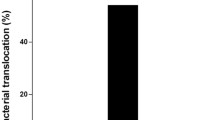Abstract
Background
Bacterial translocation is considered a major cause of initiation and development of systemic sepsis and multiple organ dysfunction in clinic. The aim of this study was to determine the ability of a defined Lactobacillus plantarum to prevent ischemia/reperfusion (I/R) induced intestinal infection.
Methods
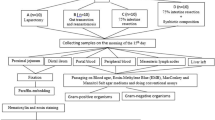
Female Sprague-Dawley rats were randomly allocated into three groups: (1) controls (sham-operated, no treatment), (2) ischemia/reperfusion and (3) ischemia/reperfusion and Lactobacillus plantarum treatment. Lactobacillus plantarum L2 was administered daily intragastrically 14 days prior to induction of I/R. Rats were then sacrificed, and tissue and blood samples were cultured to determine bacterial translocation. Cytokines in plasma were detected by ELISA. Ileal segments were removed for morphological examination.
Results
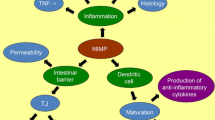
Intestinal I/R induced excess pro-inflammatory cytokine secretion and barrier dysfunction (increased epithelial cell apoptosis, cecal flora dysbiosis, disruption of mucosa and multiple erosions) in the intestine, associated with increased bacterial translocation to extraintestinal sites. Approximately 87.5% of rats exposed to I/R had bacterial translocation while there was no bacterial translocation in controls. However, pretreatment of animals with Lactobacillus plantarum completely prevented I/R induced bacterial translocation, reduced pro-inflammatory cytokine release, and intestinal epithelial cell apoptosis, resulting in recovered microflora and mucosal integrity.
Conclusions
These findings indicate that Lactobacillus plantarum L2 can prevent I/R-induced bacterial translocation and intestinal barrier dysfunction and, thereby, exert beneficial effects in the intestinal tract.



Similar content being viewed by others
References
Berg RD. The indigenous gastrointestinal microflora. Trends Microbiol. 1996;4:430–435.
Simon GL, Gorbach SL. The human intestinal microflora. Dig Dis Sci. 1986;31:147S–162S.
Hopkins MJ, Macfarlane GT. Changes in predominant bacterial populations in human faeces with age and with Clostridium difficile infection. J Med Microbiol. 2002;51:448–454.
MacFie J, O’Boyle C, Mitchell C, Buckley P, Johnstone D, Sudworth P. Gut origin of sepsis: a prospective study investigating associations between bacterial translocation, gastric microflora, and septic morbidity. Gut. 1999;45:223–228.
Berg RD. Bacterial translocation from the gastrointestinal tract. Trends Microbiol. 1995;3:149–154.
Wells CL, VandeWesterlo EM, Jechorek RP, Erlandsen SL. Effect of hypoxia on enterocyte endocytosis of enteric bacteria. Crit Care Med. 1996;24:985–991.
Kong SE, Blennerhassett LR, Heel KA, McCauley RD, Hall JC. Ischemia-reperfusion injury to the intestine. Aust N Z J Surg. 1998;68:554–561.
Kuzu MA, Kale IT, Cöl C, Tekeli A, Tanik A, Köksoy C. Obstructive jaundice promotes bacterial translocation in humans. Hepatogastroenterology. 1999;46:2159–2164.
Cicalese L, Sileri P, Green M, Abu-Elmagd K, Kocoshis S, Reyes J. Bacterial translocation in clinical intestinal transplantation. Transplantation. 2001;71:1414–1417.
Cirera I, Bauer TM, Navasa M, et al. Bacterial translocation of enteric organisms in patients with cirrhosis. J Hepatol. 2001;34:32–37.
Peitzman AB, Udekwu AO, Ochoa J, Smith S. Bacterial translocation in trauma patients. J Trauma. 1991;31:1083–1086.
Riddington DW, Venkatesh B, Boivin CM, et al. Intestinal permeability, gastric intramucosal pH, and systemic endotoxemia in patients undergoing cardiopulmonary bypass. JAMA. 1996;275:1007–1012.
Hebra A, Hong J, McGowan KL, Smith C, Mckernan ML, Ross AJ 3rd. Bacterial translocation in mesenteric ischemia-reperfusion injury: is dysfunctional motility the link. J Pediatr Surg. 1994;29:280–285.
Leaphart CL, Tepas JJ 3rd. The gut is a motor of organ system dysfunction. Surgery. 2007;141:563–569.
Mattila-Sandholm T, Mättö J, Saarela M. Lactic acid bacteria with health claims—interactions and interference with gastrointestinal flora. Int Dairy J. 1999;9:25–35.
Merk K, Borelli C, Korting HC. Lactobacilli-bacteria-host interactions with special regard to the urogenital tract. Int J Med Microbiol. 2005;295:9–18.
Foligne B, Nutten S, Grangette C, et al. Correlation between in vitro and in vivo immunomodulatory properties of lactic acid bacteria. W J Gastroenterol. 2007;13:236–243.
Sartor RB. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: antibiotics, probiotics and prebiotics. Gastroenterology. 2004;126:1620–1633.
Biller JA, Katz AJ, Flores AF, Buie TM, Gorbach SL. Treatment of recurrent Clostridium difficile colitis with Lactobacillus GG. J Pediatr Gastroenterol Nutr. 1995;21:224–226.
Madsen KL, Doyle JS, Jewell LD, Tavernini MM, Fedorak RN. Lactobacillus species prevents colitis in interleukin 10 gene-deficient mice. Gastroenterology. 1999;116:1107–1114.
Mangell P, Nejdfors P, Wang M, et al. Lactobacillus plantarum 299V inhibits Escherichia coli-induced intestinal permeability. Dig Dis Sci. 2002;47:511–516.
Zareie M, Johnson-Henry K, Jury J, et al. Probiotics prevent bacterial translocation and improve intestinal barrier function in rats following chronic psychological stress. Gut. 2006;55:1553–1560.
Wang B, Li J, Zhang H, Li Q, Li N. Isolation of adhesive strains and evaluation of the colonization and immune response by Lactobacillus plantarum L2 in the rat gastrointestinal tract. Int J Food Microbiol. 2009;132:59–66.
Yamamoto S, Tanabe M, Wakabayashi G, Shimazu M, Matsumoto K, Kitajima M. The role of tumor necrosis factor-α and interleukin-1β in ischemia-reperfusion injury of the rat small intestine. J Surg Res. 2001;99:134–141.
Chiu CJ, McArdle AH, Brown R, Scott HJ, Gurd FN. Intestinal mucosal lesion in low-flow states. I. A morphological, hemodynamic, and metabolic reappraisal. Arch Surg. 1970;101:478–483.
van Minnen LP, Timmerman HM, Lutgendorff F, et al. Modification of intestinal flora with multispecies probiotics reduces bacterial translocation and improves clinical course in a rat model of acute pancreatitis. Surgery. 2007;141:470–480.
Roller M, Rechkemmer G, Watzl B. Prebiotic inulin enriched with oligofructose in combination with the probiotics Lactobacillus rhamnosus and Bifidobacterium lactis modulates intestinal immune functions in rats. J Nutr. 2004;134:153–156.
Dalwai F, Spratt DA, Pratten J. Modeling shifts in microbial populations associated with health or disease. Appl Environ Microbiol. 2006;72:3678–3684.
Stecher B, Hardt WD. The role of microbiota in infectious disease. Trends Microbiol. 2008;16:107–114.
Grotz MR, Deitch EA, Ding J, Xu D, Huang Q, Regel G. Intestinal cytokine response after gut ischemia: role of gut barrier failure. Ann Surg. 1999;229:478–486.
Kurtel H, Fujimoto K, Zimmerman BJ, Granger DN, Tso P. Ischemia-reperfusion-induced mucosal dysfunction: role of neutrophils. Am J Physiol. 1991;261:G490–G496.
Grotz MR, Ding J, Guo W, Huang Q, Deitch EA. Comparison of plasma cytokine levels in rats subjected to superior mesenteric artery occlusion or hemorrhagic shock. Shock. 1995;3:362–368.
Souza DG, Teixeira MM. The balance between the production of tumor necrosis factor-alpha and interleukin-10 determines tissue injury and lethality during intestinal ischemia and reperfusion. Mem Inst Oswaldo Cruz. 2005;100:59–66.
Zeng H, Wu H, Sloane V, et al. Flagellin/TLR5 responses in epithelia reveal intertwined activation of inflammatory and apoptotic pathways. Am J Physiol Gastrointest Liver Physiol. 2006;290:G96–G108.
Wu B, Qiu W, Wang P, et al. p53 independent induction of PUMA mediates intestinal apoptosis in response to ischaemia-reperfusion. Gut. 2007;56:645–654.
Yan F, Polk DB. Probiotic bacterium prevents cytokine-induced apoptosis in intestinal epithelial cells. J Biol Chem. 2002;277:50959–50965.
Acknowledgments
The present work was financially supported by the National Natural Science Foundation of China (Grants 30801090 and 30830098).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, B., Huang, Q., Zhang, W. et al. Lactobacillus plantarum Prevents Bacterial Translocation in Rats Following Ischemia and Reperfusion Injury. Dig Dis Sci 56, 3187–3194 (2011). https://doi.org/10.1007/s10620-011-1747-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1747-2