Abstract
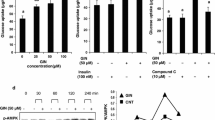
Enterolactone (ENL) is formed by the conversion of dietary precursors like strawberry lignans via the gut microbiota. Urinary concentrations of lignan metabolites are reported to be significantly associated with a lower risk of Type 2 diabetes (T2D). In the present study, antidiabetic effect of ENL and its modes of action were studied in vitro and in vivo employing a rat skeletal muscle-derived cell line, L6 myocytes in culture, and T2D model db/db mice. ENL dose-dependently increased glucose uptake in L6 myotubes under insulin absent condition. This increase by ENL was canceled by compound C, an inhibitor of 5′-adenosine monophosphate-activated protein kinase (APMK). Activation (=phosphorylation) of AMPK and translocation of glucose transporter 4 (GLUT4) to plasma membrane in L6 myotubes were demonstrated by Western blotting analyses. Promotion by ENL of GLUT4 translocation to plasma membrane was also visually demonstrated by immunocytochemistry in L6 myoblasts that were transfected with glut4 cDNA-coding vector. T2D model db/db mice were fed the basal 20 % casein diet (20C) or 20C supplemented with ENL (0.001 or 0.01 %) for 6 weeks. Fasting blood glucose (FBG) levels were measured every week and intraperitoneal glucose tolerance test (IPGTT) was conducted. ENL at a higher dose (0.01 % in 20C) suppressed the increases in FBG levels. ENL was also demonstrated to improve the index of insulin resistance (HOMA-IR) and glucose intolerance by IPGTT in db/db mice. From these results, ENL is suggested to be an antidiabetic chemical entity converted from dietary lignans by gut microbiota.





Similar content being viewed by others
Abbreviations
- AMPK:
-
5′-Adenosine monophosphate-activated protein kinase
- ENL:
-
Enterolactone
- FBG:
-
Fasting blood glucose
- GLUT4:
-
Glucose transporter 4
- HOMA-IR:
-
Homeostasis model assessment of insulin resistance
- IPGTT:
-
Intraperitoneal glucose tolerance test
- KHH buffer:
-
Krebs–Henseleit–Hepes buffer
- TBARS:
-
Thiobarbituric acid-reactive substances
- TG:
-
Triglyceride
References
Bach Knudsen KE, Serena A, Kjaer AK, Tetens I, Heinonen SM, Nurmi T, Adlercreutz H (2003) Rye bread in the diet of pigs enhances the formation of enterolactone and increases its levels in plasma, urine and feces. J Nutr 133:1368–1375
Cheong SH, Furuhashi K, Ito K, Nagaoka M, Yonezawa T, Miura Y, Yagasaki K (2014a) Daidzein promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and improves glucose homeostasis in Type 2 diabetic model mice. J Nutr Biochem 25:136–143
Cheong SH, Furuhashi K, Ito K, Nagaoka M, Yonezawa T, Miura Y, Yagasaki K (2014b) Antihyperglycemic effect of equol, a daidzein derivative, in cultured L6 myocytes and ob/ob mice. Mol Nutr Food Res 58:267–277
Fecchi K, Volonte D, Hezel MP, Schmeck K, Galbiati F (2006) Spatial and temporal regulation of GLUT4 translocation by flotillin-1 and caveolin-3 in skeletal muscle cells. FASEB J 20:705–707
Giampieri F, Tulipani S, Alvarez-Suarez JM, Quiles JL, Mezzetti B, Battino M (2012) The strawberry: composition, nutritional quality, and impact on human health. Nutrition 28:9–19
Giampieri F, Alvarez-Suarez JM, Battino M (2014) Strawberry and human health: effects beyond antioxidant activity. J Agric Food Chem 62:3867–3876
Ha BG, Nagaoka M, Yonezawa T, Tanabe R, Woo JT, Kato H, Chung UI, Yagasaki K (2012) Regulatory mechanism for the stimulatory action of genistein on glucose uptake in vitro and in vivo. J Nutr Biochem 23:501–509
Ha BG, Yonezawa T, Son MJ, Woo JT, Ohba S, Chung UI, Yagasaki K (2014) Antidiabetic effect of nepodin, a component of Rumex roots, and its modes of action in vitro and in vivo. BioFactors 40:436–447
Kamakura R, Son MJ, de Beer D, Joubert E, Miura Y, Yagasaki K (2015) Antidiabetic effect of green rooibos (Aspalathus linearis) extract in cultured cells and type 2 diabetic model KK-Ay mice. Cytotechnology 67:699–710
Kawano A, Nakamura H, Hata S, Minakawa M, Miura Y, Yagasaki K (2009) Hypoglycemic effect of aspalathin, a rooibos tea component from Aspalathus linearis, in type 2 diabetic model db/db mice. Phytomedicine 16:437–443
Krook A, Wallberg-Henriksson H, Zierath JR (2004) Sending the signal: molecular mechanisms regulating glucose uptake. Med Sci Sports Exerc 36:1212–1217
Lee YS, Cha BY, Saito K, Yamakawa H, Choi SS, Yamaguchi K, Yonezawa T, Teruya T, Nagai K, Woo JT (2010) Nobiletin improves hyperglycemia and insulin resistance in obese diabetic ob/ob mice. Biochem Pharmacol 79:1674–1683
Mazur WM, Uehara M, Wähälä K, Adlercreutz H (2000) Phyto-oestrogen content of berries, and plasma concentrations and urinary excretion of enterolactone after a single strawberry-meal in human subjects. Br J Nutr 83:381–387
Minakawa M, Kawano A, Miura Y, Yagasaki K (2011) Hypoglycemic effect of resveratrol in type 2 diabetic model db/db mice and its actions in cultured L6 myotubes and RIN-5F pancreatic β-cells. J Clin Biochem Nutr 48:237–244
Minakawa M, Miura Y, Yagasaki K (2012) Piceatannol, a resveratrol derivative, promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and suppresses blood glucose levels in type 2 diabetic model db/db mice. Biochem Biophys Res Commun 422:469–475
Miura D, Saarinen NM, Miura Y, Santti R, Yagasaki K (2007) Hydroxymatairesinol and its mammalian metabolite enterolactone reduce the growth and metastasis of subcutaneous AH109A hepatomas in rats. Nutr Cancer 58:49–59
Nishiumi S, Ashida H (2007) Rapid preparation of a plasma membrane fraction from adipocytes and muscle cells: application to detection of translocated glucose transporter 4 on the plasma membrane. Biosci Biotechnol Biochem 71:2343–2346
Park SJ, Ahmad F, Philp A, Baar K, Williams T, Luo H, Ke H, Rehmann H, Taussig R, Brown AL, Kim MK, Beaven MA, Burgin AB, Manganiello V, Chung JH (2012) Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 148:421–433
Rowland IR, Wiseman H, Sanders TA, Adlercreutz H, Bowey EA (2000) Interindividual variation in metabolism of soy isoflavones and lignans: influence of habitual diet on equol production by the gut microflora. Nutr Cancer 36:27–32
Saarinen NM, Wärri A, Airio M, Smeds A, Mäkelä S (2007) Role of dietary lignans in the reduction of breast cancer risk. Mol Nutr Food Res 51:857–866
Setchell KDR, Lawson AM, Borriello SP, Harkness R, Gordon H, Morgan DM, Kirk DN, Adlercreatz H, Anderson LC, Axelson M (1981) Lignan formation in man–microbial involvement and possible roles in relation to cancer. Lancet 2:4–7
Setchell KDR, Brown NM, Zimmer-Nechemias L, Wolfe B, Jha P, Heubi JE (2014) Metabolism of secoisolariciresinol-diglycoside the dietary precursor to the intestinally derived lignan enterolactone in humans. Food Funct 5:491–501
Son MJ, Minakawa M, Miura Y, Yagasaki K (2013) Aspalathin improves hyperglycemia and glucose intolerance in obese diabetic ob/ob mice. Eur J Nutr 52:1607–1619
Son MJ, Miura Y, Yagasaki K (2015) Mechanisms for antidiabetic effect of gingerol in cultured cells and obese diabetic model mice. Cytotechnology 67:641–652
Sun Q, Wedick NM, Pan A, Townsend MK, Cassidy A, Franke AA, Rimm EB, Hu FB, van Dam RM (2014) Gut microbiota metabolites of dietary lignans and risk of type 2 diabetes: a prospective investigation in two cohorts of U.S. women. Diabetes Care 37:1287–1295
Vanharanta M, Voutilainen S, Lakka TA, van der Lee M, Adlercreutz H, Salonen JT (1999) Risk of acute coronary events according to serum concentrations of enterolactone: a prospective population-based case-control study. Lancet 354:2112–2215
Yagasaki K (2014) Anti-diabetic phytochemicals that promote GLUT4 translocation via AMPK signaling in muscle cells. Nutr Aging 2:35–44
Yagasaki K, Morisaki N, Kitahara Y, Miura A, Funabiki R (2003) Involvement of protein kinase C activation in l-leucine-induced stimulation of protein synthesis in L6 myotubes. Cytotechnology 43:97–103
Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N, Hirshman MF, Goodyear LJ, Moller DE (2001) Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 108:1167–1174
Acknowledgments
This work was supported in part by the Regional Innovation Strategy Support Program, MEXT, Japan, in part by the Japan Society for the Promotion of Science, Japan, and in part by the Tojuro Iijima Foundation for Food Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhou, F., Furuhashi, K., Son, M.J. et al. Antidiabetic effect of enterolactone in cultured muscle cells and in type 2 diabetic model db/db mice. Cytotechnology 69, 493–502 (2017). https://doi.org/10.1007/s10616-016-9965-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-016-9965-2