Abstract
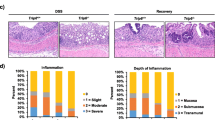
Trefoil factors (TFFs) are essential for protection and restitution of the gastrointestinal mucosa but many aspects of TFF biology are unclear. Our aim was to compare the localization of endogenous TFFs and binding sites for injected TFF3 in the colon of healthy and colitic mice and to study the effect of TFF3 on dextrane sulfate sodium (DSS)-induced colitis in mice. Expression of endogenous TFF1-3 was examined by in situ hybridization and immunohistochemistry, and the distribution of intravenously, intraperitoneally, and subcutaneously administered 125I-TFF3 by autoradiography and gamma-counting. The effect of systemically administered TFF3 on DSS-induced colitis was assessed. We found increased expression of endogenous TFF3 and increased binding of injected 125I-TFF3 in the colon of animals with DSS-induced colitis. The distribution of intraperitoneally and subcutaneously administered 125I-TFF3 was comparable. Systemic administration of the peptides reduced the severity of colitis. Expression of endogenous TFF3 and binding of systemically administered TFF3 are increased in DSS-induced colitis. Systemic administration of TFF3 attenuates the disease. These findings suggest a role of TFF3 in mucosal protection.





Similar content being viewed by others
References
Hoffmann W, Jagla W, Wiede A (2001) Molecular medicine of TFF-peptides: from gut to brain. Histol Histopathol 16:319–334
Wright NA, Poulsom R, Stamp G, et al. (1992) Trefoil peptide gene expression in gastrointestinal epithelial cells in inflammatory bowel disease. Scand J Gastroenterol Suppl 193:76–82
Alison MR, Chinery R, Poulsom R, Ashwood P, Longcroft JM, Wright NA (1995) Experimental ulceration leads to sequential expression of spasmolytic polypeptide, intestinal trefoil factor, epidermal growth factor and transforming growth factor alpha mRNAs in rat stomach. J Pathol 175:405–414
Xian CJ, Howarth GS, Mardell CE, Cool JC, Familari M, Read LC, Giraud AS (1999) Temporal changes in TFF3 expression and jejunal morphology during methotrexate-induced damage and repair. Am J Physiol 277:G785–G795
Mashimo H, Wu DC, Podolsky DK, Fishman MC (1996) Impaired defense of intestinal mucosa in mice lacking intestinal trefoil factor. Science 274:262–265
Playford RJ, Marchbank T, Goodlad RA, Chinery RA, Poulsom R, Hanby AM (1996) Transgenic mice that overexpress the human trefoil peptide pS2 have an increased resistance to intestinal damage. Proc Natl Acad Sci USA 93:2137–2142
Marchbank T, Cox HM, Goodlad RA, Giraud AS, Moss SF, Poulsom R, Wright NA, Jankowski J, Playford RJ (2001) Effect of ectopic expression of rat trefoil factor family 3 (intestinal trefoil factor) in the jejunum of transgenic mice. J Biol Chem 276:24088–24096
Farrell JJ, Taupin D, Koh TJ, Chen D, Zhao CM, Podolsky DK, Wang TC (2002) TFF2/SP-deficient mice show decreased gastric proliferation, increased acid secretion, and increased susceptibility to NSAID injury. J Clin Invest 109:193–204
Babyatsky MW, deBeaumont M, Thim L, Podolsky DK (1996) Oral trefoil peptides protect against ethanol- and indomethacin-induced gastric injury in rats. Gastroenterology 110:489–497
Playford RJ, Marchbank T, Chinery R, Evison R, Pignatelli M, Boulton RA, Thim L, Hanby AM (1995) Human spasmolytic polypeptide is a cytoprotective agent that stimulates cell migration. Gastroenterology 108:108–116
Poulsen SS, Thulesen J, Christensen L, Nexo E, Thim L (1999) Metabolism of oral trefoil factor 2 (TFF2) and the effect of oral and parenteral TFF2 on gastric and duodenal ulcer healing in the rat. Gut 45:516–522
Poulsen SS, Kissow H, Hare K, Hartmann B, Thim L (2005) Luminal and parenteral TFF2 and TFF3 dimer and monomer in two models of experimental colitis in the rat. Regul Pept 126:163–171
FitzGerald AJ, Pu M, Marchbank T, Westley BR, May FE, Boyle J, Yadollahi-Farsani M, Ghosh S, Playford RJ (2004) Synergistic effects of systemic trefoil factor family 1 (TFF1) peptide and epidermal growth factor in a rat model of colitis. Peptides 25:793–801
Taupin D, Podolsky DK (2003) Trefoil factors: initiators of mucosal healing. Nat Rev Mol Cell Biol 4:721–732
Cook GA, Familari M, Thim L, Giraud AS (1999) The trefoil peptides TFF2 and TFF3 are expressed in rat lymphoid tissues and participate in the immune response. FEBS Lett 456:155–159
Tebbutt NC, Giraud AS, Inglese M, Jenkins B, Waring P, Clay FJ, Malki S, Alderman BM, Grail D, Hollande F, Heath JK, Ernst M (2002) Reciprocal regulation of gastrointestinal homeostasis by SHP2 and STAT-mediated trefoil gene activation in gp130 mutant mice. Nat Med 8:1089–1097
Baus-Loncar M, Schmid J, Lalani EN, Rosewel I, Goodlad RA, Stamp GWH, Blin N, Kayademir T (2005) Trefoil factor 2 (Tff2) deficiency in murine digestive tract influences the immune system. Cell Physiol Biochem 16:31–42
Poulsen SS, Thulesen J, Hartmann B, Kissow HL, Nexo E, Thim L (2003) Injected TFF1 and TFF3 bind to TFF2-immunoreactive cells in the gastrointestinal tract in rats. Regul Pept 115:91–99
Chinery R, Cox HM (1995) Immunoprecipitation and characterization of a binding protein specific for the peptide, intestinal trefoil factor. Peptides 16:749–755
Tan XD, Liu QP, Hsueh W, Chen YH, Chang H, Gonzalez-Crussi F (1999) Intestinal trefoil factor binds to intestinal epithelial cells and induces nitric oxide production: priming and enhancing effects of mucin. Biochem J 338(Pt 3):745–751
Thim L, Mortz E (2000) Isolation and characterization of putative trefoil peptide receptors. Regul Pept 90:61–68
Otto W, Thim L (2005) Trefoil factor family-interacting proteins. Cell Mol Life Sci 62:2939–2946
Zhang BH, Yu HG, Sheng ZX, Luo HS, Yu JP (2003) The therapeutic effect of recombinant human trefoil factor 3 on hypoxia-induced necrotizing enterocolitis in immature rat. Regul Pept 116:53–60
Soriano-Izquierdo A, Gironella M, Massaguer A, May FEB, Salas A, Sans M, Poulsom R, Thim L, Pique JM, Panes JN (2004) Trefoil peptide TFF2 treatment reduces VCAM-1 expression and leukocyte recruitment in experimental intestinal inflammation. J Leukoc Biol 75:214–223
Thim L, Woldike HF, Nielsen PF, Christensen M, Lynch-Devaney K, Podolsky DK (1995) Characterization of human and rat intestinal trefoil factor produced in yeast. Biochemistry 34:4757– 4764
Tomita M, Itoh H, Ishikawa N, Higa A, Ide H, Murakumo Y, Maruyama H, Koga Y, Nawa Y (1995) Molecular cloning of mouse intestinal trefoil factor and its expression during goblet cell changes. Biochem J 311(Pt 1):293–297
Mashimo H, Podolsky DK, Fishman MC (1995) Structure and expression of murine intestinal trefoil factor: high evolutionary conservation and postnatal expression. Biochem Biophys Res Commun 210:31–37
Pyke C, Romer J, Kallunki P, Lund LR, Ralfkiaer E, Dano K, Tryggvason K (1994) The gamma 2 chain of kalinin/laminin 5 is preferentially expressed in invading malignant cells in human cancers. Am J Pathol 145:782–791
Vestergaard EM, Poulsen SS, Gronbaek H, Larsen R, Nielsen AM, Ejskjaer K, Clausen JT, Thim L, Nexo E (2002) Development and evaluation of an ELISA for human trefoil factor 3. Clin Chem 48:1689–1695
Murthy S, Murthy NS, Coppola D, Wood DL (1997) The efficacy of BAY y 1015 in dextran sulfate model of mouse colitis. Inflamm Res 46:224–233
Bradley PP, Christensen RD, Rothstein G (1982) Cellular and extracellular myeloperoxidase in pyogenic inflammation. Blood 60:618–622
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Williams KL, Fuller CR, Dieleman LA, DaCosta CM, Haldeman KM, Sartor RB, Lund PK (2001) Enhanced survival and mucosal repair after dextran sodium sulfate-induced colitis in transgenic mice that overexpress growth hormone. Gastroenterology 120:925–937
Byrne FR, Farrell CL, Aranda R, Rex KL, Scully S, Brown HL, Flores SA, Gu LH, Danilenko DM, Lacey DL, Ziegler TR, Senaldi G (2002) RHuKGF ameliorates symptoms in DSS and CD4(+)CD45RB(Hi) T cell transfer mouse models of inflammatory bowel disease. Am J Physiol Gastrointest Liver Physiol 282:G690–G701
Renes IB, Verburg M, Van Nispen DJPM, Taminiau JaJM, Buller HA, Dekker J, Einerhand AWC (2002) Epithelial proliferation, cell death, and gene expression in experimental colitis: alterations in carbonic anhydrase I, mucin MUC2, and trefoil factor 3 expression. Int J Colorect Dis 17:317–326
Wright NA, Poulsom R, Stamp GW, Hall PA, Jeffery RE, Longcroft JM, Rio MC, Tomasetto C, Chambon P (1990) Epidermal growth factor (EGF/URO) induces expression of regulatory peptides in damaged human gastrointestinal tissues. J Pathol 162:279–284
Poulsom R, Chinery R, Sarraf C, Lalani EN, Stamp G, Elia G, Wright N (1992) Trefoil peptide expression in intestinal adaptation and renewal. Scand J Gastroenterol Suppl 192:17–28
Vieten D, Corfield A, Carroll D, Ramani P, Spicer R (2005) Impaired mucosal regeneration in neonatal necrotising enterocolitis. Pediatr Surg Int 21:153–160
Poulsen SS, Thulesen J, Nexo E, Thim L (1998) Distribution and metabolism of intravenously administered trefoil factor 2/porcine spasmolytic polypeptide in the rat. Gut 43:240–247
Efstathiou JA, Noda M, Rowan A, Dixon C, Chinery R, Jawhari A, Hattori T, Wright NA, Bodmer WF, Pignatelli M (1998) Intestinal trefoil factor controls the expression of the adenomatous polyposis coli-catenin and the E-cadherin-catenin complexes in human colon carcinoma cells. Proc Natl Acad Sci USA 95:3122–3127
Kanai M, Mullen C, Podolsky DK (1998) Intestinal trefoil factor induces inactivation of extracellular signal-regulated protein kinase in intestinal epithelial cells. Proc Natl Acad Sci USA 95:178–182
Emami S, Le Floch N, Bruyneel E, Thim L, May F, Westley B, Rio M, Mareel M, Gespach C (2001) Induction of scattering and cellular invasion by trefoil peptides in src- and RhoA-transformed kidney and colonic epithelial cells. FASEB J 15:351–361
Rodrigues S, Van Aken E, Van Bocxlaer S, Attoub S, Nguyen QD, Bruyneel E, Westley BR, May FE, Thim L, Mareel M, Gespach C, Emami S (2003) Trefoil peptides as proangiogenic factors in vivo and in vitro: implication of cyclooxygenase-2 and EGF receptor signaling. FASEB J 17:7–16
Andoh A, Kinoshita K, Rosenberg I, Podolsky DK (2001) Intestinal trefoil factor induces decay-accelerating factor expression and enhances the protective activities against complement activation in intestinal epithelial cells. J Immunol 167:3887–3893
Tanaka S, Podolsky DK, Engel E, Guth PH, Kaunitz JD (1997) Human spasmolytic polypeptide decreases proton permeation through gastric mucus in vivo and in vitro. Am J Physiol 272:G1473–G1480
Acknowledgements
S. Kjellev was supported by a student fellowship from the Danish Ministry for Science, Technology and Innovation. The authors are grateful to the Danish Ministry of Science, Technology and Development and the Danish Research Council for financial support of this study. We thank Grazyna Poulsen, Jette Schousboe, Axel K. Hansen, Bjørg Harryson, Jette Mandelbaum, Steen Kryger, Ann-Christin Løff, and Bodil E. Andersen.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kjellev, S., Thim, L., Pyke, C. et al. Cellular Localization, Binding Sites, and Pharmacologic Effects of TFF3 in Experimental Colitis in Mice. Dig Dis Sci 52, 1050–1059 (2007). https://doi.org/10.1007/s10620-006-9256-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-9256-4