Summary
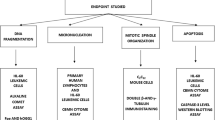
DNA intercalating molecules are promising chemotherapeutic agents. In the present study, a novel DNA intercalating compound of pyrimido[4′,5′:4,5]selenolo(2,3-b)quinoline series having 8-methyl-4-(3 diethylaminopropylamino) side chain is studied for its chemotherapeutic properties. Our results showed that 8-methyl-4-(3 diethylaminopropylamino) pyrimido [4′,5′:4,5] selenolo(2,3-b)quinoline (MDPSQ) induces cytotoxicity in a time- and concentration-dependent manner on leukemic cell lines. Both cell cycle analysis and tritiated thymidine assays revealed that MDPSQ affects DNA replication. Treatment with MDPSQ resulted in both elevated levels of DNA strand breaks and repair proteins, further indicating its cytotoxic effects. Besides, Annexin V/PI staining revealed that MDPSQ induces cell death by triggering necrosis rather than apoptosis.






Similar content being viewed by others
References
Rowley JD (2001) Chromosome translocations: dangerous liaisons revisited. Nat Rev Cancer 1:245–250
Lieber MR (1993) In: Kirsch I (ed) The causes and consequences of chromosomal translocations. CRC, Boca Raton, pp 239–275
Raghavan SC, Lieber MR (2006) DNA structures at chromosomal translocation sites. Bioessays 28(5):480–494
Raghavan SC et al (2004) A non-B-DNA structure at the Bcl-2 major breakpoint region is cleaved by the RAG complex. Nature 428(6978):88–93
Nambiar M, Kari V, Raghavan SC (2008) Chromosomal translocations in cancer. Biochim Biophys Acta 1786(2):139–152
Takeuchi Y et al (1997) Synthesis and antitumor activity of fused quinoline derivative. IV. Novel 11-aminoindolo(2, 3-b)quinolines. Chem Pharm Bull (Tokyo) 45:406–411
Auclair C et al (1987) Physicochemical and pharmacological properties of the antitumor ellipticine derivative 2-(diethylamino-2-ethyl)9-hydroxy ellipticinium-chloride. HCl. Cancer Res 47(23):6254–6261
Tilak Raj T, Ambekar SY (1988) Synthesis of 4-amino pyrimido [4′, 5′:4, 5]thieno (2, 3-b) quinolines. J Chem Eng Data 33:530–531
Tilak Raj T, Ambekar SY (1988) Synthesis of 4-amino pyrimido [4′, 5′:4, 5]thieno (2, 3-b) quinoline-4(3H)-ones. J Chem Res (S) 50:537–551
Dervan PB, Edelson BS (2003) Recognition of the DNA minor groove by pyrrole-imidazole polyamides. Curr Opin Struct Biol 13(3):284–299
Rezler EM, Bearss DJ, Hurley LH (2003) Telomere inhibition and telomere disruption as processes for drug targeting. Annu Rev Pharmacol Toxicol 43:359–379
Shahabuddin MS, Gopal M, Raghavan SC (2009) Intercalating, cytotoxic, antitumour activity of 8-chloro and 4-morpholinopyrimido [4′, 5′:4, 5]thieno(2, 3-b)quinolines. J Photochem Photobiol B 94(1):13–19
Shahabuddin MS et al (2009) A novel DNA intercalator, butylamino-pyrimido[4′,5′:4,5]selenolo(2,3-b)quinoline, induces cell cycle arrest and apoptosis in leukemic cells. Invest New Drugs
Shahabuddin MS, Gopal M, Raghavan SC (2007) Intercalating and antitumor activity of 4-Oxopyrimido [4′, 5′:4, 5]thieno(2, 3-b)quinoline-4 (3H)-one. J Cancer Mol 3:139–146
Gopal M, Shahabuddin MS (2004) Biological properties of 8-methoxypyrimido[4(1), 5(1):4, 5]thieno(2, 3-b)quinoline-4(3H)-one, a new class of DNA intercalating drugs. Indian J Med Res 119(5):198–205
Gopal M, Veeranna S (2005) 4-Anilinopyrimido[4′, 5′:4, 5]selenolo(2, 3-b)quinoline and 4-piperazino pyrimido[4′, 5′:4, 5]selenolo(2, 3-b)quinoline: new DNA intercalating chromophores with antiproliferative activity. J Photochem Photobiol B 81(3):181–189
Shenoy S et al (2007) 8-Methyl-4-(3-diethylaminopropylamino) pyrimido [4′, 5′;4, 5] thieno (2, 3-b) quinoline (MDPTQ), a quinoline derivate that causes ROS-mediated apoptosis in leukemia cell lines. Toxicol Appl Pharmacol 222(1):80–88
Gopal M, Shenoy S, Doddamani LS (2003) Antitumour activity of 4-amino and 8-methly-4-(3-diethyl aminopropylamino)pyrimido[4′, 5′:4, 5]thieno(2, 3-b)quinoline. J Photochem Photobiol B 72:69–78
Nandeeshaiah SK, Ambekar SY (1998) Synthesis, Dimroth rearrangment and blood platelet disaggregation property of pyrimido[4′, 5′:4, 5]selenolo(2, 3-b)quinolines: a new class of condensed quinoline. Indian J Chem 37:995–1000
Nandeeshaiah SK (1994) In: Department of Chemistry, University of Mysore, Mysore
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63
Chiruvella KK et al (2008) Methyl angolensate, a natural tetranortriterpenoid induces intrinsic apoptotic pathway in leukemic cells. FEBS Lett 582(29):4066–4076
Kavitha CV et al (2009) Novel derivatives of spirohydantoin induce growth inhibition followed by apoptosis in leukemia cells. Biochem Pharmacol 77(3):348–363
Vermes I et al (1995) A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J Immunol Methods 184(1):39–51
Mukhopadhyay P et al (2007) Simultaneous detection of apoptosis and mitochondrial superoxide production in live cells by flow cytometry and confocal microscopy. Nat Protoc 2(9):2295–2301
Kaufmann SH et al (1993) Specific proteolytic cleavage of poly(ADP-ribose) polymerase: an early marker of chemotherapy-induced apoptosis. Cancer Res 53(17):3976–3985
Ihmels H et al (2005) Intercalation of organic dye molecules into double-stranded DNA. Part 2: the annelated quinolizinium ion as a structural motif in DNA intercalators. Photochem Photobiol 81(5):1107–1115
Martinez R, Chacon-Garcia L (2005) The search of DNA-Intercalators as antitumoral drugs: what it worked and what did not work. Curr Med Chem 12(2):127–151
Dervan PB et al (2005) Regulation of gene expression by synthetic DNA-binding ligands. Top Curr Chem 253:1–32
Fosse P et al (1992) Stimulation of topoisomerase II-mediated DNA cleavage by ellipticine derivatives: structure-activity relationship. Mol Pharmacol 42(4):590–595
Juret P et al (1978) Preliminary trial of 9-hydroxy-2-methyl ellipticinium (NSC 264–137) in advanced human cancers. Eur J Cancer 14(2):205–206
Mathe G et al (1970) Methoxy-9-ellipticine lactate. 3. Clinical screening: its action in acute myeloblastic leukaemia. Rev Eur Etud Clin Biol 15(5):541–545
Brana MF et al (2001) Intercalators as anticancer drugs. Curr Pharm Des 7(17):1745–1780
Frei E et al (2002) Covalent binding of the anticancer drug ellipticine to DNA in V79 cells transfected with human cytochrome P450 enzymes. Biochem Pharmacol 64(2):289–295
Stiborova M et al (2001) The anticancer agent ellipticine on activation by cytochrome P450 forms covalent DNA adducts. Biochem Pharmacol 62(12):1675–1684
Ashkenazi A, Dixit VM (1998) Death receptors: signaling and modulation. Science 281(5381):1305–1308
Fantin VR, Leder P (2004) F16, a mitochondriotoxic compound, triggers apoptosis or necrosis depending on the genetic background of the target carcinoma cell. Cancer Res 64(1):329–336
Chiruvella KK et al (2007) Mechanism of DNA Double-Strand Break Repair. ICFAI J Biotech 1:7–22
Cepero V et al (2007) Trans-platinum(II) complexes with cyclohexylamine as expectator ligand induce necrosis in tumour cells by inhibiting DNA synthesis and RNA transcription. Clin Transl Oncol 9(8):521–530
Dartsch DC et al (2002) Comparison of anthracycline-induced death of human leukemia cells: programmed cell death versus necrosis. Apoptosis 7(6):537–548
Kiaris H, Schally AV (1999) Apoptosis versus necrosis: which should be the aim of cancer therapy? Proc Soc Exp Biol Med 221(2):87–88
Reiter I, Krammer B, Schwamberger G (1999) Cutting edge: differential effect of apoptotic versus necrotic tumor cells on macrophage antitumor activities. J Immunol 163(4):1730–1732
Proskuryakov SY, Konoplyannikov AG, Gabai VL (2003) Necrosis: a specific form of programmed cell death? Exp Cell Res 283(1):1–16
Dewey WC, Ling CC, Meyn RE (1995) Radiation-induced apoptosis: relevance to radiotherapy. Int J Radiat Oncol Biol Phys 33(4):781–796
Acknowledgements
We thank Dr. Kavitha C.V., Ms. Nishana M. and members of SCR laboratory for critical reading of the manuscript. This work was supported by Lady Tata Memorial Trust international award for leukemia research (London) for SCR. SMS is supported by DBT postdoctoral fellowship from India. MN is supported by Senior Research Fellowship from CSIR, India.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. S. Shahabuddin and Mridula Nambiar contributed equally to the work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Suppl. Figure 1
Quantification of cell cycle analysis of K562 and CEM cells following treatment with MDPSQ. K562 (A,B) and CEM (D,E) cells were treated with MDPSQ as described in Fig. 3. Quantification of different cell cycle phases is shown. C. Western blot showing expression of PCNA. The α-Tubulin was used as an internal loading control (PPT 283 kb)
Rights and permissions
About this article
Cite this article
Shahabuddin, M.S., Nambiar, M., Moorthy, B.T. et al. A novel structural derivative of natural alkaloid ellipticine, MDPSQ, induces necrosis in leukemic cells. Invest New Drugs 29, 523–533 (2011). https://doi.org/10.1007/s10637-009-9379-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-009-9379-5