Summary
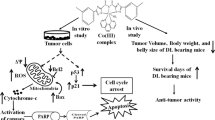
A novel ruthenium(II)-complex containing 4-carboxy N-ethylbenzamide (Ru(II)-CNEB) was found to interact with and inhibit M4-lactate dehydrogenase (M4-LDH), a tumor growth supportive enzyme, at the tissue level. The present article describes modulation of M4-LDH by this compound in a T-cell lymphoma (Dalton’s Lymphoma: DL) vis a vis regression of the tumor in vivo. The compound showed a dose dependent cytotoxicity to DL cells in vitro. When a non toxic dose (10 mg/kg bw i.p.) of Ru(II)-CNEB was administered to DL bearing mice, it also produced a significant decline in DL cell viability in vivo. The DL cells from Ru(II)-CNEB treated DL mice showed a significant decline in the level of M4-LDH with a concomitant release of this protein in the cell free ascitic fluid. A significant increase of nuclear DNA fragmentation in DL cells from Ru(II)-CNEB treated DL mice also coincided with the release of mitochondrial cytochrome c in those DL cells. Importantly, neither blood based biochemical markers of liver damage nor the normal patterns of LDH isozymes in other tissues were affected due to the treatment of DL mice with the compound. These results were also comparable with the effects of cisplatin (an anticancer drug) observed simultaneously on DL mice. The findings suggest that Ru(II)-CNEB is able to regress Dalton’s lymphoma in vivo via declining M4-LDH and inducing mitochondrial dysfunction–apoptosis pathway without producing any toxicity to the normal tissues.











Similar content being viewed by others
References
Clarke MJ (2003) Ruthenium metallopharamceuticals. Coord Chem Rev 236:209–233. doi:10.1016/S0010-8545(02)00312-0
Mishra L, Singh AK, Trigun SK, Singh SK, Pandey SM (2004) Anti HIV and cytotoxic Ruthenium (II) complexes containing flavones: biochemical evaluation in mice. Indian J Exp Biol 42:660–666
Kostova I (2006) Ruthenium complexes as anticancer agents. Curr Med Chem 13:1085–1107. doi:10.2174/092986706776360941
Keppler BK, Berger MR, Klenner T, Heim ME (1990) Metal complexes as antitumour agents. Adv Drug Res 19:243–310
Novakova O, Chen H, Vrana O, Rodger A, Sadler PJ, Brabee Y (2003) DNA interaction of mono functional organometallic Ru (II) anti tumor complexes in cell free media. Biochemistry 42:11544–11554. doi:10.1021/bi034933u
Dyson PJ, Sava G (2006) Metal based anti tumor drugs in the post genomic era. Dalton Trans 16:1929–1933. doi:10.1039/b601840h
Bergamo A, Sava G (2007) Ruthenium complexes can target determinants of tumour malignancy. Dalton Trans 13:1267–1272. doi:10.1039/b617769g
Christofk HR, Heiden MGV, Harris MH, Ramanathan A, Gerszten RE, Wei R et al (2008) The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 452:230–233. doi:10.1038/nature06734
Bregman H, Carroll PJ, Meggers E (2006) Rapid access to unexplored chemical space by ligand scanning around a ruthenium center: discovery of potent and selective protein kinase inhibitors. J Am Chem Soc 128:877–884. doi:10.1021/ja055523r
Kim JW, Gardner LB, Dang CV (2005) Oncogenic alterations of metabolism and the Warburg effect. Drug Discov Today 2:233–238. doi:10.1016/j.ddmec.2005.04.001
Fantin VR, St-Pierre J, Leder P (2006) Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology and tumour maintenance. Cancer Cell 9:425–434. doi:10.1016/j.ccr.2006.04.023
Kim JW, Dang CV (2006) Cancer's molecular sweet tooth and the Warburg effect. Cancer Res 66:8927–8930. doi:10.1158/0008-5472.CAN-06-1501
Kondoh H, Lleonart ME, Gil J, Beach D, Peters G (2005) Glycolysis and cellular immortalization. Drug Discov Today 2:263–267. doi:10.1016/j.ddmec.2005.05.001
Gaber K (2006) Energy deregulation: licensing tumors to grow. Science 312:1158–1159. doi:10.1126/science.312.5777.1158
Moreno-Sánchez R, Rodríguez-Enríquez S, Marín-Hernández A, Saavedra E (2007) Energy metabolism in tumor cells. FEBS J 274:1393–1418. doi:10.1111/j.1742-4658.2007.05686.x
Maher C, Krishan JA, Lampidis TJ (2004) Greater cell cycle inhibition and cytotoxicity induced by 2-deoxy-d-glucose in tumor cells treated under hypoxic vs aerobic conditions. Cancer Chemother Pharmacol 53:116–122. doi:10.1007/s00280-003-0724-7
Geschwind JF, Georgiades CS, Ko YH, Pedersen PL (2004) Recently elucidated energy catabolism pathways provide opportunities for novel treatments in hepatocellular carcinoma. Expert Rev Anticancer Ther 4:449–457. doi:10.1586/14737140.4.3.449
Xu RH, Pelicano H, Zhou Y, Carew JS, Feng L, Bhalla KN et al (2005) Inhibition of glycolysis in cancer cells: a novel strategy to overcome drug resistance associated with mitochondrial respiratory defect and hypoxia. Cancer Res 65:613–621. doi:10.1158/0008-5472.CAN-04-4313
Semenza GL (2003) Targeting HIF-1 for cancer therapy. Nat Rev Cancer 3:721–732. doi:10.1038/nrc1187
Koukourakis M, Giatromanolaki A, Harris AL, Sivridis E (2006) Comparison of metabolic pathways between cancer cells and stromal cells in colorectal carcinomas: a metabolic survival role for tumor associated stroma. Cancer Res 66:632–637. doi:10.1158/0008-5472.CAN-05-3260
Koukourakis M, Giatromanolaki A, Sivridis E (2003) Lactate dehydrogenase isoenzymes 1 and 5: differential expression by neoplastic and stromal cells in non-small cell lung cancer and other epithelial malignant tumors. Tumour Biol 24:199–202. doi:10.1159/000074430
Jaroszewski JW, Kaplan O, Cohen JS (1990) Action of gossypol and rhodamine 123 on wild-type and multidrug-resistant MCF-7 human breast cancer cells: 31P nuclear magnetic resonance and toxicity studies. Cancer Res 50:6936–6943
Coyle T, Levante S, Shetler M, Wintield J (1994) In vitro and in vivo cytotoxicity of gossypol against central nervous system tumor cell lines. J Neurooncol 19:25–35. doi:10.1007/BF01051046
Trigun SK, Koiri RK, Mishra L, Dubey S, Singh S, Pandey P (2007) Ruthenium complex as enzyme modulator: modulation of lactate dehydrogenase by a novel ruthenium(II) complex containing 4-carboxy N-ethylbenzamide as a ligand. Curr Enzym Inhib 3:243–253. doi:10.2174/157340807781369010
Sullivan BP, Salmon DJ, Meyer T (1978) Mixed phosphine 2,2′-bipyridine complexes of ruthenium. Inorg Chem 17:3334–3341. doi:10.1021/ic50190a006
Prasad SB, Giri A (1999) Effect of cisplatin on the lactate dehydrogenase activity and its isozyme pattern in Dalton's lymphoma bearing mice. Cytologia (Tokyo) 64:259–267
Pathak C, Vinayak M (2005) Modulation of lactate dehydrogenase isozymes by modified base queuine. Mol Biol Rep 32:191–196. doi:10.1007/s11033-004-6941-2
Sellins KS, Cohen JJ (1987) Gene induction by γ-irradiation leads to DNA fragmentation in lymphocytes. J Immunol 139:3199–3206
Kuo CL, Chou CC, Yung BY (1995) Berberine complexes with DNA in the berberine-induced apoptosis in human leukemic HL-60 cells. Cancer Lett 93:193–200. doi:10.1016/0304-3835(95)03809-B
Pandey P, Singh SK, Trigun SK (2005) Fructose-2, 6-bisphosphate associated regulatory enzymes develop in concordance in mice brain during early postnatal life. Neurol Psychiatry Brain Res 12:69–74
Wang H (2000) Over expression of L-PhGPx in MCF-7 cells. In: The role of mitochondrial phospholipids hydroperoxide glutathione peroxide in cancer therapy, Ph.D. thesis, The University of Iowa, Iowa. 2000, pp 16–56
Koiri RK, Trigun SK, Dubey SK, Singh S, Mishra L (2008) Metal Cu(II) and Zn(II) bipyridyls as inhibitors of lactate dehydrogenase. Biometals 21:117–126. doi:10.1007/s10534-007-9098-3
Singh S, Koiri RK, Trigun SK (2008) Acute and chronic hyperammonemia modulate antioxidant enzymes differently in cerebral cortex and cerebellum. Neurochem Res 33:103–113. doi:10.1007/s11064-007-9422-x
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Parajuli P, Singh SM (1996) Alteration of IL-1 and arginase activity of tumor-associated macrophages: a role in the promotion of tumor growth. Cancer Lett 107:249–256. doi:10.1016/0304-3835(96)04378-9
Prasad SB, Giri A (1994) Antitumor effect of cisplatin against murine ascites Dalton’s lymphoma. Indian J Exp Biol 32:155–162
Khynriam D, Prasad SB (2003) Cisplatin-induced genotoxic effects and endogenous glutathione levels in mice bearing ascites Dalton’s lymphoma. Mutat Res 526:9–18. doi:10.1016/S0027-5107(03)00005-8
Sava G, Pacor S, Bergamo A, Cocchietto M, Mestroni G, Alessio E (1995) Effects of ruthenium complexes on experimental tumors: irrelevance of cytotoxicity for metastasis inhibition. Chem Biol Interact 95:109–126. doi:10.1016/0009-2797(94)03350-1
Stefanini M (1985) Enzymes, isozymes, and enzyme variants in the diagnosis of cancer. A short review. Cancer 55:1931–1936. doi:10.1002/1097-0142(19850501)55:9<1931::AID-CNCR2820550917>3.0.CO;2-M
Rudnicki M, Oliveira MR, Pereira TV, Reginatto FH, Pizzol DF, Moreira JCF (2007) Antioxidant and antiglycation properties of Passiflora alata and Passiflora edulis extracts. Food Chem 100:719–724. doi:10.1016/j.foodchem.2005.10.043
Giraldi T, Sava G, Bertoli G, Mestroni G, Zassinovich G (1977) Antitumor action of two rhodium and ruthenium complexes in comparison with cis-Diamminedichloroplatinum(II). Cancer Res 37:2662–2666
Ajith TA, Janardhanan KK (2003) Cytotoxic and antitumor activities of a polypore macrofungus, Phellinus rimosus (Berk) Pilat. J Ethnopharmacol 84:157–162. doi:10.1016/S0378-8741(02)00292-1
Christina AJM, Joseph GD, Packialakshmi M, Kothai R, Robert SJH, Chidambaranathan N et al (2004) Anticarcinogenic activity of Withania somnifera Dunal against Dalton’s ascitic lymphoma. J Ethnopharmacol 93:359–361. doi:10.1016/j.jep.2004.04.004
Nicholson DW (1996) From the bench to clinic with apoptosis-based therapeutic agents. Nature 407:810–816. doi:10.1038/35037747
Kuwahara D, Tsutsumi K, Kobayashi T, Hasunuma T, Nishioka K (2000) Caspace-9 regulates cisplatin-induced apoptosis in human head and neck squamous cell carcinoma cells. Cancer Lett 148:65–71. doi:10.1016/S0304-3835(99)00315-8
Gaiddon C, Jeannequin P, Bischoff P, Pferrer M, Sirlin C, Loeffler JP (2005) Ruthenium (II)-derived organometallic compounds induce cytostatic and cytotoxic effects on mammalian cancer cell lines through p53-dependent and p53-independent mechanisms. J Pharmacol Exp Ther 315:1403–1411. doi:10.1124/jpet.105.089342
Jiang X, Wang X (2004) Cytochrome c mediated apoptosis. Annu Rev Biochem 73:87–106. doi:10.1146/annurev.biochem.73.011303.073706
López-Lázaro M (2007) Digitoxin as an anticancer agent with selectivity for cancer cells: possible mechanisms involved. Expert Opin Ther Targets 11:1043–1053. doi:10.1517/14728222.11.8.1043
Pederson PL (1978) Tumor mitochondria and the bioenergetics of cancer cells. Prog Exp Tumor Res 22:198–274
Hockenbery DM, Oltavi ZN, Yin XM, Milliman CL, Korsmeyer SJ (1993) Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 75:241–251. doi:10.1016/0092-8674(93)80066-N
Forastiere A (1994) Overview of platinum chemotherapy in head and neck cancer. Semin Oncol 21:20–27
Siddik ZH (2003) Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 22:7265–7279. doi:10.1038/sj.onc.1206933
Khalaila I, Bergamo A, Bussy F, Sava G, Dyson PJ (2006) The role of cisplatin and NAMI-A plasma–protein interactions in relation to combination therapy. Int J Oncol 29:261–268
Fraiser LH, Kanekal S, Kehrer JP (1991) Cyclophosphamide toxicity. Characterizing and avoiding the problem. Drugs 42:781–795
Acknowledgment
This work was financially supported by a project from Department of Biotechnology (DBT), Govt. of India, (BT/PR5910/BRB/10/406/2005) sanctioned jointly to LM and SKT. The authors are thankful to UGC Centre of Advanced Studies programme to Department of Zoology, BHU, for providing infrastructural facilities. The help extended by Mr. S. Bhattacharyya, Ms. S. Srivastav, and Ms. B. Mishra is also acknowledged.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koiri, R.K., Trigun, S.K., Mishra, L. et al. Regression of Dalton’s lymphoma in vivo via decline in lactate dehydrogenase and induction of apoptosis by a ruthenium(II)-complex containing 4-carboxy N-ethylbenzamide as ligand. Invest New Drugs 27, 503–516 (2009). https://doi.org/10.1007/s10637-008-9202-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-008-9202-8