Summary
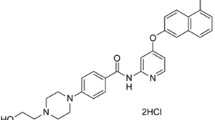
LY2457546 is a potent and orally bioavailable inhibitor of multiple receptor tyrosine kinases involved in angiogenic and tumorigenic signalling. In biochemical and cellular assays, LY2457546 demonstrates potent activity against targets that include VEGFR2 (KDR), PDGFRβ, FLT-3, Tie-2 and members of the Eph family of receptors. With activities against both Tie2 and Eph receptors, LY2457546 possesses an activity profile that distinguishes it from multikinase inhibitors. When compared head to head with sunitinib, LY2457546 was more potent for inhibition of endothelial tube formation in an in vitro angiogenesis co-culture model with an intermittent treatment design. In vivo, LY2457546 inhibited VEGF-driven autophosphorylation of lung KDR in the mouse and rat in a dose and concentration dependent manner. LY2457546 was well tolerated and exhibited efficacy in a 13762 syngeneic rat mammary tumor model in both once and twice daily continuous dosing schedules and in mouse human tumor xenograft models of lung, colon, and prostate origin. Additionally, LY2457546 caused complete regression of well-established tumors in an acute myelogenous leukemia (AML) FLT3-ITD mutant xenograft tumor model. The observed efficacy that was displayed by LY2457546 in the AML FLT3-ITD mutant tumor model was superior to sunitinib when both were evaluated using equivalent doses normalized to in vivo inhibition of pKDR in mouse lung. LY2457546 was well tolerated in non-clinical toxicology studies conducted in rats and dogs. The majority of the toxicities observed were similar to those observed with other multi-targeted anti-angiogenic kinase inhibitors (MAKs) and included bone marrow hypocellularity, hair and skin depigmentation, cartilage dysplasia and lymphoid organ degeneration and necrosis. Thus, the unique spectrum of target activity, potent in vivo anti-tumor efficacy in a variety of rodent and human solid tumor models, exquisite potency against a clinically relevant model of AML, and non-clinical safety profile justify the advancement of LY2457546 into clinical testing.












Similar content being viewed by others
References
Hicklin DJ, Ellis LM (2005) Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 23:1011–1027
Takahashi H, Shibuya M (2005) The vascular endothelial growth factor (VEGF)/VEGF receptor system and its role under physiological and pathological conditions. Clin Sci 109:227–241
Mendel DB, Laird AD, Xin X, Louie SG, Christensen JG, Li G, Schreck RE, Abrams TJ, Ngai TJ, Lee LB, Murray LJ, Carver J, Chan E, Moss KG, Haznedar JO, Sukbuntherng J, Blake RA, Sun L, Tang C, Miller T, Shirazian S, McMahon G, Cherrington JM (2003) In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmacodynamic relationship. Clin Cancer Res 9:327–337
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, Cao Y, Shujath J, Gawlak S, Eveleigh D, Rowley B, Liu L, Adnane L, Lynch M, Auclair D, Taylor I, Gedrich R, Voznesensky A, Riedl B, Post LE, Bollag G, Trail PA (2004) BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 64:7099–7109
Casanovas O, Hicklin DJ, Bergers G, Hanahan D (2005) Drug resistance by evasion of antiangiogenic targeting of VEGF signalling in late-stage pancreatic islet tumors. Cancer Cell 8:299–309
Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand SJ, Holash J (2000) Vascular-specific growth factors and blood vessel formation. Nature 407:242–248
Barda DA, Burkholder TP, Clayton JR, Hao Y, Henry JR, Knobeloch JM, McLean JA, Mendel D, Rempala ME, Wang ZQ, Yip YY, Zhong B (2010) U.S. Patent 7 666 879
Okram B, Nagle A, Adrian FJ, Lee C, Ren P, Wang X, Sim T, Xie Y, Wang X, Xia G, Spraggon G, Warmuth M, Liu Y, Gray N (2006) A general strategy for creating “inactive-conformation” Abl inhibitors. Chem Biol 13:779–786
Schindler T, Bornmann W, Pellicena P, Miller WT, Clarkson B, Kuriyan J (2000) Sructural mechanism for STI-571 inhibition of abelson tyrosine kinase. Science 289:1938–1942
Manley PW, Bold G, Bruggen J, Fendrich G, Furet P, Mestan J, Schnell C, Stolz B, Meyer T, Meyhack B, Stark W, Strauss A, Wood J (2004) Advances in the structural biology, design and clinical development of VEGF-R kinase inhibitors for the treatment of angiogenesis. Biochim Biophys Acta 1697:17–27
Sammond DM, Nailor KE, Veal JM, Nolte RT, Wang L, Knick VB, Rudolph SK, Truesdale AT, Nartey EN, Stafford JA, Kumar R, Cheung M (2005) Discovery of a novel and potent series of dianilinopyrimidineurea and urea isostere inhibitors of VEGFR2 tyrosine kinase. Bioorg Med Chem Lett 15:3519–3523
Jung FH, Pasquet G, Lambert-van der Brempt C, Lohman JM, Warin N, Renaud F, Germain H, De Savi C, Roberts N, Johnson T, Dousson C, Hill GB, Mortlock AA, Heron N, Wilkinson RW, Wedge SR, Heaton SP, Odedra R, Keen NJ, Green S, Brown E, Thompson K, Brightwell S (2006) Discovery of novel and potent thiazoloquinazolines as selective Aurora A and B kinase inhibitors. J Med Chem 49:955–970
Huss KL, Blonigen PE, Campbell RM (2007) Development of a TranscreenerTM kinase assay for protein kinase A and demonstration of concordance of data with a filter-binding assay format. J Biomol Screen 12:578–584
Wang X, Mader MM, Toth JE, Yu X, Jin N, Campbell RM, Smallwood JE, Christe ME, Chatterjee A, Goodson T Jr, Vlahos CJ, Matter WF, Bloem LJ (2005) Complete inhibition of anisomycin and UV radiation but not cytokine induced JNK and p38 activation by an aryl-substituted dihydropyrrolopyrazole quinoline and mixed lineage kinase 7 small interfering RNA. J Biol Chem 280:19298–19305
Hasumi Y, Babaa M, Ajimab R, Hasumi H, Valeraa VA, Kleina ME, Hainesc DC, Merinod MJ, Honga S-B, Yamaguchib TP, Schmidt LS, Linehana WM (2009) Homozygous loss of BHD causes early embryonic lethality and kidney tumor development with activation of mTORC1 and mTORC2 PNAS, 106: 18722–18727
Chen Y, Wei T, Yan L, Lawrence L, Qian H-R, Burkholder TP, Starling JJ, Yingling JM, Shou J (2008) Developing and applying a gene functional association network for anti-angiogenic kinase inhibitor activity assessment in an angiogenesis co-culture model. BMC Genomics 9:264
Abu-Ehuler FM, Goodeve AC, Wilson GA, Gari MA, Peake IR, Rees DC, Vandenberghe EA, Winship PR, Reilly JT (2000) FLT3 internal tandem duplication mutations in adult acute myeloid leukemia define a high-risk group. Br J Haematol 111:190–195
Rombouts WJ, Blokland I, Lowenberg B, Ploemacher RE (2000) (2000) Biological characteristics and prognosis of adult acute myeloid leukemia with internal tandem duplications in the Flt3 gene. Leukemia 14(4):675–683
Meshinchi S, Woods WG, Stirewalt DL, Sweetser DA, Buckley JD, Tjoa TK, Bernstein ID, Radich JP (2001) Prevalence and prognostic significance of Flt3 internal tandem duplication in pediatric acute myeloid leukemia. Blood 97:89–94
Obermann EC, Arber C, Jotterand M, Tichelli A, Hirschmann P, Tzankov A (2010) Expression of pSTAT5 predicts FLT3 internal tandem duplications in acute myeloid leukemia. Ann Hematol 89:663–669
Hall AP, Westwood FR, Wadsworth PF (2006) Review of the effects of anti-angiogenic compounds on the epiphyseal growth plate. Toxicol Pathol 34(2):131–4
Sutent (Sunitinib Malate) (2006) Pharmacology review, NDA Application No.: 021938 & 021968, Pfizer Inc
Nexavar (Sorafenib Tosylate): Pharmacology Review, NDA Application No.: 021923, 2005. Bayer Pharmaceuticals Corporation
Avastin (Bevacizumab): Pharmacology Review, BLA Application No.: 125085, 2004. Genetech, Inc
Moss KG, Toner GC, Cherrington JM, Mendel DB, Laird AD (2003) Hair depigmentation is a biological readout for pharmacological inhibition of KIT in mice and humans. J Pharmacol Exp Ther 307(2):476–80
Robert C, Soria JC, Spatz A, Le Cesne A, Malka D, Pautier P, Wechsler J, Lhomme C, Escudier B, Boige V, Armand JP, Le Chevalier T (2005) Cutaneous side-effects of kinase inhibitors and blocking antibodies. Lancet Oncol 6(7):491–500
Robert C, Spatz A, Faivre S, Armand JP, Raymond E (2003) Tyrosine kinase inhibition and grey hair. Lancet 361(9362):1056
Acknowledgements
The authors would like to thank Steve Hatch, Baohui Zhao, Julie A. Stewart, Jason R. Manro, Andrew R. Capen, Lysiane Huber, Phil Schwier, Lisa Kays, Spring Weir, James E. McGee, Jeffrey K. Smallwood, Robert B. Peery, Jianyong Shou, Joan H. Carter, Karen S. Britt, Bryan Dirk Anderson, Robert M. Campbell, and Aimee K. Bence for helpful discussions and for conducting experiments and data analyses in support of these studies.
Conflict of interest
All Lilly employees are employed by and hold stocks in Eli Lilly and Company, which owns LY2457546.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Funding
The study was sponsored by Eli Lilly and Company.
Rights and permissions
About this article
Cite this article
Burkholder, T.P., Clayton, J.R., Rempala, M.E. et al. Discovery of LY2457546: a multi-targeted anti-angiogenic kinase inhibitor with a novel spectrum of activity and exquisite potency in the acute myelogenous leukemia-Flt-3-internal tandem duplication mutant human tumor xenograft model. Invest New Drugs 30, 936–949 (2012). https://doi.org/10.1007/s10637-011-9640-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-011-9640-6