Abstract
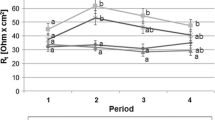
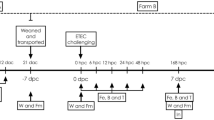
Pretreatment with the probiotic Escherichia colistrain Nissle 1917 (EcN) was assessed in a pig model of intestinal infection to prevent acute secretory diarrhea. In the model 1010 colony forming units of the porcine enterotoxigenic Escherichia coli Abbotstown (EcA) was given via orogastric tube to weaned piglets at day 21 postpartum (−EcN/+EcA group, n = 7). Forty-eight hours after challenge electrophysiological parameters of isolated intact jejunal epithelia were characterized in Ussing chambers. In agreement with clinical signs of diarrhea, tissues of challenged animals showed an overshoot of secretory response after stimulation of the cAMP-mediated second messenger pathway by forskolin, indicating higher excitability of chloride secretory systems under infected conditions. The data were compared with respective measurements from animals that got a daily dose of 1010 cfu of the probiotic EcN over 10 days before EcA challenge (+EcN/+EcA group; n = 4), from a group that received only EcN (+EcN/–EcA; n = 4), or from a group that remained totally untreated (−EcN/−EcA; n = 6). EcN pretreatment completely abolished clinical signs of secretory diarrhea in +EcN/+EcA animals. Furthermore, jejunum epithelia of these animals did not exhibit an overshoot of secretory response upon stimulation with forskolin. Our studies demonstrate for the first time the efficacy of prophylactic EcN in pig small intestine for preventing an effect of toxigenic EcA. This infection model with freshly weaned piglets may be predestinated to further characterize EcN effects on the cellular level, i.e., involved second messenger pathways, or it may also be useful to examine the efficacy of other substrates or microbe strains against secretory stimuli.
Similar content being viewed by others
References
Schrezenmeier J, de Vrese M: Probiotics, prebiotics, and synbiotics—approaching a definition. Am J Clin Nutr 73:361S–364S, 2001
Gionchetti P, Rizzello F, Venturi, A, Campieri M: Probiotics in infective diarroea and inflammatory diseases. J Gastroenterol Hepatol 15:489–493, 2000
Gill HS, Guarner F: Probiotics and human health: a clinical perspective. Postgrad Med J 80:516–526, 2004
Fedorak RN, Madsen KL: Probiotics and the management of inflammatory bowel disease. Inflamm Bowel Dis 10:286–299, 2004
Grozdanov L, Raasch C, Schulze J, Sonnenborn U, Gottschalk G, Hacker J, Dobrindt U: Analysis of the genome structure of the nonpathogenic probiotic Escherichia coli strain Nissle 1917. J Bacteriol 186:5432–5441, 2004
Kruis W, Fric P, Pokrotnieks J, Lukas M, Fixa B, Kascak M, Kamm MA, Weismueller J, Beglinger C, Stolte M, Wolff C, Schulze J: Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut 53:1617–1623, 2004
Rembacken BJ, Snelling AM, Hawkey PM, Chalmers DM, Axon AT: Non-pathogenic Escherichia coli versus mesalazine for the treatment of ulcerative colitis: a randomised trial. Lancet 354:635–639, 1999
Tannock GW, Munro K, Harmsen HJ, Welling GW, Smart J, Gopal PK: Analysis of the fecal microflora of human subjects consuming a probiotic product containing Lactobacillus rhamnosusDR20. Appl Environ Microbiol 66:2578–2588, 2000
Cross ML, Ganner A, Teilab D, Fray LM: Patterns of cytokine induction by gram-positive and gram-negative probiotic bacteria. FEMS Immunol Med Microbiol 42:173–180, 2004
Schultz M, Strauch UG, Linde HJ, Watzl S, Obermeier F, Gottl C, Dunger N, Grunwald N, Schölmerich J, Rath HC: Preventive effects of Escherichia coli strain Nissle 1917 on acute and chronic intestinal inflammation in two different murine models of colitis. Clin Diagn Lab Immunol 11:372–378, 2004
Schultz M, Linde HJ, Lehn N, Zimmermann K, Grossmann J, Falk W, Scholmerich J: Immunomodulatory consequences of oral administration of Lactobacillus rhamnosus strain GG in healthy volunteers. J Dairy Res 70:165–173, 2003
Wehkamp J, Harder J, Wehkamp K, Wehkamp–von Meissner B, Schlee M, Enders C, Sonnenborn U, Nuding S, Bengmark S, Fellermann K, Schroder JM, Stange EF: NF-kappaB- and AP-1-mediated induction of human beta defensin-2 in intestinal epithelial cells by Escherichia coli Nissle 1917: a novel effect of a probiotic bacterium. Infect Immun 72:5750–5758, 2004
Altenhoefer A, Oswald S, Sonnenborn U, Enders C, Schulze J, Hacker J, Oelschlaeger TA: The probiotic Escherichia coli strain Nissle 1917 interferes with invasion of human intestinal epithelial cells by different enteroinvasive bacterial pathogens. FEMS Immunol Med Microbiol 40:223–229, 2004
Boudeau J, Glasser AL, Julien S, Colombel JF, Darfeuille–Michaud A: Inhibitory effect of probiotic Escherichia coli strain Nissle 1917 on adhesion to and invasion of intestinal epithelial cells by adherent–invasive E. coli strains isolated from patients with Crohn’s disease. Aliment Pharmacol Ther 18:45–56, 2003
Otte JM, Podolsky DK: Functional modulation of enterocytes by gram-positive and gram-negative microorganisms. Am J Physiol Gastrointest Liver Physiol 286:G613–G626, 2004
Jurjus AR, Khoury NN, Reimund JM: Animal models of inflammatory bowel disease. J Pharmacol Toxicol Methods 50:81–92, 2004
Dieleman LA, Hoentjen F, Qian BF, Sprengers D, Tjwa E, Torres MF, Torrice CD, Sartor RB, Tonkonogy SL: Reduced ratio of protective versus proinflammatory cytokine responses to commensal bacteria in HLA-B27 transgenic rats. Clin Exp Immunol 136:30–39, 2004
Schultz M, Veltkamp C, Dieleman LA, Grenther WB, Wyrick PB, Tonkonogy SL, Sartor RB: Lactobacillus plantarum 299V in the treatment and prevention of spontaneous colitis in interleukin-10-deficient mice. Inflamm Bowel Dis 8:71–80, 2002
Cahill, EE, Glantz PJ: Demonstration of K88ac and K88ab antigens of Escherichia coli by means of immunoelectrophoresis and immunodiffusion. Infect Immun 20:811–815, 1978
Anonymous: Amtliche Sammlung von Untersuchungsverfahren nach S 35 LMBG. Untersuchung von Lebensmitteln, Bestimmung der aeroben Keimzahl bei 30°C in Fleisch und Fleischerzeugnissen. Tropfplatten-Verfahren 1984, L06.0/0/9
Blum-Oehler G, Oswald S, Eiteljorge K: Development of strain-specific PCR reactions for the detection of the probiotic Escherichia coli strain Nissle 1917 in fecal samples. Res Microbiol 154:59–66, 2003
To TL, Ward LA, Yuan L, Saif LJ: Serum and intestinal isotype antibody responses and correlates of protective immunity to human rotavirus in a gnotobiotic pig model of disease. J Gen Virol 79:2661–2672, 1998
Holt, JG, Krieg NR, Sneath PHA, Staley JT, Williams ST: Bergey’s Manual of Determinative Bacteriology, 9th ed. Baltimore, MD, Lippincott Williams & Wilkins, 1994
Wieler LH, Bauerfeind R: STEC as a veterinary problem. Diagnostics and prophylaxis in animals. Meth Mol Med 73:75–89, 2003
Bauerfeind R, Wieler LH, Weiss R, Baljer G: Vergleichende Plasmidprofilanalyse von Salmonella typhimuriumvar. Copenhagen-Stàmmen aus einem Salmonellenausbruch bei Klinikpferden. Berl Münch Tieràrztl Wochenschr 105:38–42, 1992
Jones GF, Ward GE, Murtaugh MP, Lin G, Gebhart CJ: Enhanced detection of intracellular organisms of swine proliferative enteritis, ileal symbiont intracellularis, in feces by polymerase chain reaction. J Clin Microbiol 31:2611–2615, 1993
Herbst W, Willems H, Baljer G: Verbreitung von Brachyspira hyodysenteriae und Lawsonia intracellularis bei gesunden und durchfallkranken Schweinen. Berl Münch Tieràrztl Wochenschr 117:493–498, 2004
Herbst W, Lange H, Zschöck M, Krauss H: Nachweis von Kàlberrotavirus mit dem Latextest Rotascreen® und dem Elektronenmikroskop—eine vergleichende Untersuchung. Dtsch Tieràrztl Wochenschr 93:317–319, 1986
Schröder B, Winckler C, Failing K, Breves G: Studies on the time course of the effects of the probiotic yeast Saccharomyces boulardii on electrolyte transport in pig jejunum. Dig Dis Sci 49:1311–1317, 2004
Schröder B, Kaune R, Harmeyer J: Effects of calcitriol on stimulation of ion transport in pig jejunal mucosa. J Physiol (London) 433:451–465, 1991
Schultz SG, Zalusky R: Ion transport in isolated rabbit ileum: I. Short-circuit current and Na fluxes. J Gener Physiol 47:567–584, 1964
Buenau R von, Jaekel L, Schubotz E, Schwarz S, Stroff T, Krueger M: Escherichia coli strain Nissle 1917: Significant reduction of neonatal calf diarrhea. J Dairy Sci 88:317–323, 2005
Lammers KM, Helwig U, Swennen E, Rizzello F, Venturi A, Caramelli E, Kamm MA, Brigidi P, Gionchetti P, Campieri M: Effect of probiotic strains on interleukin 8 production by HT2/9/9A cells. Am J Gastroenterol 97:1182–1186, 2002
Hampson DJ, Hinton M, Kidder DE: Coliform numbers in the stomach and small intestine of healthy pigs following weaning at three weeks of age. J Comp Pathol 95:353–62, 1985
Hinton M, Hampson DJ, Hampson E, Linton AH: A comparison of the ecology of Escherichia coli in the intestine of healthy unweaned pigs and pigs after weaning. J Appl Bacteriol 58:471–477, 1985
Egberts HJ, de Groot EC, van Dijk JE, Vellenga L, Mouwen JM: Tight junctional structure and permeability of porcine jejunum after enterotoxic Escherichia coli infection. Res Vet Sci 55:10–14, 1993
Breves G, Walter C, Burmester M, Schröder B: In vitro studies on the effects of Saccharomyces boulardii and Bacillus cereus var. toyoi on nutrient transport in pig jejunum. J Anim Physiol Anim Nutr 84:9–20, 2000
Carey HV, Hayden UL, Tucker KE: Fasting alters basal and stimulated ion transport in piglet jejunum. Am J Physiol 267:R156–R163, 1994
Cermak R, Follmer U, Wolffram S: Dietary flavonol quercetin induces chloride secretion in rat colon. Am J Physiol 275:G1166–G1172, 1998
Eto B, Boisset M, Desjeux JF: Sodium fluoride inhibits the antisecretory effect of peptide YY and its analog in rabbit jejunum. Arch Physiol Biochem 104:180–184, 1996
Brown DR, Overend MF, Treder BG: Neurohormonal regulation of ion transport in the porcine distal jejunum. Actions of somatostatin-14 and its natural and synthetic homologs. J Pharmacol Exp Ther 252:126–134, 1990
Winckler C, Schröder B, Breves G: Effects of Saccharomyces boulardii, Bacillus cereus var. caron and Bacillus cereus var. toyoi on epithelial transport functions in pig jejunum. Z Gastroenterol 36 (Suppl 1):30–37, 1998
Czerucka D, Roux I, Rampal P: Saccharomyces boulardii inhibits secretagogue-mediated adenosine 3′,5′-cyclic monophosphate induction in intestinal cells. Gastroenterol 106:65–72, 1994
Czerucka D, Nano JL, Bernasconi P, Rampal P: Response to cholera toxin of 2 epithelial intestinal cell lines. Effect of Saccharomyces boulardii. Gastroenterol Clin Biol 13:383–387, 1989
Michail S, Abernathy F: Lactobacillus plantarum reduces the in vitro secretory response of intestinal epithelial cells to enteropathogenic Escherichia coli infection. J Pediatr Gastroenterol Nutr 35:350–355, 2002
Thiagarajah JR, Verkman AS: CFTR pharmacology and ist role in intestinal fluid secretion. Curr Op Pharmacol 3:594–599, 2003
Nilius B, Droogmans G: Amazing chloride channels: an overview. Acta Physiol Scan 177:119–147, 2003
Alcantara C, Stenson WF, Steiner TS, Guerrant RL: Role of inducible cyclooxygenase and prostaglandins in Clostridium difficile toxin A-induced secretion and inflammation in an animal model. J Infect Dis 184:648–652, 2001
Castagliuolo I, Riegler M, Pasha A, Nikulasson S, Lu B, Gerard C, Gerard NP, Pothoulakis C: Neurokinin-1 (NK-1) receptor is required in Clostridium difficile- induced enteritis. J Clin Invest 101:1547–1550, 1998
Santos J, Perdue MH: Stress and neuroimmune regulation of gut mucosal function. Gut 47 (Suppl 4):49–51, 2000
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schroeder, B., Duncker, S., Barth, S. et al. Preventive Effects of the Probiotic Escherichia coli Strain Nissle 1917 on Acute Secretory Diarrhea in a Pig Model of Intestinal Infection. Dig Dis Sci 51, 724–731 (2006). https://doi.org/10.1007/s10620-006-3198-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-3198-8