Abstract
Background
To date, mechanisms of intestinal immunoglobulin (Ig) dysfunction following intestinal ischemia/reperfusion (I/R) remain unclear. Programmed death 1 (PD-1) is associated with immune responses of lymphocytes.
Aim
We aimed to verify the hypothesis that activation of PD-1 may improve intestinal immune dysfunction by regulating IL-10/miR-155 production after intestinal IR injury.
Methods
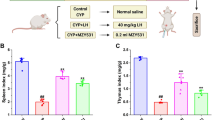
Intestinal I/R injury was induced in mice by clamping the superior mesenteric artery for 1 h followed by 2-h reperfusion. PD-L1 fusion Ig, anti-interleukin (IL)-10 monoclonal antibody (mAb), and microRNA (miR)-155 agomir were administered. PD-1 expression, IL-10 mRNA, and protein expression in Peyer’s patches (PP) CD4+ cells were measured. MiR-155 levels, tumor necrosis factor (TNF)-α and IL-1β concentration, and activation-induced cytidine deaminase (AID), a key enzyme for intestinal immune antibodies, in PP tissues were measured, respectively. Importantly, the production and cecal bacteria-binding capacity of IgA and IgM were detected.
Results
Intestinal I/R led to decreased PD-1 expression, imbalanced production, and impaired bacteria-binding capacity of IgA and IgM. Activating PD-1 by PD-L1 Ig facilitated IL-10 synthesis, then decreased miR-155 levels, and subsequently promoted AID expression and reduced TNF-α, IL-1β concentration. Upregulation of AID improved the disruptions of intestinal immune barrier caused by IgA and IgM dysfunction. Anti-IL-10 mAb and miR-155 agomir abolished the protective effects of PD-L1 Ig on the intestinal immune defense.
Conclusion
Activation of PD-1 with PD-L1 Ig relieves intestinal immune defensive injury through IL-10/miR-155 pathway following intestinal I/R attack. PD-1, IL-10, and miR-155 may be potential targets for the damages of intestinal barrier and immunity.





Similar content being viewed by others
References
Mallick IH, Yang W, Winslet MC, et al. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49:1359–1377. https://doi.org/10.1023/B:DDAS.0000042232.98927.91.
Zhang XY, Liu ZM, Wen SH, et al. Dexmedetomidine administration before, but not after, ischemia attenuates intestinal injury induced by intestinal ischemia-reperfusion in rats. Anesthesiology. 2012;116:1035–1046.
Fukatsu K, Sakamoto S, Hara E, et al. Gut ischemia-reperfusion affects gut mucosal immunity: a possible mechanism for infectious complications after severe surgical insults. Crit Care Med. 2006;34:182–187.
Wang F, Li Q, Wang C, et al. Dynamic alteration of the colonic microbiota in intestinal ischemia-reperfusion injury. PLoS ONE. 2012;7:e42027.
MacFie J, O’Boyle C, Mitchell CJ, et al. Gut origin of sepsis: a prospective study investigating associations between bacterial translocation, gastric microflora, and septic morbidity. Gut. 1999;45:223–228.
Deitch EA. Gut-origin sepsis: evolution of a concept. Surgeon. 2012;10:350–356.
Pabst O. New concepts in the generation and functions of IgA. Nat Rev Immunol. 2012;12:821–832.
Li H, Wu GH, Chen J. Effect of glucagon-like peptide 2 on the intestinal mucosal immunity and correlative cytokines in mice with gut ischemia/reperfusion injury. Zhonghua Wei Chang Wai Ke Za Zhi. 2006;9:67–70.
Zhang XY, Liu ZM, Zhang HF, et al. TGF-β1 improves mucosal IgA dysfunction and dysbiosis following intestinal ischaemia-reperfusion in mice. J Cell Mol Med. 2016;20:1014–1023.
Lenaerts K, Ceulemans LJ, Hundscheid IH, et al. New insights in intestinal ischemia-reperfusion injury: implications for intestinal transplantation. Curr Opin Organ Transplant. 2013;18:298–303.
Francisco LM, Sage PT, Sharpe AH. The PD-1 pathway in tolerance and autoimmunity. Immunol Rev. 2010;236:219–242.
Brown KE, Freeman GJ, Wherry EJ, et al. Role of PD-1 in regulating acute infections. Curr Opin Immunol. 2010;22:397–401.
Esch KJ, Juelsgaard R, Martinez PA, et al. Programmed death 1-mediated T cell exhaustion during visceral leishmaniasis impairs phagocyte function. J Immunol. 2013;191:5542–5550.
Kawamoto S, Tran TH, Maruya M, et al. The inhibitory receptor PD-1 regulates IgA selection and bacterial composition in the gut. Science. 2012;336:485–489.
Zhang XY, Liu ZM, Zhang HF, et al. Decreased PD-1/PD-L1 expression is associated with the reduction in mucosal immunoglobulin A in mice with intestinal ischemia reperfusion. Dig Dis Sci. 2015;60:2662–2669. https://doi.org/10.1007/s10620-015-3684-y.
Jankovic D, Kugler DG, Sher A. IL-10 production by CD4 + effector T cells: a mechanism for self-regulation. Mucosal Immunol. 2010;3:239–246.
Fairfax KA, Gantier MP, Mackay F, et al. IL-10 regulates Aicda expression through miR-155. J Leukoc Biol. 2015;97:71–78.
Liao W, Zheng H, Wu S, et al. The systemic activation of programmed death 1-PD L1 axis protects systemic lupus erythematosus model from nephritis. Am J Nephrol. 2017;46:371–379.
Chen WJ, Hu XF, Yan M, et al. Human umbilical vein endothelial cells promote the inhibitory activation of CD4(+)CD25(+)Foxp3(+) regulatory T cells via PD-L1. Atherosclerosis. 2016;244:108–112.
Zhang XY, Chen X, Zhang HF, et al. Propofol does not reduce pyroptosis of enterocytes and intestinal epithelial injury after lipopolysaccharide challenge. Dig Dis Sci. 2018;63:81–91. https://doi.org/10.1007/s10620-017-4801-x.
Proietti M, Cornacchione V, RezzonicoJost T, et al. ATP-gated ionotropic P2X7 receptor controls follicular T helper cell numbers in Peyer’s patches to promote host-microbiota mutualism. Immunity. 2014;41:789–801.
Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009;9:799–809.
Zan H, Casali P. Regulation of Aicda expression and AID activity. Autoimmunity. 2013;46:83–101.
Fagarasan S, Muramatsu M, Suzuki K, et al. Critical roles of activation-induced cytidine deaminase in the homeostasis of gut flora. Science. 2002;298:1424–1427.
Ji H, Shen X, Gao F, et al. Programmed death-1/B7-H1 negative costimulation protects mouse liver against ischemia and reperfusion injury. Hepatology. 2010;52:1380–1389.
Jaworska K, Ratajczak J, Huang L, et al. Both PD-1 ligands protect the kidney from ischemia reperfusion injury. J Immunol. 2015;194:325–333.
Roussey JA, Viglianti SP, Teitz-Tennenbaum S, et al. Anti-PD-1 antibody treatment promotes clearance of persistent cryptococcal lung infection in mice. J Immunol. 2017;199:3535–3546.
Zhang Y, Du W, Chen Z, Xiang C. Upregulation of PD-L1 by SPP1 mediates macrophage polarization and facilitates immune escape in lung adenocarcinoma. Exp Cell Res. 2017;359:449–457.
Manukyan MC, Alvernaz CH, Poynter JA, et al. Interleukin-10 protects the ischemic heart from reperfusion injury via the STAT3 pathway. Surgery. 2011;150:231–239.
Wan X, Huang WJ, Chen W, et al. IL-10 deficiency increases renal ischemia-reperfusion injury. Nephron Exp Nephrol. 2014;128:37–45.
Fouda AY, Pillai B, Dhandapani KM, et al. Role of interleukin-10 in the neuroprotective effect of the angiotensin type 2 receptor agonist, compound 21, after ischemia/reperfusion injury. Eur J Pharmacol. 2017;799:128–134.
Basso K, Schneider C, Shen Q, et al. BCL6 positively regulates AID and germinal center gene expression via repression of miR-155. J Exp Med. 2012;209:2455–2465.
Eisenhardt SU, Weiss JB, Smolka C, et al. MicroRNA-155 aggravates ischemia-reperfusion injury by modulation of inflammatory cell recruitment and the respiratory oxidative burst. Basic Res Cardiol. 2015;110:32.
Pena-Philippides JC, Caballero-Garrido E, Lordkipanidze T, Roitbak T. In vivo inhibition of miR-155 significantly alters post-stroke inflammatory response. J Neuroinflammation. 2016;13:287.
Li Y, Ma D, Wang Z, Yang J. MicroRNA-155 deficiency in Kupffer cells ameliorates liver ischemia-reperfusion injury in mice. Transplantation. 2017;101:1600–1608.
Acknowledgments
The authors thank Jie Li (Laboratory Animal Center, Sun Yat-sen University) for her help in animal experiments.
Funding
The present study was supported by Grants from the Natural Science Foundation of Guangdong Province, China (Grant 2017A030313572 to Xu-Yu Zhang) and grants from the National Natural Science Foundation of China (Grant 81701874 to Zi-meng Liu).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, XY., Guan, S., Zhang, HF. et al. Activation of PD-1 Protects Intestinal Immune Defense Through IL-10/miR-155 Pathway After Intestinal Ischemia Reperfusion. Dig Dis Sci 63, 3307–3316 (2018). https://doi.org/10.1007/s10620-018-5282-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-5282-2