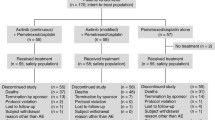
Abstract
Purpose Studies indicate that recombinant human endostatin (rh-endostatin) can inhibit tumor endothelial cell proliferation, angiogenesis, and tumor growth. This study assessed the efficacy of the combination of standard gemcitabine plus cisplatin chemotherapy with rh-endostatin in patients with non-small-cell lung cancer (NSCLC). Patients and Methods Chemotherapy-naive patients with stage IIIB to IV NSCLC were randomly (1:1) assigned to receive gemcitabine/cisplatin chemotherapy alone or with 7.5 mg/ m2 of intravenously rh-endostatin on days 1 to 14 of each 3-week cycle. The primary end point was objective response rate (ORR). Results Baseline characteristics were similar between treatment arms. The best ORRs for rh-endostatin arm (n = 33) and chemotherapy-alone arm were 37.5% (95% CI: 21.3 to 47.2%) and 28.6% (95% CI: 19.8 to 37.6%), respectively. Median survival was 12.4 months in the rh-endostatin arm and 9.8 months in the chemotherapy-alone arm, and 1-year survival was 51.6% and 38.7%, respectively. Mild palpitions, diarrhea, and liver dysfunction were the most common rh-endostatin-related adverse events. Grade 3/4 hematological toxicities were all reported similar for patients in the two arms. Conclusion The addition of rh-endostatin to gemcitabine plus cisplatin chemotherapy for first-line treatment of NSCLC improves objective response and may improve survival.

Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J et al (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Ardizzoni A, Boni L, Tiseo M et al (2007) Cisplatin- versus carboplatin-based chemotherapy in first-line treatment of advanced non-small-cell lung cancer: an individual patient data meta-analysis. J Natl Cancer Inst 99:847–857
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285:1182–1186
Italiano JE Jr, Richardson JL, Patel-Hett S et al (2008) Angiogenesis is regulated by a novel mechanism: pro- and antiangiogenic proteins are organized into separate platelet granules and differentially released. Blood 111:1227–1233
Tosetti F, Ferrari N, De Flora S et al (2002) ‘Angioprevention’: angiogenesis is a common and key target for cancer chemopreventive agents. FASEB J 16:2–14
Boehle AS, Kurdow R, Schulze M et al (2001) Human endostatin inhibits growth of human non-small-cell lung cancer in a murine xenotransplant model. Int J Cancer 94:420–428
Iizasa T, Chang H, Suzuki M, et al (2005) Expression of collagen XVIII in non-small cell lung carcinoma and its prognostic value. AACR Meeting Abstracts, 944
Ni Q, Ji H, Zhao Z et al (2009) Endostar, a modified endostatin inhibits non small cell lung cancer cell in vitro invasion through osteopontin-related mechanism. Eur J Pharmacol 614:1–6
Sun Y, Wang J, Liu Y, et al (2005) Results of phase III trial of rh-endostatin (YH-16) in advanced non-small cell lung cancer (NSCLC) patients. ASCO Meeting Abstracts :7138
Han B, Xiu Q, Wang H, et al (2008) Rh-endostatin injection plus paclitaxel and carboplatin therapy for non-small-cell lung cancer: randomized, double-blind, placebo-controlled, multicentre study. ASCO Meeting Abstracts :19126
Le Chevalier T, Scagliotti G, Natale R et al (2005) Efficacy of gemcitabine plus platinum chemotherapy compared with other platinum containing regimens in advanced non-small-cell lung cancer: a meta-analysis of survival outcomes. Lung Cancer 47:69–80
Arrieta O, González-De la Rosa CH, Aréchaga-Ocampo E et al (2010) Randomized phase II trial of all-trans-retinoic acid with chemotherapy based on paclitaxel and cisplatin as first-line treatment in patients with advanced non-small-cell lung cancer. J Clin Oncol 28:3463–3471
Schiller JH, Harrington D, Belani CP et al (2002) Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346:92–98
Sandler A, Gray R, Perry MC et al (2006) Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355:2542–2550
Reck M, von Pawel J, Zatloukal P et al (2009) Phase III trial of cisplatin plus gemcitabine with either placebo or bevacizumab as first-line therapy for nonsquamous non–small-cell lung cancer: AVAiL. J Clin Oncol 27:1227–1234
Lynch TJ, Patel T, Dreisbach L et al (2010) Cetuximab and first-line taxane/carboplatin chemotherapy in advanced non-small-cell lung cancer: results of the randomized multicenter phase III trial BMS099. J Clin Oncol 28:911–917
Pirker R, Pereira JR, Szczesna A et al (2009) Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an open-label randomized phase III trial. Lancet 373:1525–1531
Acknowledgements
The authors wish to thank all the patients who chose to participate in this trial.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, X., Mei, K., Cai, X. et al. A randomized phase II study of recombinant human endostatin plus gemcitabine/cisplatin compared with gemcitabine/cisplatin alone as first-line therapy in advanced non-small-cell lung cancer. Invest New Drugs 30, 1144–1149 (2012). https://doi.org/10.1007/s10637-011-9631-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-011-9631-7