid tumors. Proc Am Soc Clin Oncol, abstract 2024
Piro L (2003) KOS-862 (epothilone D): a comparison of two schedules in patients with advanced malignancies. Proc Am Soc Clin Oncol 22(abstract 539)
Arnold D et al (2009) Weekly administration of sagopilone (ZK-EPO), a fully synthetic epothilone, in patients with refractory solid tumours: results of a phase I trial. Br J Cancer 101(8):1241–1247
Lam ET et al (2011) Phase I dose escalation study of KOS-1584, a novel epothilone, in patients with advanced solid tumors. Cancer Chemother Pharmacol
Perez EA et al (2007) Efficacy and safety of ixabepilone (BMS-247550) in a phase II study of patients with advanced breast cancer resistant to an anthracycline, a taxane, and capecitabine. J Clin Oncol 25(23):3407–3414
Roche H et al (2007) Phase II clinical trial of ixabepilone (BMS-247550), an epothilone B analog, as first-line therapy in patients with metastatic breast cancer previously treated with anthracycline chemotherapy. J Clin Oncol 25(23):3415–3420
Chen T, Molina A, Moore S et al (2004) Epothilone B analog (BMS-247550) at the recommended phase II dose (RPTD) in patients (pts) with gynecologic (gyn) and breast cancers. Proc Am Soc Clin Oncol 22(2115a).
Smit W, Sufliarsky J, Spanik S et al (2005) Phase I/II dose-escalation trial of patupilone every 3 weeks in patients with relapsed/refractory ovarain cancer. . Proc Am Soc Clin Oncol 23(5056a).
Vansteenkiste J et al (2007) Phase II clinical trial of the epothilone B analog, ixabepilone, in patients with non small-cell lung cancer whose tumors have failed first-line platinum-based chemotherapy. J Clin Oncol 25(23):3448–3455
Burtness B, Goldwasser M, Axelrod R et al (2006) A randomized phase II study of BMS-247550 (ixabepilone) given daily × 5 days every 3 weeks or weekly in patients with metastatic or recurrent squamous cell cancer of the head and neck. . Proc Am Soc Clin Oncol 24(5532a)
Altmann KH (2005) Recent developments in the chemical biology of epothilones. Curr Pharm Des 11(13):1595–1613
Beer TM et al (2007) Phase II study of KOS-862 in patients with metastatic androgen independent prostate cancer previously treated with docetaxel. Invest New Drugs 25(6):565–570
Feldman DR et al (2007) Phase II trial of ixabepilone in patients with cisplatin-refractory germ cell tumors. Invest New Drugs 25(5):487–490
Whitehead RP et al (2006) A Phase II trial of epothilone B analogue BMS-247550 (NSC #710428) ixabepilone, in patients with advanced pancreas cancer: a Southwest Oncology Group study. Invest New Drugs 24(6):515–520
Ajani JA et al (2006) A multi-center phase II study of BMS-247550 (Ixabepilone) by two schedules in patients with metastatic gastric adenocarcinoma previously treated with a taxane. Invest New Drugs 24(5):441–446
Eng C et al (2004) A phase II trial of the epothilone B analog, BMS-247550, in patients with previously treated advanced colorectal cancer. Ann Oncol 15(6):928–932
Posadas EM et al (2007) A phase II study of ixabepilone (BMS-247550) in metastatic renal-cell carcinoma. Cancer Biol Ther 6(4):490–493
Smith S, Pro B, Van Besien K et al (2005) A phase II study of epothilone B analog BMS-247550 (NSC 710428) in patients with relapsed aggresive non-Hodgkin's lymphomas. Proc Am Soc Clin Oncol 23(6625a)
Galsky MD et al (2005) Multi-institutional randomized phase II trial of the epothilone B analog ixabepilone (BMS-247550) with or without estramustine phosphate in patients with progressive castrate metastatic prostate cancer. J Clin Oncol 23(7):1439–1446
Hensley ML et al (2007) A phase I trial of BMS-247550 (NSC# 710428) and gemcitabine in patients with advanced solid tumors. Invest New Drugs 25(4):335–341
Forster M et al (2007) A phase Ib and pharmacokinetic trial of patupilone combined with carboplatin in patients with advanced cancer. Clin Cancer Res 13(14):4178–4184
Plummer R (2002) Phase I and pharmakokinetic study of BMS-247-550 in combination with carboplatin in patients with advanced solid malignancies. . Proc Am Soc Clin Oncol, abstract 2125
Faivre S (2004) Phase I study of ixabepilone given every other week in combination with irinotecan in patients with advanced malignancies. Proc Am Soc Clin Oncol, abstract 2051
Wojtowicz M (2004) Phase I dose-escalation trial investigating the safety and tolerability of EPO906 plus estramustine in patients with advanced cancer Proc Am Soc Clin Oncol, abstract 4623
Conflict of Interest Statement
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported in part by funds granted by *The American Society of Clinical Oncology Career Development Awards (JD), and by The Damon Runyon-Lilly Clinical Investigator Award (JD). OAO was supported in part by a Leukemia and Lymphoma Society Research Scholar Award.
Rights and permissions
About this article
Cite this article
Konner, J., Grisham, R.N., Park, J. et al. Phase I clinical, pharmacokinetic, and pharmacodynamic study of KOS-862 (Epothilone D) in patients with advanced solid tumors and lymphoma. Invest New Drugs 30, 2294–2302 (2012). https://doi.org/10.1007/s10637-011-9765-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-011-9765-7