Summary
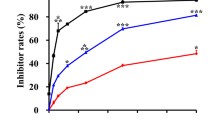
MMPT, a thiazolidin compound, was identified in our laboratory as a novel antineoplastic agent with a broad spectrum of antitumor activity against many human cancer cells. However, the related mechanism has yet not been revealed. In this study, we investigated the cellular and molecular events underlying the antitumous function of this compound in human lung adenocarcinoma H1792 cells, focusing on the early cytotoxic effect. Treatment of H1792 cancer cells with MMPT (0.1–100 μM for 24–72 h) resulted in a growth inhibition in a dose and time-dependent manner, determined by MTT assay. This effect was accompanied by apoptosis, evidenced by Nucleosome ELISA, H33258 stained assay, and Sub-G1 analysis. Our data showed that MMPT caused activation of caspase-3, caspase-6 and caspase-8, but not caspase-9. The finding that MMPT induced apoptosis through a membrane-mediated mechanism was supported by the up-regulated expression of Fas (CD95/APO-1), and Fas ligand. Overall, our results demonstrated that MMPT induced growth inhibition of H1792 cells through a Fas-mediated and caspase-dependent apoptosis pathway, which suggested that MMPT might be used as a Fas/FasL and caspases promoter to initiate lung cancer cell apoptosis.






Similar content being viewed by others
References
Petty RD, Nicolson MC, Kerr KM, Collie-Duguid E, Murray GI (2004) Gene expression profiling in non-small cell lung cancer: from molecular mechanisms to clinical application. Clin Cancer Res 10:3237–3248. doi:10.1158/1078-0432.CCR-03-0503
Kim PK, Park SY, Koty PP, Hua Y, Luketich JD, Billiar TR (2003) Fas-associating death domain protein overexpression induces apoptosis in lung cancer cells. J Thorac Cardiovasc Surg 125(6):1336–1342. doi:10.1016/S0022-5223(02)73227-3
Kandasamy K, Srinivasula SM, Alnemri ES, Thompson CB, Korsmeyer SJ, Bryant JL, Srivastava RK (2003) Involvement of proapoptotic molecules Bax and Bak in tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced mitochondrial disruption and apoptosis: differential regulation of cytochrome c and Smac/DIABLO release. Cancer Res 63(7):1712–1721
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407:770–776. doi:10.1038/35037710
Okun I, Balakin KV, Tkachenko SE, Ivachtchenko AV (2008) Caspase activity modulators as anticancer agents. Anticancer Agents Med Chem 8(3):322–341. doi:10.2174/187152008783961914
Kim R, Tanabe K, Uchida Y, Emi M, Inoue H, Toge T (2002) Current status of the molecular mechanisms of anticancer drug-induced apoptosis. The contribution of molecular-level analysis to cancer chemotherapy. Cancer Chemother Pharmacol 50(5):343–352. doi:10.1007/s00280-002-0522-7
Lu J, Bai L, Sun H, Nikolovska-Coleska Z, McEachern D, Qiu S, Miller RS, Yi H, Shangary S, Sun Y, Meagher JL, Stuckey JA, Wang S (2008) SM-164: a novel, bivalent Smac mimetic that induces apoptosis and tumor regression by concurrent removal of the blockade of cIAP-1/2 and XIAP. Cancer Res 68(22):9384–9393. doi:10.1158/0008-5472.CAN-08-2655
Mousavi SH, Tavakkol-Afshari J, Brook A, Jafari-Anarkooli I (2009) Direct toxicity of Rose Bengal in MCF-7 cell line: role of apoptosis. Food Chem Toxicol 47(4):855–859. doi:10.1016/j.fct.2009.01.018
Su H, Yang JR, Xu T, Huang J, Xu L, Yuan Y, Zhuang SM (2009) MicroRNA-101, down-regulated in hepatocellular carcinoma, promotes apoptosis and suppresses tumorigenicity. Cancer Res 69(3):1135–1142. doi:10.1158/0008-5472.CAN-08-2886
Salgame P, Varadhachary AS, Primiano LL, Fincke JE, Muller S, Monestier M (1997) An ELISA for detection of apoptosis. Nucleic Acids Res 25(3):680–681. doi:10.1093/nar/25.3.680
Herrmann M, Lorenz HM, Voll R, Grünke M, Woith W, Kalden JR (1994) A rapid and simple method for the isolation of apoptotic DNA fragments. Nucleic Acids Res 22(24):5506–5507. doi:10.1093/nar/22.24.5506
Nagata S, Golstein P (1995) The Fas death factor. Science 267:1449–1456. doi:10.1126/science.7533326
Zakeri Z, Lockshin RA (2008) Cell death: history and future. Adv Exp Med Biol 615:1–11. doi:10.1007/978-1-4020-6554-5_1
Stockwell BR (2000) Chemical genetics: ligand-based discovery of gene function. Nat Rev Genet 2:116–125. doi:10.1038/35038557
Schreiber SL (1998) Chemical genetics resulting from a passion for synthetic organic chemistry. Bioorg Med Chem 8:1127–1152. doi:10.1016/S0968-0896(98)00126-6
Noble M, Barrett P, Endicott J (2005) Exploiting structural principles to design cyclin-dependent kinase inhibitors. Biochim Biophys Acta 1754:58–64
Chen J, Nikolovska-Coleska Z, Wang G, Qiu S, Wang S (2006) Design, synthesis, and characterization of new embelin derivatives as potent inhibitors of X-linked inhibitor of apoptosis protein. Bioorg Med Chem Lett 16(22):5805–5808. doi:10.1016/j.bmcl.2006.08.072
Servida F, Soligo D, Delia D, Henderson C, Brancolini C, Lombardi L, Deliliers GL (2005) Sensitivity of human multiple myelomas and myeloid leukemias to the proteasome inhibitor I. Leukemia 19(12):2324–2331. doi:10.1038/sj.leu.2403987
Landis-Piwowar KR, Milacic V, Chen D, Yang H, Zhao Y, Chan TH, Yan B, Dou QP (2006) The proteasome as a potential target for novel anticancer drugs and chemosensitizers. Drug Resist Updat 9(6):263–273. doi:10.1016/j.drup.2006.11.001
Teraishi F, Wu S, Sasaki J, Zhang L, Zhu HB, Davis JJ, Fang B (2005) P-glycoprotein-independent apoptosis induction by a novel synthetic compound, MMPT [5-[(4-methylphenyl) methylene]-2-(phenylamino)-4(5H)-thiazolone]. J Pharmacol Exp Ther 314(1):355–362. doi:10.1124/jpet.105.085654
MacKenzie SH, Clark AC (2008) Targeting cell death in tumors by activating caspases. Curr Cancer Drug Targets 8:98–109. doi:10.2174/156800908783769391
Boatright KM, Salvesen GS (2003) Mechanisms of caspase activation. Curr Opin Cell Biol 15(6):725–731. doi:10.1016/j.ceb.2003.10.009
Debatin KM (2004) Apoptosis pathways in cancer and cancer therapy. Cancer Immunol Immunother 53(3):153–159. doi:10.1007/s00262-003-0474-8
Porter AG, Jänicke RU (1999) Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6(2):99–104. doi:10.1038/sj.cdd.4400476
Roos WP, Kaina B (2006) DNA damage-induced cell death by apoptosis. Trends Mol Med 12(9):440–450. doi:10.1016/j.molmed.2006.07.007
Acknowledgments
This project was supported by the Doctoral Foundation of Shandong Province (2007BS02024), Natural Science Foundation of Shandong Province (Y2008D05) and the 11th Five-year Plan Provincial Key Construction of Qufu Normal University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Yf., Li, Xl., Sun, Yx. et al. MMPT: a thiazolidin compound inhibits the growth of lung cancer H1792 cells via Fas-mediated and caspase-dependent apoptosis pathway. Invest New Drugs 28, 318–325 (2010). https://doi.org/10.1007/s10637-009-9259-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-009-9259-z