Summary
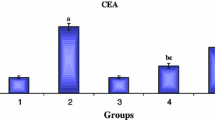
Liver cancer is the sixth most common cancer worldwide but because of very poor prognosis, it is the third most common cause of death from cancer. There are currently limited therapeutic regimens available for effective treatment of this cancer. Silymarin is a naturally derived polyphenolic antioxidant, is the active constituent in a widely consumed dietary supplement milk thistle (Silybum marianum) extract. Mast cells play an important role in the inflammatory component of a developing neoplasm; they are also a major source for matrix metalloproteinases (MMPs), which are involved in invasion and angiogenesis. In the present study, we investigated whether dietary supplementation of silymarin has any role in mast cell density (MCD) and in the expressions of MMP-2 and MMP-9 in N-nitrosodiethylamine induced (NDEA) liver cancer in Wistar albino male rats. NDEA administered rats showed increased MCD as revealed by toluidine blue staining along with upregulated expressions of MMP-2 and MMP-9. Silymarin treatment inhibited this increase in MCD and downregulated the expressions of MMP-2 and MMP-9 as revealed by Western blotting and immunohistochemistry. In conclusion, silymarin exerted beneficial effects on liver carcinogenesis by attenuating the recruitment of mast cells and thereby decreased the expressions of MMP-2 and MMP-9.




Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
El-Serag HB, Mason AC (1999) Rising incidence of hepatocellular carcinoma in the United States. N Engl J Med 340:745–750. doi:10.1056/NEJM199903113401001
El-Serag HB, Mason AC, Key C (2001) Trends in survival of patients with hepatocellular carcinoma between 1977 and 1996 in the United States. Hepatology 33:62–65. doi:10.1053/jhep.2001.21041
Hecht SS (1997) Approaches to cancer prevention based on an understanding of N-nitrosamine carcinogenesis. Proc Soc Exp Biol Med 216(2):181–191
Fine DH, Ross R, Rounbehler DP, Silvergleid A, Song L (1977) Formation in vivo of volatile N-nitrosamines in man after ingestion of cooked bacon and spinach. Nature 265:753–755. doi:10.1038/265753a0
Zhao J, Agarwal R (1999) Tissue distribution of silibinin, the major active constituent of silymarin, in mice and its association with enhancement of phase II enzymes: implications in cancer chemoprevention. Carcinogenesis 20(11):2101–2108. doi:10.1093/carcin/20.11.2101
Kohno H, Suzuki R, Sugie S, Tsuda H, Tanaka T (2005) Dietary supplementation with silymarin inhibits 3,2¢-dimethyl-4-aminobiphenyl-induced prostate carcinogenesis in male F344 rats. Clin Cancer Res 11(13):4962–4967. doi:10.1158/1078-0432.CCR-05-0137
Ramakrishnan G, Muzio LL, Elinos-Baez CM, Jagan S, Augustine TA, Kamaraj S, Devaki T (2008) Silymarin inhibited proliferation and induced apoptosis in hepatic cancer cells. Cell Prolif (in press)
Kaur M, Agarwal R (2007) Silymarin and epithelial cancer chemoprevention: how close are we to bedside? Toxicol Appl Pharmacol 224(3):350–359. doi:10.1016/j.taap.2006.11.011
Ramakrishnan G, Raghavendran HR, Vinodhkumar R, Devaki T (2006) Suppression of N-nitrosodiethylamine induced hepatocarcinogenesis by silymarin in rats. Chem Biol Interact 161(2):104–114. doi:10.1016/j.cbi.2006.03.007
Ramakrishnan G, Augustine TA, Jagan S, Vinodhkumar R, Devaki T (2007) Effect of silymarin on N-nitrosodiethylamine induced hepatocarcinogenesis in rats. Exp Oncol 29(1):39–44
Bhatia N, Zhao J, Wolf DM, Agarwal R (1999) Inhibition of human carcinoma cell growth and DNA synthesis by silibinin, an active constituent of milk thistle: comparison with silymarin. Cancer Lett 147:77–84. doi:10.1016/S0304-3835(99)00276-1
Katiyar SK (2005) Silymarin and skin cancer prevention: anti-inflammatory, antioxidant and immunomodulatory effects. Int J Oncol 26:169–176
Kleiner DE, Stetler-Stevenson WG (1993) Structural biochemistry and activation of matrix metalloproteinases. Curr Opin Cell Biol 5:891–897. doi:10.1016/0955-0674(93)90040-W
Uchima Y, Sawada T, Hirakawa K (2007) Action of antiproteases on pancreatic cancer cells. JOP 8(4 Suppl):479–487
Bergers G, Javaherian K, Lo K, Folkman J, Hanahan D (1999) Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science 284:808–812. doi:10.1126/science.284.5415.808
Coussens ML, Fingleton B, Matrisian LM (2002) Matrix metalloproteinase inhibitors and cancer: trials and tribulations. Science 295:2387–2392. doi:10.1126/science.1067100
Deryugina EI, Quigley JP (2006) Matrix metalloproteinases and tumor metastasis. Cancer Metastasis Rev 25:9–34. doi:10.1007/s10555-006-7886-9
Brower GL, Chancey AL, Thanigaraj S, Matsubara BB, Janicki JS (2002) Cause and effect relationship between myocardical mast cell number and matrix metalloproteinase activity. Am J Physiol Heart Circ Physiol 283:H518–H525
Heissig B, Rafii S, Akiyama H, Ohki Y, Sato Y, Rafael T et al (2005) Low-dose irradiation promotes tissue revascularization through VEGF release from mast cells and MMP-9-mediated progenitor cell mobilization. J Exp Med 202(6):739–750. doi:10.1084/jem.20050959
Coussens LM, Raymond WW, Bergers G, Laig-Webster M, Bendrendtsen O, Werb Z et al (1999) Inflammatory mast cells up-regulated angiogenesis during squamous epithelial carcinogenesis. Genes Dev 13:1382–1397. doi:10.1101/gad.13.11.1382
Ranieri G, Labriola A, Achille G, Florio G, Zito AF, Grammatica L et al (2002) Microvessel density, mast cell density and thymidine phosphorylase expression in oral squamous carcinoma. Int J Oncol 21(6):1317–1323
Peng SH, Deng H, Yang J, Xie PP, Li C, Li H et al (2005) Significance and relationship between infiltrating inflammatory cell and tumor angiogenesis in hepatocellular carcinoma tissues. World J Gastroenterol 11(41):6521–6524
Minn AJ, Gupta GP, Siegel PM, Bos PD, Shu W, Giri DD et al (2005) Genes that mediate breast cancer metastasis to lung. Nature 436:518–524. doi:10.1038/nature03799
Gounaris E, Erdman SE, Restaino C, Gurish MF, Friend DS, Gounari F et al (2007) Mast cells are an essential hematopoietic component for polyp development. Proc Natl Acad Sci USA 104(50):19977–19982. doi:10.1073/pnas.0704620104
Rodewald HR, Dessing M, Dvorak AM, Galli SJ (1996) Identification of a committed precursor for the mast cell lineage. Science 271:818–822. doi:10.1126/science.271.5250.818
Dundar E, Oner U, Peker BC, Metintas M, Isiksoy S, Ak G (2008) The significance and relationship between mast cells and tumour angiogenesis in non-small cell lung carcinoma. J Int Med Res 36(1):88–95
Rajput AB, Turbin DA, Cheang MC, Voduc DK, Leung S, Gelmon KA et al (2008) Stromal mast cells in invasive breast cancer are a marker of favourable prognosis: a study of 4,444 cases. Breast Cancer Res Treat 107(2):249–257. doi:10.1007/s10549-007-9546-3
Grizzi F, Franceschini B, Chiriva-Internati M, Liu Y, Hermonat PL, Dioguardi N (2003) Mast cells and human hepatocellular carcinoma. World J Gastroenterol 9(7):1469–1473
DeLaPuerta R, Martinez E, Bravo L, Ahumada MC (1996) Effect of silymarin on different acute inflammation models and on leukocyte migration. J Pharm Pharmacol 48(9):968–970
Ramakrishnan G, Elinos-Baez CM, Jagan S, Augustine TA, Kamaraj S, Anandakumar P et al (2008) Silymarin downregulates COX-2 expression and attenuates hyperlipidaemia during NDEA induced rat hepatocellular carcinoma. Mol Cell Biochem 313(1–2):53–61. doi:10.1007/s11010-008-9741-5
Jia JD, Bauer M, Cho JJ, Ruehl M, Milani S, Boigk G et al (2001) Antifibrotic effect of silymarin in rat secondary biliary fibrosis is mediated by downregulation of procollagen alpha1(I) and TIMP-1. J Hepatol 35(3):392–398. doi:10.1016/S0168-8278(01)00148-9
Zheng M, Ruan Y, Wu Z (2000) Correlation study of TGF beta expression in diethylnitrosamine-induced rat liver cancer and mast cell in its vicinity. Zhonghua Zhong Liu Za Zhi 22(6):463–465
Tchnougounova E, Lundequist A, Fajardo I, Winberg J, Abrink M, Pejler G (2005) A key role for mast cell chymase in the activation of pro-matrix metalloprotease-9 and pro-matrix metalloprotease-2. J Biol Chem 280(10):9291–9296. doi:10.1074/jbc.M410396200
Hiratsuka S, Nakamura K, Iwai S, Murakami M, Itoh T, Kijima H et al (2002) MMP-9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell 2(4):289–300. doi:10.1016/S1535-6108(02)00153-8
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174. doi:10.1038/nrc745
Liotta LA, Tryggvason K, Garbisa S, Hart I, Foltz CM, Shatic S (1980) Metastatic potential correlates with enzymatic degradation of basement-membrane collagen. Nature 284:67–68. doi:10.1038/284067a0
Hanahan D, Folkman J (1996) Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86:353–364. doi:10.1016/S0092-8674(00)80108-7
Stetler-Stevenson WG (1999) Matrix metalloproteinases in angiogenesis: a moving target for therapeutic intervention. J Clin Invest 103:1237–1241. doi:10.1172/JCI6870
Chantrian CF, Henriet P, Jodele S, Emonard H, Feron O, Courtoy PJ et al (2006) Mechanisms of pericyte recruitment in tumor angiogenesis: a new role for metalloproteinases. Eur J Cancer 42(3):310–318 doi:10.1016/j.ejca.2005.11.010
Coussens LM, Tinkle CL, Hanahan D, Werb Z (2000) MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 103:481–490. doi:10.1016/S0092-8674(00)00139-2
Bergers G, Brekken R, McMahon G, Vu TH, Itoh T, Tamaki K et al (2000) Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nat Cell Biol 2:737–744. doi:10.1038/35036374
Itoh T, Tanioka M, Yoshida H, Yoshioka T, Nishimoto H, Itohara S (1998) Reduced angiogenesis and tumor progression in gelatinase A-deficient mice. Cancer Res 58:1048–1051
Girolamo ND, Indoh I, Jackson N, Wakefield D, McNeil HP, Yan W et al (2006) Human mast cell-derived gelatinase B (matrix metalloproteinase-9) is regulated by inflammatory cytokines: role in cell migration. J Immunol 177:2638–2650
Davidson B, Givant-Horwitz V, Lazarovici P, Risberg B, Nesland JM, Trope CG et al (2003) Matrix metalloproteinases (MMP), EMMPRIN (extracellular matrix metalloproteinase inducer) and mitogen-activated protein kinases (MAPK): co-expression in metastatic serous ovarian carcinoma. Clin Exp Metastasis 20(7):621–631. doi:10.1023/A:1027347932543
Singh RP, Tyagi AK, Zhao J, Agarwal R (2002) Silymarin inhibits growth and causes regression of established skin tumors in SENCAR mice via modulation of mitogen-activated protein kinases and induction of apoptosis. Carcinogenesis 23(3):499–510. doi:10.1093/carcin/23.3.499
Acknowledgements
The authors wish to thank Prof. W.G. Stetler-Stevenson, NIH, USA for his kind gift of antibodies to MMP-2 and MMP-9. This work was supported in part by Indian Council for Medical Research (ICMR), Government of India, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found online at http://dx.doi.org/10.1007/s10637-012-9892-9.
Rights and permissions
About this article
Cite this article
Ramakrishnan, G., Jagan, S., Kamaraj, S. et al. Silymarin attenuated mast cell recruitment thereby decreased the expressions of matrix metalloproteinases-2 and 9 in rat liver carcinogenesis. Invest New Drugs 27, 233–240 (2009). https://doi.org/10.1007/s10637-008-9163-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-008-9163-y