Abstract
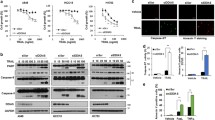
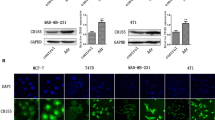
The tumor suppressor DLEC1 has been shown to promote cell proliferation when AP-2α2 is down-regulated in HCT116 stable clones, suggesting its pro-survival nature. However, the pro-survival function of DLEC1 has not been confirmed in other cells and its underlying mechanisms remain elusive. Therefore, we knocked down DLEC1 in a panel of cell lines and found that DLEC1 depletion caused various extents of cell death through intrinsic pathway. DLEC1 overexpression promoted cell survival and reduced cell death in cancer cells after 5-FU treatment, while DLEC1 down-regulation sensitized cancer cells to 5-FU. Further studies demonstrated that DLEC1 attenuated the increase in cleaved PARP, caspase-3 and caspase-7, the activity of caspase-9 and the diffusion of cytosolic cytochrome c from mitochondria. Our data also showed that BCL-XL was up-regulated by DLEC1 in stable clones after 5-FU treatment. Altogether, these results indicated that DLEC1 protects cells against cell death induced by 5-FU through the attenuation of active proteins in caspase cascade and the up-regulation of BCL-XL. Therefore, DLEC1 can be a pro-survival protein under certain circumstances and a potential therapeutic target for increasing sensitivity of cancer cells to 5-FU.





Similar content being viewed by others
References
Brunelle JK, Letai A (2009) Control of mitochondrial apoptosis by the Bcl-2 family. J Cell Sci 122:437–441
Chen S, Dai Y, Pei XY, Grant S (2009) Bim upregulation by histone deacetylase inhibitors mediates interactions with the Bcl-2 antagonist ABT-737: evidence for distinct roles for Bcl-2, Bcl-xL, and Mcl-1. Mol Cell Biol 29:6149–6169. https://doi.org/10.1128/mcb.01481-08
Cotter TG (2009) Apoptosis and cancer: the genesis of a research field. Nat Rev Cancer 9:501–507
Daigo Y, Nishiwaki T, Kawasoe T, Tamari M, Tsuchiya E, Nakamura Y (1999) Molecular cloning of a candidate tumor suppressor gene, DLC1, from chromosome 3p21.3. Can Res 59:1966–1972
Ghiringhelli F, Apetoh L (2015) Enhancing the anticancer effects of 5-fluorouracil: current challenges and future perspectives. Biomed J 38:111–116. https://doi.org/10.4103/2319-4170.130923
Hennessy EJ (2016) Selective inhibitors of Bcl-2 and Bcl-xL: balancing antitumor activity with on-target toxicity. Bioorg Med Chem Lett 26:2105–2114. https://doi.org/10.1016/j.bmcl.2016.03.032
Huang BH, Zhuo JL, Leung CH, Lu GD, Liu JJ, Yap CT, Hooi SC (2012) PRAP1 is a novel executor of p53-dependent mechanisms in cell survival after DNA damage. Cell Death Dis 3:e442. https://doi.org/10.1038/cddis.2012.180
Kwong J et al (2006) Candidate tumor-suppressor gene DLEC1 is frequently downregulated by promoter hypermethylation and histone hypoacetylation in human epithelial ovarian cancer. Neoplasia 8:268–278. https://doi.org/10.1593/neo.05502
Lavrik IN, Krammer PH (2012) Regulation of CD95/Fas signaling at the DISC. Cell Death Differ 19:36–41. https://doi.org/10.1038/cdd.2011.155
Li J, Yuan J (2008) Caspases in apoptosis and beyond. Oncogene 27:6194–6206. https://doi.org/10.1038/onc.2008.297
Longley DB, Harkin DP, Johnston PG (2003) 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer 3:330–338. https://doi.org/10.1038/nrc1074
Longley DB, Wilson TR, McEwan M, Allen WL, McDermott U, Galligan L, Johnston PG (2006) c-FLIP inhibits chemotherapy-induced colorectal cancer cell death. Oncogene 25:838–848. https://doi.org/10.1038/sj.onc.1209122
Miura M (2012) Apoptotic and nonapoptotic caspase functions in animal development. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a008664
Muller FU et al (2004) Transcription factor AP-2alpha triggers apoptosis in cardiac myocytes. Cell Death Differ 11:485–493. https://doi.org/10.1038/sj.cdd.4401383
Nita ME et al (2000) Bcl-X(L) antisense sensitizes human colon cancer cell line to 5-fluorouracil Japanese journal of cancer research. Gann 91:825–832
Orso F et al (2008) AP-2alpha and AP-2gamma regulate tumor progression via specific genetic programs. FASEB J 22:2702–2714. https://doi.org/10.1096/fj.08-106492
Ow YP, Green DR, Hao Z, Mak TW (2008) Cytochrome c: functions beyond respiration. Nat Rev Mol Cell Biol 9:532–542. https://doi.org/10.1038/nrm2434
Qiu GH et al (2004) The candidate tumor suppressor gene BLU, located at the commonly deleted region 3p21.3, is an E2F-regulated, stress-responsive gene and inactivated by both epigenetic and genetic mechanisms in nasopharyngeal carcinoma. Oncogene 23:4793–4806
Qiu GH et al (2008) The tumor suppressor gene DLEC1 is frequently silenced by DNA methylation in hepatocellular carcinoma and induces G1 arrest in cell cycle. J Hepatol 48:433–441. https://doi.org/10.1016/j.jhep.2007.11.015
Qiu GH, Xie X, Deng L, Hooi SC (2015) Tumor suppressor DLEC1 can stimulate the proliferation of cancer cells when AP-2a2 is down-regulated in HCT116. Hepat Mon 15:e29829. https://doi.org/10.5812/hepatmon.29829
Rolland SG, Conradt B (2010) New role of the BCL2 family of proteins in the regulation of mitochondrial dynamics. Curr Opin Cell Biol 22:852–858. https://doi.org/10.1016/j.ceb.2010.07.014
Safa AR (2012) c-FLIP, a master anti-apoptotic regulator. Exp Oncol 34:176–184
Seng TJ et al (2008) DLEC1 and MLH1 promoter methylation are associated with poor prognosis in non-small cell lung carcinoma. Br J Cancer 99:375–382. https://doi.org/10.1038/sj.bjc.6604452
Seven D, Yavuz E, Kilic E, Baltaci E, Karaman E, Ulutin T, Buyru N (2015) DLEC1 is not silenced solely by promoter methylation in head and neck squamous cell carcinoma. Gene 563:83–86. https://doi.org/10.1016/j.gene.2015.03.004
Simonian PL, Grillot DA, Nunez G (1997) Bcl-2 and Bcl-XL can differentially block chemotherapy-induced cell death. Blood 90:1208–1216
Vilenchik M, Raffo AJ, Benimetskaya L, Shames D, Stein CA (2002) Antisense RNA down-regulation of bcl-xL expression in prostate cancer cells leads to diminished rates of cellular proliferation and resistance to cytotoxic chemotherapeutic agents. Can Res 62:2175–2183
Wlodkowic D, Telford W, Skommer J, Darzynkiewicz Z (2011) Apoptosis and beyond: cytometry in studies of programmed cell death. Methods Cell Biol 103:55–98. https://doi.org/10.1016/b978-0-12-385493-3.00004-8
Wyatt MD, Wilson DM III (2009) Participation of DNA repair in the response to 5-fluorouracil. Cell Mol Life Sci 66:788–799. https://doi.org/10.1007/s00018-008-8557-5
Yang SY, Sales KM, Fuller B, Seifalian AM, Winslet MC (2009) Apoptosis and colorectal cancer: implications for therapy. Trends Mol Med 15:225–233. https://doi.org/10.1016/j.molmed.2009.03.003
Ye X, Feng G, Jiao N, Pu C, Zhao G, Sun G (2014) Methylation of DLEC1 promoter is a predictor for recurrence in Chinese patients with gastric cancer. Dis Mark 2014:804023. https://doi.org/10.1155/2014/804023
Ying J et al (2009) DLEC1 is a functional 3p22.3 tumour suppressor silenced by promoter CpG methylation in colon and gastric cancers. Br J Cancer 100:663–669. https://doi.org/10.1038/sj.bjc.6604888
Zhang L et al (2015) DLEC1, a 3p tumor suppressor, represses NF-kappaB signaling and is methylated in prostate cancer. J Mol Med 93:691–701. https://doi.org/10.1007/s00109-015-1255-5
Zhu H et al (2005) Bcl-XL small interfering RNA suppresses the proliferation of 5-fluorouracil-resistant human colon cancer cells. Mol Cancer Ther 4:451–456. https://doi.org/10.1158/1535-7163.mct-04-0162
Zinkel S, Gross A, Yang E (2006) BCL2 family in DNA damage and cell cycle control. Cell Death Differ 13:1351–1359. https://doi.org/10.1038/sj.cdd.4401987
Acknowledgements
This study was supported by the Major Project of Fujian Provincial Science and Technology Department (2014NZ0002-3), Longyan Science and Technology Scheme (2017LY73), Natural Science Foundation of Fujian Province (2018J01456) and the National University of Singapore ARF Grants R-185-000-074-112 and R-185-000-087-112.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Qiu, GH., Que, W., Yan, S. et al. The pro-survival function of DLEC1 and its protection of cancer cells against 5-FU-induced apoptosis through up-regulation of BCL-XL. Cytotechnology 71, 23–33 (2019). https://doi.org/10.1007/s10616-018-0258-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-018-0258-9