Summary
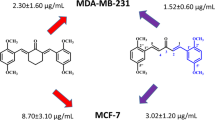
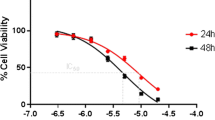
Estrogen receptor (ER)-negative breast cancer is an aggressive form that currently requires more drug treatment options. Thus, we have further modified cyclohexanone derivatives of curcumin and examined them for cytotoxicity towards ER-negative human breast cancer cells. Two of the analogs screened elicited increased cytotoxic potency compared to curcumin and other previously studied derivatives. Specifically, 2,6-bis(pyridin-3-ylmethylene)-cyclohexanone (RL90) and 2,6-bis(pyridin-4-ylmethylene)-cyclohexanone (RL91) elicited EC50 values of 1.54 and 1.10 µM, respectively, in MDA-MB-231 cells and EC50 values of 0.51 and 0.23 in SKBr3 cells. All other new compounds examined were less potent than curcumin, which elicited EC50 values of 7.6 and 2.4 µM in MDA-MB-231 and SKBr3 cells, respectively. Mechanistic analyses demonstrated that RL90 and RL91 significantly induced G2/M-phase cell cycle arrest and apoptosis. RL90 and RL91 also modulated the expression of key cell signaling proteins, specifically, in SKBr3 cells, protein levels of Her-2, Akt, and NFκB were decreased in a time-dependent manner, while activity of stress kinases JNK1/2 and P38 MAPK were increased. Signaling events in MDA-MB-231 cells were differently implicated, as EGFR protein levels were decreased and activity of GSK-3β transiently decreased, while β-catenin protein level and activity of P38 MAPK, Akt, and JNK1/2 were transiently increased. In conclusion replacement of the phenyl group of cyclohexanone derived curcumin derivatives with heterocyclic rings forms a class of second-generation analogs that are more potent than both curcumin and other derivatives. These new derivatives provide a platform for the further development of drugs for the treatment of ER-negative breast cancer.








Similar content being viewed by others
References
Parkin D, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Putti T, El-Rehim D, Rakha E, Paish C, Lee A, Pinder S, Ellis I (2005) Estrogen receptor-negative breast carcinomas: a review of morphology and immunophenotypical analysis. Mod Pathol 18:26–35
Azuine M, Bhide S (1992) Chemopreventive effect of turmeric against stomach and skin tumors induced by chemical carcinogens in swiss mice. Nutr Cancer 17:77–83
Huang M, Lou Y, Ma W, Newmark H, Reuhl K, Conney A (1994) Inhibitory effects of dietary curcumin on forestomach, duodenal, and colon carcinogenesis in mice. Cancer Res 54:5841–5847
Ikezaki S, Nishikawa A, Furukawa F, Kudo K, Nakamura H, Tamura K, Mori H (2001) Chemopreventive effects of curcumin on glandular stomach carcinogenesis induced by n-methyl-n′-nitro-n-nitrosoguanidine and sodium chloride in rats. Anticancer Res 21:3407–3411
Rao C, Simi B, Reddy B (1993) Inhibition by dietary curcumin of azoxymethane-induced ornithine decarboxylase, tyrosine protein kinase, arachidonic acid metabolism and aberrant crypt foci formation in the rat colon. Carcinogenesis 11:2219–2225
Rao C, Rivenson A, Simi B, Reddy B (1995) Chemoprevention of colon cancer by dietary curcumin. Ann NY Acad Sci USA 768:201–204
Sharma RA, Iresone CR, Verschoyle RD, Hill KA, Williams ML, Leuratti C, Manson MM, Marnett LJ, Steward WP, Gescher A (2001) Effects of dietary curcumin on glutathione s-transferase and malondialdehyde-DNA adducts in rat liver and colon mucosa: relationship with drug levels. Clin Cancer Res 7:1452–1458
Inano H, Onoda M (2002) Prevention of radiation-induced mammary tumors. Int J Radiat Oncol Biol Phys 52:212–223
Inano H, Onoda M, Inafuku N, Kubota M, Kamada Y, Osawa T, Kobayashi H, Wakabayashi K (1999) Chemoprevention by curcumin during the promotion stage of tumorigenesis of mammary gland in rats irradiated with gamma-rays. Carcinogenesis 20:1011–1018
Pereira MA, Grubbs CJ, Barnes LH, Li H, Olson GR, Eto I, Juliana M, Whitaker LM, Kelloff GJ, Steele VE, Lubet RA (1996) Effects of the phytochemicals, curcumin and quercetin, upon azoxymethane-induced colon cancer and 7, 12-dimethylbenz[a]anthracene-induced mammary cancer in rats. Carcinogenesis 17:1305–1311
Singletary K, MacDonald C, Wallig M, Fisher C (1996) Inhibition of 7, 12-dimethylbenz[a]anthracene (dmba)-induced mammary tumorigenesis and dmba-DNA adduct formation by curcumin. Cancer Lett 103:137–141
Chuang S, Cheng A, Lin J, Kuo M (2000) Inhibition by curcumin of diethylnitrosamine-induced hepatic hyperplasia, inflammation, cellular gene products and cell-cycle-related proteins in rats. Food ChemToxicol 38:991–995
Squires MS, Hudson EA, Howells L, Sale S, Houghton CE, Jones JL, Fox LH, Dickens M, Prigent SA, Manson MM (2003) Relevance of mitogen activated protein kinase (mapk) and phosphotidylinositol-3-kinase/protein kinase b (pi3k/pkb) pathways to induction of apoptosis by curcumin in breast cells. Biochem Pharmacol 65:361–376
Shao ZM, Shen ZZ, Liu CH, Sartippour MR, Go VL, Heber D, Nguyen M (2002) Curcumin exerts multiple suppressive effects on human breast carcinoma cells. Int J Cancer 98:234–240
Mehta K, Pantazis P, Mcqueen T, Aggarwal B (1997) Antiproliferative effect of curcumin (diferuloylmethane) against human breast tumor cell lines. Anticancer Drugs 8:470–481
Verma SP, Salamone E, Goldin B (1997) Curcumin and genistein, plant natural products, show synergistic inhibitory effects on the growth of human breast cancer mcf-7 cells induced by estrogenic pesticides. Biochem Biophys Res Commun 233:692–696
Ramachandran C, You W (1999) Differential sensitivity of human mammary epithelial and breast carcinoma cell lines to curcumin. Breast Cancer Res Treat 54:269–278
Hanif R, Qiao L, Shiff S, Rigas B (1997) Curcumin, a natural plant phenolic food additive, inhibits cell proliferation and induces cell cycle changes in colon adenocarcinoma cell lines by a prostaglandin-independent pathway. J Lab Clin Med 130:576–584
Mukhopadhyay A, Bueso-Ramos C, Chatterjee D, Pantazis P, Aggarwal BB (2001) Curcumin downregulates cell survival mechanisms in human prostate cancer cell lines. Oncogene 20:7597–7609
Jee S, Shen S, Tseng C, Chiu H, Kuo M (1998) Curcumin induces a p53-dependent apoptosis in human basal cell carcinoma cells. J Invest Dermatol 111:656–661
Somers-Edgar TJ, Scandlyn MJ, Stuart EC, Le Nedelec MJ, Valentine SP, Rosengren RJ (2008) The combination of epigallocatechin gallate and curcumin suppresses era- breast cancer cell growth in vitro and in vivo. Inter J Cancer 122:1966–1971
Cheng AL, Hsu CH, Lin J-K, Hsu MM, Ho YF, Shen TS, Ko JY, Lin JT, Lin BR, Ming-Shiang W, Yu HS, Jee SH, Chen GS, Chen TM, Chen CA, Lai MK, Pu YS, Pan MH, Wang YJ, Tsai CC, Hsieh CY (2001) Phase i clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer Res 21:2895–2900
Liang G, Shao L, Wang Y, Zhao C, Chu Y, Xiao J, Zhao Y, Li X, Yang S (2009) Exploration and synthesis of curcumin analogues with improved structural stability both in vitro and in vivo as cytotoxic agents. Bioorg Med Chem 17:2623–2631
Markaverich B, Schauweker T, Gregory R, Varma M, Kittrell F, Medina D, Varma R (1992) Nuclear type ii sites and malignant cell proliferation: inhibition by 2, 6-bis-benzylidenecyclohexanones. Cancer Res 52:2482–2488
Adams BK, Ferstl EM, Davis MC, Herold M, Kurtkaya S, Camalier RF, Hooingshead MG, Kaur G, Sausville EA, Rickles FR, Snyder JP, Liotta DC, Shoji M (2004) Synthesis and biological evaluation of novel curcumin analogs as anti-cancer and anti-angiogenesis agents. Bioorg Med Chem 12:3871–3883
Adams BK, Cai J, Armstrong J, Herold M, Lu YJ, Sun A, Snyder JP, Liotta DC, Jones JL, Shoji M (2005) Ef24, a novel synthetic curcumin analog, induces apoptosis in cancer cells via a redox-dependent mechanism. Anticancer Drugs 16:263–275
Das B, Thirupathi P, Mahender I, Reddy KR (2006) Convenient and facile cross-aldol condensation catalyzed by molecular iodine. An efficient synthesis of α, α ′-bis(substituted-benzylidene) cycloalkanones. J Mol Cat 247:182–185
Costi R, Di Santo R, Artico M, Massa S, Ragno R, Loddo R, La Colla M, Tramontano E, La Colla P, Pani A (2004) 2, 6-bis(3, 4, 5-trihydroxybenzylydene) derivatives of cyclohexanone: novel potent hiv-1 integrase inhibitors that prevent hiv-1 multiplication in cell-based assays. Bioorg Med Chem 12:199–215
Bhagat S, Sharma R, Chakraborti AK (2006) Dual-activation protocol for tandem cross-aldol condensation: an easy and highly efficient synthesis of α, α ′-bis(aryl/alkylmethylidene)ketones. J Mol Cat 260:235–240
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JT, Mokesch H, Kenney S, Boyd MR (1990) New colorimetric cytotoxicity assay for anti-cancer drug screening. J Natl Cancer Inst 82:1107–1112
Stuart EC, Rosengren RJ (2008) The combination of raloxifene and epigallocatechin gallate suppresses growth and induces apoptosis in mda-mb-231 cells. Life Sciences 82:943–948
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85
Wosikowski K, Schuurhuis D, Kops G, Saceda M, Bates S (1997) Altered gene expression in drug-resistant human breast cancer cells. Clin Cancer Res 3:2405–2414
Robinson T, Ehlers T, Hubbard I, Bai X, Arbiser J, Goldsmith D, Bowen J (2003) Design, synthesis, and biological evaluation of angiogenesis inhibitors: aromatic enone and dienone analogues of curcumin. Bioorg Med Chem Lett 13:115–117
Grandis J, Sok J (2004) Signaling through the epidermal growth factor receptor during the development of malignancy. Pharmacol Ther 102:37–46
DeFazio A, Chiew Y, McEvoy M, Watts C, Sutherland R (1997) Antisense estrogen receptor rna expression increases epidermal growth factor receptor gene expression in breast cancer cells. Cell Growth Differ 8:903–911
Roos W, Fabbro D, Kung W, Costa SD, Eppenberger U (1986) Correlation between hormone dependency and the regulation of epidermal growth factor receptor by tumour promoters in human mammary carcinoma cells. Proc Natl Acad Sci USA 83:991–995
Slamon D, Clark G, Wong S, Levin W, Ullrich A, Mcguire W (1987) Human breast cancer: correlation of relapse and survival with amplification of the her-2/neu oncogene. Science 235:177–182
Biswas DK, Shi Q, Baily S, Strickland I, Ghosh S, Perdee AB, Iglehart JD (2004) Nf-kb activation in human breast cancer specimens and its role in cell proliferation and apoptosis. Proc Natl Acad Sci USA 101:10137–10142
Korutla L, Cheung J, Mendelsohn J, Kumar R (1995) Inhibition of ligand-induced activation of epidermal growth factor receptor tyrosine phosphorylation by curcumin. Carcinogenesis 16:1741–1745
Korutla L, Kumar R (1994) Inhibitory effect of curcumin on epidermal growth factor receptor kinase activity in a431 cells. Biochim Biophys Acta 1224:597–600
Hong R, Spohn W, Hung M (1999) Curcumin inhibits tyrosine kinase activity of p185neu and also depletes p185neu. Clin Cancer Res 5:1884–1891
Dorai T, Cao YC, Dorai B, Buttyan R, Katz AE (2001) Therapeutic potential of curcumin in human prostate cancer. Iii. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of lncap prostate cancer cells in vivo. Prostate 47:293–303
Patel B, Sengupta R, Qazi S, Vachhani H, Yu Y, Rishi A, Majumdar A (2008) Curcumin enhances the effects of 5-fluorouracil and oxaliplatin in mediating growth inhibition of colon cancer cells by modulating egfr and igf-1r. Int J Cancer 122:267–273
Chen A, Xu J, Johnson A (2006) Curcumin inhibits human colon cancer cell growth by suppressing gene expression of epidermal growth factor receptor through reducing the activity of the transcription factor egr-1. Oncogene 25:278–287
Longva KE, Pedersen NM, Haslekas C, Stang E, Madshus IH (2005) Herceptin-induced inhibition of erbb2 signaling involves reduced phosphorylation of akt but not endocytic down-regulation of erbb2. Int J Cancer 116:359–367
Dillon RL, White DE, Muller WJ (2007) The phosphatidyl inositol 3-kinase signaling network: implications for human breast cancer. Oncogene 26:1338–1345
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-kinase akt pathway in human cancer. Nat Rev Cancer 2:489–501
Ichijo H, Nishida E, Irie K, ten Dijke P, Saitoh M, Moriguchi T, Takagi M, Matsumoto K, Miyazono K, Gotoh Y (1997) Induction of apoptosis by ask1, a mammalian mapkkk that activates sapk/jnk and p38 signaling pathways. Science 275:90–94
Nishitoh H, Saitoh M, Mochida Y, Takeda K, Nakano H, Rothe M, Miyazono K, Ichijo H (1998) Ask1 is essential for jnk/sapk activation by traf2. Mol Cell 2:389–395
Ho KK, Rosivatz E, Gunn RM, Smith ME, Stavropoulou AV, Rosivatz E, Numbere MG, Wong JB, Lafitte VG, Behrendt JM, Myatt SS, Hailes HC, Woscholski R, Lam EW (2009) The novel molecule 2-[5-(2-chloroethyl)-2-acetoxy-benzyl]-4-(2-chloroethyl)-phenyl acetate inhibits phosphoinositide 3-kinase/akt/mammalian target of rapamycin signalling through jnk activation in cancer cells. Febs J
Yamamoto Y, Hoshino Y, Ito T, Nariai T, Mohri T, Obana M, Hayata N, Uozumi Y, Maeda M, Fujio Y, Azuma J (2008) Atrogin-1 ubiquitin ligase is upregulated by doxorubicin via p38-map kinase in cardiac myocytes. Cardiovasc Res 79:89–96
Barkett M, Gilmore TD (1999) Control of apoptosis by rel/nf-kappab transcription factors. Oncogene 18:6910–6924
Lauder A, Castellanos A, Weston K (2001) C-myb transcription is activated by protein kinase b (pkb) following interleukin 2 stimulation of t cells and is required for pkb-mediated protection from apoptosis. Mol Cell Biol 21:5797–5805
Burow ME, Weldon CB, Melnik LI, Duong BN, Collins-Burow BM, Beckman BS, McLachlan JA (2000) Pi3-k/akt regulation of nf-kappab signaling events in suppression of tnf-induced apoptosis. Biochem Biophys Res Commun 271:342–345
Gong L, Li Y, Nedeljkovic-Kurepa A, Sarkar FH (2003) Inactivation of nf-kappab by genistein is mediated via akt signaling pathway in breast cancer cells. Oncogene 22:4702–4709
Anand P, Thomas S, Kunnumakkara A, Sundarama C, Harikumar K, Sung B, Tharakan S, Misra K, Priyadarsini I, Rajasekharan K, Aggarwal BB (2008) Biological activities of curcumin and its analogues (congeners) made by man and mother nature. Biochem Pharmacol 76:1590–1611
Acknowledgments
This work was supported by a grant from the Breast Cancer Research Trust (RJR) and a University of Otago Postgraduate Scholarship (TJS-E).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Somers-Edgar, T.J., Taurin, S., Larsen, L. et al. Mechanisms for the activity of heterocyclic cyclohexanone curcumin derivatives in estrogen receptor negative human breast cancer cell lines. Invest New Drugs 29, 87–97 (2011). https://doi.org/10.1007/s10637-009-9339-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-009-9339-0