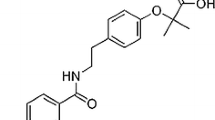
Abstract
Vildagliptin and sitagliptin are commonly used antidiabetic drugs. Chromosomal aberration (CA), sister chromatid exchange (SCE) and cytokinesis-block micronucleus (CBMN) assays were employed to assess and compare cytotoxic and genotoxic effects of these drugs. Peripheral lymphocytes were exposed to 125 μg/ml, 250 μg/ml and 500 μg/ml of vildagliptin and 250 μg/ml, 500 μg/ml and 1000 μg/ml of sitagliptin for 24 h and 48 h with and without exogenous metabolic activation. At the end of the study, it was determined that these drugs and their metabolites had no genotoxic effects on CA, SCE and CBMN. On the other hand, parallel to the increase in dose, vildagliptin showed weak cytotoxicity on the mitotic index, and depending on its increase in dose; sitagliptin caused potential cytotoxicity and cytostatic effect on the mitotic index, nuclear division index and proliferation index. Due to their cytotoxic and cytostatic potential, these drugs inhibit cell proliferation.
Similar content being viewed by others
References
Amritha CA, Kumaravelu P, Chellathai DD (2015) Evaluation of anti cancer effects of DPP-4 inhibitors in colon cancer- an invitro study. J Clin Diagn Res 9:FC14–FC16. https://doi.org/10.7860/jcdr/2015/16015.6979
Andrade SE et al (2004) Prescription drug use in pregnancy. Am J Obstet Gynecol 191:398–407. https://doi.org/10.1016/j.ajog.2004.04.025
Atlı Şekeroğlu Z, Şekeroğlu V (2011) Genetik toksisite testleri. TÜBAV Bilim Dergisi 4:221–229
Caballero AE (2017) Long-term studies of treatments for type 2 diabetes. Postgrad Med 129:352–365. https://doi.org/10.1080/00325481.2017.1265417
Cordero OJ, Salgado FJ, Nogueira M (2009) On the origin of serum CD26 and its altered concentration in cancer patients Cancer Immunology. Immunotherapy 58:1723–1747. https://doi.org/10.1007/s00262-009-0728-1
Corvi R, Madia F (2017) In vitro genotoxicity testing—can the performance be enhanced? Food Chem Toxicol 106:600–608. https://doi.org/10.1016/j.fct.2016.08.024
DeFronzo RA et al (2015) Type 2 diabetes mellitus. Nat Rev Dis Primers 1:15019. https://doi.org/10.1038/nrdp.2015.19
Eastmond DA et al (2009) Mutagenicity testing for chemical risk assessment: update of the WHO/IPCS harmonized scheme. Mutagenesis 24:341–349
EMA (2007) Sitagliptin (Januvia) scientific discussion. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Scientific_Discussion/human/000722/WC500039057.pdf. Accessed 06 Feb 2019
EMA EMA (2012) Vildagliptin (Galvus) scientific discussion. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Summary_for_the_public/human/000771/WC500020328.pdf. Accessed 06 Feb 2019
Engüzel C (2015) Bir anti-diyabetik ilaç etken maddesi olan sitagliptin’in tek başına ve metformin ile birlikte ın vıtro genotoksik etkilerinin belirlenmesi. Yüksek Lisans Tezi, Gazi Üniversitesi
Evans H, O’Riordan ML (1975) Human peripheral blood lymphocytes for the analysis of chromosome aberrations in mutagen tests. Mutat Res 31:135–148
Femia AP, Raimondi L, Maglieri G, Lodovici M, Mannucci E, Caderni G (2013) Long-term treatment with Sitagliptin, a dipeptidyl peptidase-4 inhibitor, reduces colon carcinogenesis and reactive oxygen species in 1,2-dimethylhydrazine-induced rats. Int J Cancer 133:2498–2503. https://doi.org/10.1002/ijc.28260
Fenech M (2000) The in vitro micronucleus technique. Mutat Res 455:81–95. https://doi.org/10.1016/S0027-5107(00)00065-8
Giovannucci E et al (2010) Diabetes and cancer: a consensus report. CA Cancer J Clin 60:207–221. https://doi.org/10.3322/caac.20078
Gonzalez-Gil G, Navarrete M (1982) On the mechanism of differential Giemsa staining of BrdU-substituted chromatids. Chromosoma 86:375–382
Harishankar MK, Logeshwaran S, Sujeevan S, Aruljothi KN, Dannie MA, Devi A (2015) Genotoxicity evaluation of metformin and glimepiride by micronucleus assay in exfoliated urothelial cells of type 2 diabetes mellitus patients. Food Chem Toxicol 83:146–150. https://doi.org/10.1016/j.fct.2015.06.013
Hayashi M (2016) The micronucleus test—most widely used in vivo genotoxicity test. Genes Environ 38:18. https://doi.org/10.1186/s41021-016-0044-x
IDF (2015) IDF Diabetes Atlas Karakas Print. https://www.idf.org/e-library/epidemiology-research/diabetes-atlas/13-diabetes-atlas-seventh-edition.html. Accessed 07 July 2017
Jena GB, Kaul CL, Ramarao P (2002) Genotoxicity testing. A regulatory requirement for drug discovery and development: impact of Ich guidelines. Indian J Pharmacol 34:86–99
Kocaman AY, Rencüzoğulları E, Topaktaş M (2014) In vitro investigation of the genotoxic and cytotoxic effects of thiacloprid in cultured human peripheral blood lymphocytes. Environ Toxicol 29:631–641
Kontaş S, Atlı Şekeroğlu Z (2015) Investigation of cytotoxic and genotoxic effects of the antihistaminic drug, loratadine, on human lymphocytes. Drug Chem Toxicol 38:57–62
Laffon B, Pásaro E, Méndez J (2001) Genotoxic effects of styrene-7, 8-oxide in human white blood cells: comet assay in relation to the induction of sister-chromatid exchanges and micronuclei. Mutat Res 491:163–172
Levetan C (2007) Oral antidiabetic agents in type 2 diabetes. Curr Med Res Opin 23:945–952. https://doi.org/10.1185/030079907x178766
Ma J et al (2017) CMD-05, a novel promising clinical anti-diabetic drug candidate, in vivo and vitro studies. Sci Rep 7:46628
Maddika S et al (2007) Cell survival, cell death and cell cycle pathways are interconnected: implications for cancer therapy. Drug Resist Updates 10:13–29
Marble A (1934) Diabetes and cancer. N Engl J Med 211:339–349. https://doi.org/10.1056/NEJM193408232110801
Mark H et al (1994) A practical cytogenetic protocol for in vitro cytotoxicity and genotoxicity testing. Ann Clin Lab Sci 24:387–395
Mateuca R, Lombaert N, Aka PV, Decordier I, Kirsch-Volders M (2006) Chromosomal changes: induction, detection methods and applicability in human biomonitoring. Biochimie 88:1515–1531. https://doi.org/10.1016/j.biochi.2006.07.004
Matteucci E, Giampietro O (2011) Dipeptidyl peptidase-4 inhibition: linking chemical properties to clinical safety. Curr Med Chem 18:4753–4760
Moorhead PS, Nowell P, Mellman WJ, Battips DT, Hungerford D (1960) Chromosome preparations of leukocytes cultured from human peripheral blood. Exp Cell Res 20:613–616
OECD (2016a) Test No. 473: In vitro mammalian chromosomal aberration test. OECD Publishing, Paris
OECD (2016b) Test No 487: In vitro mammalian cell micronucleus test. OECD Publishing, Paris
Okutur SK (2015) İki Eski Dost: Diyabet ve Kanser Okmeydanı Tıp Dergisi 31:23–32. https://doi.org/10.5222/otd.2015.023
Oz Gul O et al (2013) Comparative genotoxic and cytotoxic effects of the oral antidiabetic drugs sitagliptin, rosiglitazone, and pioglitazone in patients with type-2 diabetes: a cross-sectional, observational pilot study. Mutat Res 757:31–35. https://doi.org/10.1016/j.mrgentox.2013.04.024
Pinheiro MM, Stoppa CL, Valduga CJ, Okuyama CE, Gorjao R, Pereira RM, Diniz SN (2017) Sitagliptin inhibit human lymphocytes proliferation and Th1/Th17 differentiation in vitro. Eur J Pharm Sci 100:17–24. https://doi.org/10.1016/j.ejps.2016.12.040
Plosker GL (2014) Sitagliptin: a review of its use in patients with type 2 diabetes mellitus. Drugs 74:223–242. https://doi.org/10.1007/s40265-013-0169-1
Rojas E, Herrera LA, Sordo M, Gonsebatt ME, Montero R, Rodriguez R, Ostrosky-Wegman P (1993) Mitotic index and cell proliferation kinetics for identification of antineoplastic activity. Anticancer Drugs 4:637–640
Roshdy HM, Kassem SM (2013) Genetic effects of Januvia and Galvus alone or with metformin on pregnant female mice and their embryos. World Appl Sci J 25:1690–1698. https://doi.org/10.5829/idosi.wasj.2013.25.12.76142
Scott LJ (2017) Sitagliptin: a review in type 2 diabetes. Drugs. 77:1–16
Sen S, He Y, Koya D, Kanasaki K (2014) Cancer biology in diabetes. J Diabetes Invest 5:251–264. https://doi.org/10.1111/jdi.12208
Spagnuolo PA, et al. (2013) Inhibition of intracellular dipeptidyl peptidases 8 and 9 enhances parthenolide’s anti-leukemic activity Leukemia 27:1236 https://doi.org/10.1038/leu.2013.9 https://www.nature.com/articles/leu20139#supplementary-information
Speit G (1984) Considerations on the mechanism of differential Giemsa staining of BrdU-substituted chromosomes. Hum Genet 67:264–269
TEMD (2014) Türkiye Endokrinoloji ve Metabolizma Derneği, Diabetes Mellitus ve Komplikasyonlarının Tanı, Tedavi Ve İzlem Kılavuzu. TEMD Yayınları. Ankara. ISBN 978-605-4011-16-2
Tokur O, Aksoy A (2017) In vitro Sitotoksisite Testleri. Harran Üniversitesi Veteriner Fakültesi Dergisi 6(1):112–118
Tucker JD, Preston RJ (1996) Chromosome aberrations, micronuclei, aneuploidy, sister chromatid exchanges, and cancer risk assessment. Mutat Res/Rev Genet Toxicol 365:147–159
Warburg O (1956) On the origin of cancer. Science 123:309–314
Yang X, Ma R, So WY, Kong A, Xu G, Chan J (2012) Addressing different biases in analysing drug use on cancer risk in diabetes in non-clinical trial settings—what, why and how? Diabetes, Obes Metab 14:579–585
Yuzbasioglu D, Enguzel-Alperen C, Unal F (2018) Investigation of in vitro genotoxic effects of an anti-diabetic drug sitagliptin. Food Chem Toxicol 112:235–241. https://doi.org/10.1016/j.fct.2018.01.003
Acknowledgements
This study was supported by Ondokuz Mayıs University with PYO.TIP.1904-15.030 project numbers. We also thank Seval Kontaş Yedier for her technical help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Börçek Kasurka, C., Elbistan, M., Atmaca, A. et al. In vitro cytogenetic assessment and comparison of vildagliptin and sitagliptin. Cytotechnology 71, 1063–1077 (2019). https://doi.org/10.1007/s10616-019-00345-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-019-00345-y