Abstract
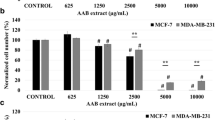
Plants are still to be explored for new anti-cancer compounds because overall success in cancer treatment is still not satisfactory. As a new possible source for such compounds, the lichens are recently taking a great attention. We, therefore, explored both the genotoxic and anti-growth properties of lichen species Parmelia sulcata Taylor. The chemical composition of P. sulcata was analyzed with comprehensive gas chromatography–time of flight mass spectrometry. Anti-growth effect was tested in human breast cancer cell lines (MCF-7 and MDA-MB-231) by the MTT and ATP viability assays, while the genotoxic activity was studied by assays for micronucleus, chromosomal aberration and DNA fragmentation in human lymphocytes culture. Cell death modes (apoptosis/necrosis) were morphologically assessed. P. sulcata inhibited the growth in a dose-dependent manner up to a dose of 100 μg/ml and induced caspase-independent apoptosis. It also showed genotoxic activity at doses (>125 μg/ml) higher than that required for apoptosis. These results suggest that P. sulcata may induce caspase-independent apoptotic cell death at lower doses, while it may be genotoxic at relatively higher doses.





Similar content being viewed by others
References
Andreotti PE, Cree IA, Kurbacher CM, Hartmann DM, Linder D, Harel G, Gleiberman I, Caruso PA, Ricks SH, Untch M (1995) Chemosensitivity testing of human tumors using a microplate adenosine triphosphate luminescence assay: clinical correlation for cisplatin resistance of ovarian carcinoma. Cancer Res 55:5276–5282
Ari F, Celikler S, Oran S, Balikci N, Ozturk S, Ozel MZ, Ozyurt D, Ulukaya E (2012) Genotoxic, cytotoxic and apoptotic effects of Hypogymnia physodes (L.) Nyl. on breast cancer cells. Environ Toxicol. doi:10.1002/tox.21809
Aslan A, Gulluce M, Agar G, Karadayi M, Bozari S, Orhan F (2012) Mutagenic and antimutagenic properties of some lichen species grown in the Eastern Anatolia Region of Turkey. Tsitol Genet 46:36–42
Benn PA, Perle MA (1992) Chromosome staining and banding techniques. In: Rooney DE, Czepulkowsky BH (eds) Human cytogenetics: a practical approach I–II. IRL Press, Oxford, pp 7–83
Bezivin C, Tomasi S, Lohezic-Le Devehat F, Boustie C (2003) Cytotoxic activity of some lichen extracts on murine and human cancer cell lines. Phytomedicine 10:499–503
Bonassi S, Fenech M, Lando C (2001) Human micronucleus project: international database comparison for results with the cytokinesis block micronucleus assay in human lymphocytes: effect of laboratory protocol, scoring criteria, and host factors on the frequency of micronuclei. Environ Mol Mutagen 37:31–45
Brodo IM, Sharnoff SD, Sharnoff S (2001) Lichens of North America. Yale University Press, New Haven
Fenech M (2000) The in vitro micronucleus technique. Mutat Res 455:81–95
Gown AM, Willingham MC (2002) Improved detection of apoptotic cells in archival paraffin sections: immunohistochemistry using antibodies to cleaved caspase 3. J Histochem Cytochem 50:449–454
IPCS (1985) Guide to short-term tests for detecting mutagenic and carcinogenic chemicals, vol. 208. Environmental Health Criteria, World Health Organization, Geneva, pp 102–103
Koparal AT, Tüylü BA, Türk H (2006) In vitro cytotoxic activities of (+)-usnic acid and (−)-usnic acid on V79, A549, and human lymphocyte cells and their non-genotoxicity on human lymphocytes. Nat Prod Res 20:1300–1307
Kosanić M, Ranković B, Vukojević J (2011) Antioxidant properties of some lichen species. J Food Sci Technol 48:584–590
Kosanić M, Ranković RB, Stanojković TP (2012) Antioxidant, antimicrobial and anticancer activities of three Parmelia species. J Sci Food Agric 92:1909–1916
Manojlović NT, Vasiljevic P, Juskovic M, Najman S, Jankovic S, Milenkovic-Andjelkovic A (2010) HPLC analysis and cytotoxic potential of extracts from the lichen, Thamnolia vermicularis var. subuliformis. J Med Plant Res 49:817–823
Manojlović N, Ranković B, Kosanić M, Vasiljević P, Stanojković T (2012) Chemical composition of three Parmelia lichens and antioxidant, antimicrobial and cytotoxic activities of some their major metabolites. Phytomedicine 19:1166–1172
Mitrović T, Stamenković S, Cvetković V, Tośić S, Stanković M, Radojević I, Stefanović O, Ljiljana Č, Dragana Ð, Ćurčić M, Marković S (2011) Antioxidant, antimicrobial and antiproliferative activities of five lichen species. Int J Mol Sci 12:5428–5448
Molnar K, Farkas E (2010) Current results on biological activities of lichen secondary metabolites: a review. Z Naturforsch 65:157–173
Müller K (2001) Pharmaceutically relevant metabolites from lichens. Appl Microbiol Biotechnol 56:9–16
Oksanen I (2006) Ecological and biotechnological aspects of lichens. Appl Microbiol Biotechnol 73:723–734
Ozel MZ, Gogus F, Lewis AC (2006) Determination of Teucrium chamaedrys volatiles by using direct thermal desorption–comprehensive two-dimensional gas chromatography–time-of-flight mass spectrometry. J Chromatogr A 1114:164–169
Park SJ, Wu CH, Gordon JD, Zhong X, Emami A, Safa AR (2004) Taxol induces caspase-10-dependent apoptosis. J Biol Chem 279:51057–51067
Perry NB, Benn MH, Brennan NJ, Burgess EJ, Elliss G, Galloway DJ, Lorimer SD, Tangney RS (1999) Antimicrobial, antiviral and cytotoxic activity of New Zealand lichens. Lichenologist 31:627–636
Purvis OW, Coppins BJ, Hawskworth DL, James PW, Moore DM (1994) The lichen flora of Great Britain and Ireland. Natural History Museum Publications. The British Lichen Society, London, pp 421–437
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Sommers CL, Walker-Jones D, Heckford SE, Worland P, Valverius E, Clark R, McCormick F, Stampfer M, Abularach S, Gelmann EP (1989) Vimentin rather than keratin expression in some hormone-independent breast cancer cell lines and in oncogene-transformed mammary epithelial cells. Cancer Res 49:4258–4263
Triggiani D, Ceccarelli D, Tiezzi A, Pisani T, Munzi S, Gaggi C, Loppi S (2009) Antiproliferative activity of lichen extracts on murine myeloma cells. Biologia 64:59–62
Ulukaya E, Ozdikicioglu F, Oral AY, Demirci M (2008) The MTT assay yields a relatively lower result of growth inhibition than the ATP assay depending on the chemotherapeutic drugs tested. Toxicol In Vitro 22:232–239
Wirth W (1995) Die Flechten Baden-Württembergs. Teil 1-2. Verlag Eugen Ulmer, Stuttgart, pp 637–669
Zeytinoglu H, Incesu Z, Tuylu BA, Turk AO, Barutca B (2008) Determination of genotoxic, antigenotoxic and cytotoxic potential of the extract from lichen Cetraria aculeata (Schreb.) Fr. in vitro. Phytother Res 22:118–123
Acknowledgments
We appreciate the Research Fund of Uludag University for the Project that is numbered UAP(F)-2011/42 as for providing us with the kits/chemicals. The authors would like to thank Neslihan Onder Ozdemir (Lecturer of Medical English, Uludag University) for her comments on linguistic improvement. We thank Prof. Dr. Ayhan Bilir (Istanbul University, Turkey) for the cell lines and thank Nazlihan Aztopal, Mehmet Sarimahmut and Sezin Bozdemir for technical assistance.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ari, F., Ulukaya, E., Oran, S. et al. Promising anticancer activity of a lichen, Parmelia sulcata Taylor, against breast cancer cell lines and genotoxic effect on human lymphocytes. Cytotechnology 67, 531–543 (2015). https://doi.org/10.1007/s10616-014-9713-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-014-9713-4