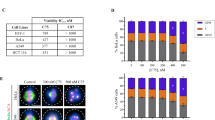
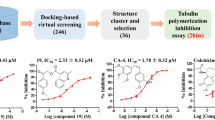
Summary
Targeting cellular mitosis is an attractive antitumor strategy. Here, we reported MT7, a novel compound from the 6H-Pyrido[2′,1′:2,3]imidazo [4,5-c]isoquinolin- 5(6H)-one library generated by using the multi-component reaction strategy, as a new mitotic inhibitor. MT7 elicited apparent inhibition of cell proliferation by arresting mitosis specifically and reversibly in various tumor cell lines originating from different human tissues. Detailed mechanistic studies revealed that MT7 induced typical gene expression profiles related to mitotic arrest shown by cDNA microarray assays. Connectivity Map was used to analyze the microarray data and suggested that MT7 was possibly a tubulin inhibitor due to its similar gene expression profiles to those of the known tubulin inhibitors demecolcine, celastrol and paclitaxel. Further analyses demonstrated that MT7 inhibited the polymerization of cellular microtubules although it was not detectable to bind to purified tubulin. The inhibition of cellular tubulin polymerization by MT7 subsequently resulted in the disruption of mitotic spindle formation, activated the spindle assembly checkpoint and consequently arrested the cells at mitosis. The persistent mitotic arrest by the treatment with MT7 led the tested tumor cells to apoptosis. Our data indicate that MT7 could act as a promising lead for further optimization, in hopes of developing new anticancer therapeutics and being used to probe the biology of mitosis, specifically, the mode of interference with microtubules.






Similar content being viewed by others
Abbreviations
- MT7:
-
6-(4-methoxybenzyl)pyrido[2′,1′:2,3] imidazo[4,5-c]isoquinolin-5(6H)-one
- VCR:
-
vincristine
- DMSO:
-
dimethyl sulfoxide
- SRB:
-
sulforhodamine B
- siRNA:
-
small interfering RNA
- GSEA:
-
gene set enrichment analysis
- SAC:
-
spindle assembly checkpoint
- IC50 :
-
50% inhibitory concentration
- Topo I:
-
topoisomerase I
- Topo II:
-
topoisomerase II.
References
Sherr CJ (1996) Cancer Cell Cycles. Science 274:1672–1677
Castedo M, Perfettini J-L, Roumier T et al (2004) Cell death by mitotic catastrophe: a molecular definition. Oncogene 23:2825–2837
Wood KW (2001) Past and future of the mitotic spindle as an oncology target. Curr. Opin. Pharmacol. 1:370–377
Jackson JR, Patrick DR, Dar MM et al (2007) Targeted anti-mitotic therapies: can we improve on tubulin agents? Nat Rev Cancer 7:107–117
Jordan MA, Wilson L (2004) Microtubules as a target for anticancer drugs. Nat Rev Cancer 4:253–265
Pinguet F, Mavel S, Galtier C et al (1999) Synthesis and cytotoxicity of novel pyrido[1, 2-e]purines on multidrug resistant human MCF7 cells. Pharmazie 54:876–878
Adhikary PF, Das SK, Hess BA Jr (1976) Synthesis and antihypertensive activity of some imidazoindole derivatives. J Med Chem 19:1352–1354
Meng T, Zhang Z, Hu D et al (2007) Three-component combinatorial synthesis of a substituted 6H-pyrido[2′, 1′:2, 3]imidazo- [4, 5-c]isoquinolin-5(6H)-one library with cytotoxic activity. J Comb Chem 9:739–741
Ditchfield C, Johnson VL, Tighe A et al (2003) Aurora B couples chromosome alignment with anaphase by targeting BubR1, Mad2, and Cenp-E to kinetochores. J. Cell Biol. 161:267–280
Andreassen PR, Skoufias DA, Margolis RL (2004) Analysis of the spindle-assembly checkpoint in HeLa cells. Methods Mol Biol 281:213–225
Shelanski ML, Gaskin F, Cantor CR (1973) Microtubule assembly in the absence of added nucleotides. Proc Natl Acad Sci U S A 70:765–768
Subramanian A, Tamayo P, Mootha VK et al (2005) Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 102:15545–15550
Mootha VK, Lindgren CM, Eriksson K-F et al (2003) PGC-1[alpha]-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat Genet 34:267–273
Lamb J, Crawford ED, Peck D et al (2006) The Connectivity Map: Using Gene-Expression Signatures to Connect Small Molecules, Genes, and Disease. Science 313:1929–1935
Lamb J (2007) The Connectivity Map: a new tool for biomedical research. Nat Rev Cancer 7:54–60
Yamashita Y, Fujii N, Murakata C et al (1992) Induction of mammalian DNA topoisomerase I mediated DNA cleavage by antitumor indolocarbazole derivatives. Biochemistry 31:12069–12075
Meng LH, Zhang JS, Ding J (2001) Salvicine, a novel DNA topoisomerase II inhibitor, exerting its effects by trapping enzyme-DNA cleavage complexes. Biochem Pharmacol 62:733–741
Qin Y, Meng L, Hu C et al (2007) Gambogic acid inhibits the catalytic activity of human topoisomerase IIalpha by binding to its ATPase domain. Mol Cancer Ther 6:2429–2440
Tanabe K, Ikegami Y, Ishida R et al (1991) Inhibition of topoisomerase II by antitumor agents bis(2, 6-dioxopiperazine) derivatives. Cancer Res 51:4903–4908
Bruce Alberts AJ, Lewis J, Raff M, Roberts K, Walter P (2002) Molecular Biology of the Cell. Garland Science, New York
Pagano M, Pepperkok R, Verde F et al (1992) Cyclin A is required at two points in the human cell cycle. EMBO J 11:961–971
Clute P, Pines J (1999) Temporal and spatial control of cyclin B1 destruction in metaphase. Nat Cell Biol 1:82–87
di Bernardo D, Thompson MJ, Gardner TS et al (2005) Chemogenomic profiling on a genome-wide scale using reverse-engineered gene networks. Nat Biotechnol 23:377–383
Cho RJ, Campbell MJ, Winzeler EA et al (1998) A genome-wide transcriptional analysis of the mitotic cell cycle. Mol Cell 2:65–73
Whitfield ML, Sherlock G, Saldanha AJ et al (2002) Identification of genes periodically expressed in the human cell cycle and their expression in tumors. Mol Biol Cell 13:1977–2000
Jiang Y, Liu M, Spencer CA et al (2004) Involvement of transcription termination factor 2 in mitotic repression of transcription elongation. Mol Cell 14:375–385
Parsons GG, Spencer CA (1997) Mitotic repression of RNA polymerase II transcription is accompanied by release of transcription elongation complexes. Mol Cell Biol 17:5791–5802
Gottesfeld JM, Forbes DJ (1997) Mitotic repression of the transcriptional machinery. Trends Biochem Sci 22:197–202
Hartl P, Gottesfeld J, Forbes DJ (1993) Mitotic repression of transcription in vitro. J Cell Biol 120:613–624
Spencer CA, Kruhlak MJ, Jenkins HL et al (2000) Mitotic transcription repression in vivo in the absence of nucleosomal chromatin condensation. J Cell Biol 150:13–26
Vivanco I, Palaskas N, Tran C et al (2007) Identification of the JNK signaling pathway as a functional target of the tumor suppressor PTEN. Cancer Cell 11:555–569
Baur JA, Pearson KJ, Price NL et al (2006) Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444:337–342
Cabral F, Sobel ME, Gottesman MM (1980) CHO mutants resistant to colchicine, colcemid or griseofulvin have an altered beta-tubulin. Cell 20:29–36
Banerjee AC, Bhattacharyya B (1979) Colcemid and colchicine binding to tubulin. Similarity and dissimilarity. FEBS Lett 99:333–336
Morita H, Hirasawa Y, Muto A et al (2008) Antimitotic quinoid triterpenes from Maytenus chuchuhuasca. Bioorg Med Chem Lett 18:1050–1052
Peters NT, Logan KO, Miller AC et al (2007) Phospholipase D signaling regulates microtubule organization in the fucoid alga Silvetia compressa. Plant Cell Physiol 48:1764–1774
Dhonukshe P, Laxalt AM, Goedhart J et al (2003) Phospholipase D Activation Correlates with Microtubule Reorganization in Living Plant Cells. Plant Cell 15:2666–2679
Kadura S, Sazer S (2005) SAC-ing mitotic errors: how the spindle assembly checkpoint (SAC) plays defense against chromosome mis-segregation. Cell Motil Cytoskeleton 61:145–160
Yamada HY, Gorbsky GJ (2006) Spindle checkpoint function and cellular sensitivity to antimitotic drugs. Mol Cancer Ther 5:2963–2969
Rieder CL, Maiato H (2004) Stuck in division or passing through: what happens when cells cannot satisfy the spindle assembly checkpoint. Dev Cell 7:637–651
Steegmaier M (2005) BI 2536, a potent and highly selective inhibitor of Polo-like kinase 1 (Plk1), induces mitotic arrest and apoptosis in a broad spectrum of tumor cell lines. Clin. Cancer Res. 11:9147
Mayer TU (1999) Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science 286:971–974
Tao W (2005) Induction of apoptosis by an inhibitor of the mitotic kinesin KSP requires both activation of the spindle assembly checkpoint and mitotic slippage. Cancer Cell 8:49–59
Keen N, Taylor S (2004) Aurora-kinase inhibitors as anticancer agents. Nature Rev. Cancer 4:927–936
Soncini C, Carpinelli P, Gianellini L et al (2006) PHA-680632, a novel Aurora kinase inhibitor with potent antitumoral activity. Clin Cancer Res 12:4080–4089
Donaldson MM, Tavares AA, Hagan IM et al (2001) The mitotic roles of Polo-like kinase. J Cell Sci 114:2357–2358
Marumoto T, Zhang D, Saya H (2005) Aurora-A — a guardian of poles. Nat Rev Cancer 5:42–50
Huang M, Gao H, Chen Y et al (2007) Chimmitecan, a novel 9-substituted camptothecin, with improved anticancer pharmacologic profiles in vitro and in vivo. Clin Cancer Res 13:1298–1307
Bhat KM, Setaluri V (2007) Microtubule-associated proteins as targets in cancer chemotherapy. Clin Cancer Res 13:2849–2854
Charbaut E, Curmi PA, Ozon S et al (2001) Stathmin Family Proteins Display Specific Molecular and Tubulin Binding Properties. J. Biol. Chem. 276:16146–16154
Wignall SM, Gray NS, Chang YT et al (2004) Identification of a novel protein regulating microtubule stability through a chemical approach. Chem Biol 11:135–146
Fourest-Lieuvin A, Peris L, Gache V et al (2006) Microtubule Regulation in Mitosis: Tubulin Phosphorylation by the Cyclin-dependent Kinase Cdk1. Mol Biol Cell 17:1041–1050
Matsuyama A, Shimazu T, Sumida Y et al (2002) In vivo destabilization of dynamic microtubules by HDAC6-mediated deacetylation. EMBO J 21:6820–6831
Zhang Y, Li N, Caron C et al (2003) HDAC-6 interacts with and deacetylates tubulin and microtubules in vivo. EMBO J 22:1168–1179
Schiff PB, Horwitz SB (1981) Taxol assembles tubulin in the absence of exogenous guanosine 5′-triphosphate or microtubule-associated proteins. Biochemistry 20:3247–3252
Edsall AB, Mohanakrishnan AK, Yang D et al (2004) Effects of Altering the Electronics of 2-Methoxyestradiol on Cell Proliferation, on Cytotoxicity in Human Cancer Cell Cultures, and on Tubulin Polymerization. J Med Chem 47:5126–5139
Acknowledgements
This study was supported by the grants from the National Natural Science Foundation of China (NSFC) (No.30873092 and No.30721005) and the Science and Technology Commission of Shanghai Municipality (STCSM) (No.08PJ14113 and No.08DZ1980200), respectively.
We sincerely thank Dr. Yi Chen, Mrs. Li-Juan Lu, Mr. Yong Xi and Dr. Hong-Chun Liu for their technical supports.
The authors have no conflicting financial interests.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, Z., Meng, T., He, J. et al. MT7, a novel compound from a combinatorial library, arrests mitosis via inhibiting the polymerization of microtubules. Invest New Drugs 28, 715–728 (2010). https://doi.org/10.1007/s10637-009-9303-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-009-9303-z