Abstract
Purpose
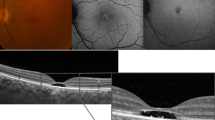
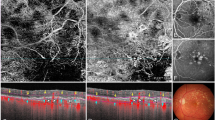
Pericentral visual field changes and disruption of the ellipsoid layer on spectral domain optical coherence tomography (SD-OCT) are the main features of antimalarial retinal toxicity. C-Scan OCT or “en face” enables a topographic frontal view of the changes observed within the different retinal layers in particular the ellipsoid layer. The aim of this prospective study was to compare multifocal ERG (mfERG) responses with the results of C-Scan OCT (“en face” OCT) in patients with abnormal visual field and to analyze relationships between the structural and functional abnormalities.
Methods
In 354 consecutive patients screened for antimalarial toxicity between January 1, 2014 and December 31, 2016, central visual field, mfERG recording, C-Scan OCT and short-wavelength fundus autofluorescent imaging were performed.
Results
Among the 17/354 patients with abnormal central visual field results, all presented with abnormalities on the mfERG at least in one eye. In 16/33 eyes, there was a good concordance between focal loss of the mfERG response and the disruption of the ellipsoid layer on C-Scan OCT. In one eye with characteristic changes in the ellipsoid layer on the C-Scan OCT, the mfERG was normal, whereas in three eyes the mfERG was abnormal in eyes with a normal C-Scan OCT.
Conclusions
The contribution of the C-Scan OCT changes remains difficult to establish as there is no strict concordance with the local ERG responses. Although C-Scan OCT technology provides a new approach in analyzing focal abnormalities in the photoreceptor–retinal pigment epithelium interface, the sensitivity of this method compared with mfERG and other tests (central visual field, B-Scan OCT) needs to be evaluated. This study is still ongoing on a larger cohort.











Similar content being viewed by others
References
Ingster-Moati I, Orssaud C (2009) Protocole de surveillance ophtalmologique des patients traités par antipaludéens de synthèse ou par vigabatrin au long cours. J Fr Ophtalmol 32:83–88
Rynes RI (1997) Antimalarial drugs in the treatment of rheumatological diseases. Rheumatology 36:799–805
Brinkley JR, Dubois EL, Ryan SJ (1979) Long-term course of chloroquine retinopathy after cessation of medication. Am J Ophthalmol 88:1–11
Moschos M, Nitoda E, Chatziralli IP et al (2015) Assessment of hydroxychloroquine maculopathy after cessation of treatment: an optical coherence tomography and multifocal electroretinography study. Drug Des Dev Ther 9:2993
Marmor MF, Kellner U, Lai TYY et al (2016) Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology 123:1386–1394
Marmor MF, Kellner U, Lai TYY et al (2011) Revised Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy. Ophthalmology 118:415–422
Marmor MF (2016) The demise of the bulls eye (Screening for hydroxychloroquine retinopathy). Retina 36:1803–1805
Kellner U, Renner AB, Tillack H (2006) Fundus autofluorescence and mfERG for early detection of retinal alterations in patients using chloroquine/hydroxychloroquine. Invest Opthalmol Vis Sci 47:3531
Tsang AC, Ahmadi Pirshahid S, Virgili G, Gottlieb CC, Hamilton J, Coupland SG (2015) Hydroxychloroquine and chloroquine retinopathy: a systematic review evaluating the multifocal electroretinogram as a screening test. Ophthalmology 122:1239–1251.
Østerberg GA (1935) Topography of the layer of rods and cones in the human retina. Acta Ophthalmol 6:1–102
Browning DJ, Lee C (2014) Relative sensitivity and specificity of 10-2 visual fields, multifocal electroretinography, and spectral domain optical coherence tomography in detecting hydroxychloroquine and chloroquine retinopathy. Clin Ophthalmol 8:1389–1399
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakera’s bureaus; membership; employment; consultancies; stock ownership; or other equity interest; and expert testimony or patent-licensing arrangements) or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Arndt, C., Costantini, M., Chiquet, C. et al. Comparison between multifocal ERG and C-Scan SD-OCT (“en face” OCT) in patients with a suspicion of antimalarial retinal toxicity: preliminary results. Doc Ophthalmol 136, 97–111 (2018). https://doi.org/10.1007/s10633-018-9625-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-018-9625-2