Abstract
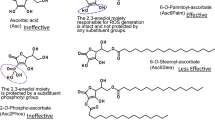
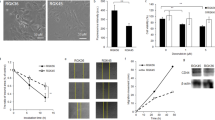
The aim of the present study is to determine the anti-proliferative activity of 6-o-palmitoyl-l-ascorbic acid (Asc6Palm) that is a lipophilic derivative of l-ascorbic acid (Asc), on human tongue squamous carcinoma HSC-4 cells by combined use of hyperthermia in comparison to Asc. Asc6Palm or Asc were administered to HSC-4 cells for 1 h, to which hyperthermia at 42 °C was applied for initial 15 min. After further 1–72 h incubation at 37 °C, cell proliferation was determined with Crystal Violet staining. Ascorbyl radical (AscR) in HSC-4 cell suspension was measured by electron spin resonance (ESR), and cell morphology was observed with scanning electron microscopy (SEM). At 37 °C, 4 mM Asc or 0.35 mM Asc6Palm were enough to suppress proliferation of HSC-4 cells. By combined use of hyperthermia at 42 °C, cell proliferation was decreased when compared to 37 °C. After Asc of 4 mM was incubated with HSC-4 cell suspensions at 37 °C or 42 °C for 0–180 min, the signal intensity of ascorbyl radical (AscR) by ESR was not different regardless of the presence or absence of cells at 37 °C, whereas AscR signal was enlarged in the presence of HSC-4 cells at 42 °C. It was suggested that oxidation of Asc occurred rapidly in HSC-4 cells by hyperthermia, and thereby enhanced the anti-proliferative activity. By SEM observation, the surface of HSC-4 cells treated with Asc6Palm revealed distinct morphological changes. Thus, the combined regimen of Asc6Palm and hyperthermia is expected to exert a marked antitumor activity.






Similar content being viewed by others
Abbreviations
- Asc6Palm:
-
6-o-palmitoyl-l-ascorbic acid
- Asc:
-
l-ascorbic acid
- AscR:
-
Ascorbyl radical
- ESR:
-
Electron spin resonance
- SEM:
-
Scanning electron microscopy
- CRet system:
-
Capacitive-resistive transfer system
References
Cameron E, Pauling L, Leibovitz B (1979) Ascorbic acid and cancer. Cancer Res 39:663–681
Chen P, Yang LL, Wang YS, Li G, Fang F, Liu K, Li J, Zhao X, Hu HZ, Wei YQ (2008) Synergistic antitumor effect of CXCL10 with hyperthermia. J Cancer Res Clin Oncol 134:679–687
Chitnis MP, Adwankar MK (1986) Enhancement of the in vitro cytotoxicity of bouvardin by verapamil alone and combined with hyperthermia in Sarcoma 180 and Ehrlich ascites carcinoma cells. J Cancer Res Clin Oncol 112:131–134
Cress AE, Culver PS, Moon TE, Garner EW (1982) Correlation between amounts of cellular membrane components and sensitivity to hyperthermia in a variety of mammalian cell lines in culture. Cancer Res 42:1716–1721
Currie RW, White FP (1981) Trauma-induced protein in rat tissues: a physiological role for a “heat shock” protein? Science 214:72–73
Dietzel F (1983) Basic principles in hyperthermic tumor therapy. Recent Results Cancer Res 86:177–190
Dodd NJ (1973) Some EPR signals in tumour tissue. Br J Cancer 28:257–262
Dodd NJ, Swartz HM (1984) The nature of the ESR signal in lyophilized tissue and its relevance to malignancy. Br J Cancer 49:65–71
Engin K (1996) Biological rationale and clinical experience with hyperthermia. Control Clin Trials 17:316–342
Fiaschi AI, Cozzolino A, Ruggiero G, Giorgi G (2005) Glutathione, ascorbic acid and antioxidant enzymes in the tumor tissue and blood of patients with oral squamous cell carcinoma. Eur Rev Med Pharmacol Sci 9:361–367
Flick DA, Gifford GE (1984) Comparison of in vitro cell cytotoxic assays for tumor necrosis factor. J Immunol Methods 68:167–175
Gillies RJ, Didier N, Denton M (1976) Determination of cell number in monolayer cultures. Anal Biochem 159:109–113
Giovanella BC, Stehlin JS, Yim SO (1980) Correlation of the thermosensitivity of cells to their malignant potential. Ann N Y Acad Sci 335:206–214
Hacker MP, Khokhar AR, Brown DB, McCormack JJ, Kracoff IH (1985) Ascorbate (1, 2-diaminocyclohexane): platinum (II) complexes, a new series of water-soluble antitumor drugs. Cancer Res 45:4748–4753
Hernández-Bule ML, Trillo MA, Cid MA, Leal J, Ubeda A (2007) In vitro exposure to 0.57-MHz electric currents exerts cytostatic effects in HepG2 human hepatocarcinoma cells. Int J Oncol 30:583–592
Kageyama K, Onoyama Y, Kimura M, Yamazaki H, Miwa N (1991) Enhanced inhibition of DNA synthesis and release of membrane phospholipids in tumour cells treated with a combination of acylated ascorbate and hyperthermia. Int J Hyperthermia 7:85–91
Kageyama K, Onoyama Y, Otani S, Matsui-Yuasa I, Nagao N, Miwa N (1995) Enhanced inhibitory effects of hyperthermia combined with ascorbic acid on DNA synthesis in Ehrlich ascites tumor cells grown at a low cell density. Cancer Biochem Biophys 14:273–280
Kageyama K, Onoyama Y, Otani S, Kimura M, Matsui-Yuasa I, Nagao N, Miwa N (1996) Promotive action of acylated ascorbate on cellular DNA synthesis and growth at low doses in contrast to inhibitory action at high doses or upon combination with hyperthermia. J Cancer Res Clin Oncol 122:41–44
Kampinga HH (2006) Cell biological effects of hyperthermia alone or combined with radiation or drugs: a short introduction to newcomers in the field. Int J Hyperthermia 22:191–196
Kusumoto K, Kageyama K, Matsuda T, Tomura TT, Munakata H, Tanaka H, Yazama F, Miwa N (2007) Effects of ethyl-esterization, chain length, unsaturation degrees, and hyperthermia on carcinostatic effect of omega-hydroxylated fatty acids. Exp Oncol 29:106–110
Liu JW, Nagao N, Kageyama K, Miwa N (2000) Anti-metastatic effect of an autooxidation-resistant and lipophilic ascorbic acid derivative through inhibition of tumor invasion. Anticancer Res 20:113–118
Liu JW, Kayasuga A, Nagao N, Masatsuji-Kato E, Tuzuki T, Miwa N (2003) Repressions of actin assembly and RhoA localization are involved in inhibition of tumor cell motility by lipophilic ascorbyl phosphate. Int J Oncol 23:1561–1567
Lohmann W, Holz D (1984) Structure of ascorbic acid and its biological function. I. ESR determination of the ascorbyl radical in biological samples and in model systems. Biophys Struct Mech 10:197–204
Miwa N, Yamazaki H (1986) Potentiated susceptibility of ascites tumor to acyl derivatives of ascorbate caused by balanced hydrophobicity in the molecule. Exp Cell Biol 54:245–249
Miwa N, Yamazaki H, Ikari Y (1986) Enhancement of ascorbate cytotoxicity by chelation with ferrous ions through prolonged duration of the action. Anticancer Res 6:1033–1036
Miwa N, Yamazaki H, Nagaoka Y, Kageyama K, Onoyama Y, Matsui-Yuasa I, Otani S, Morisawa S (1988) Altered production of the active oxygen species is involved in enhanced cytotoxic action of acylated derivatives of ascorbate to tumor cells. Biochim Biophys Acta 97:144–151
Pajonk F, Ophoven A, McBride WH (2005) Hyperthermia-induced proteasome inhibition and loss of androgen receptor expression in human prostate cancer cells. Cancer Res 65:4836–4843
Pauling L (1991) Effect of ascorbic acid on incidence of spontaneous mammary tumors and UV-light-induced skin tumors in mice. Am J Clin Nutr 54:1252S–1255S
Pauling L, Willoughby R, Reynolds R, Blaisdell BE, Lawson S (1982) Incidence of squamous cell carcinoma in hairless mice irradiated with ultraviolet light in relation to intake of ascorbic acid (vitamin C) and of D, L-alpha-tocopheryl acetate (vitamin E). Int J Vitam Nutr Res Suppl 23:53–82
Pennacchioli E, Fiore M, Gronchi A (2009) Hyperthermia as an adjunctive treatment for soft-tissue sarcoma. Expert Rev Anticancer Ther 9:199–210
Roginsky VA, Stegmann HB (1994) Ascorbyl radical as natural indicator of oxidative stress: quantitative regularities. Free Radic Biol Med 17:93–103
Satoh K, Sakagami H, Nakamura K (1996) Enhancement of radical intensity and cytotoxic activity of ascorbate by hyperthermia. Anticancer Res 16:2987–2991
Shammari MA, Watmough DJ, Kerr CL, Gregory DW, Wheatley DN (1990) Combined effects of hyperthermia (to 45 degrees C) and ultrasound irradiation on the surface ultrastructure of HeLa cells. Int J Hyperthermia 6:571–580
Silcock JM, Dodd NJ (1976) ESR study of changes during development of solid Yoshida tumour I: Ascorbyl radical. Br J Cancer 34:550–555
Stich HF, Wei L, Whiting RF (1979) Enhancement of the chromosome-damaging action of ascorbate by transition metals. Cancer Res 39:4145–4151
Symonds RP, Wheldon TE (1984) Morphological changes in L1210 leukaemic cells induced by hyperthermia. Scan Electron Microsc Pt 2:825–830
Takahashi I, Emi Y, Hasuda S, Kakeji Y, Maehara Y, Sugimachi K (2002) Clinical application of hyperthermia combined with anticancer drugs for the treatment of solid tumors. Surgery 131:S78–S84
Tanaka H, Kageyama K, Kusumoto K, Asada R, Miwa N (2007) Antitumor and anti-invasive effects of diverse new macrocyclic lactones, alkylolides and alkenyloides, and their enhancement by hyperthermia. Oncol Rep 18:1257–1262
van Bree C, van der Maat B, Ceha HM, Franken NA, Haveman J, Bakker PJ (1999) Inactivation of p53 and of pRb protects human colorectal carcinoma cells against hyperthermia-induced cytotoxicity and apoptosis. J Cancer Res Clin Oncol 125:549–555
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kato, S., Asada, R., Kageyama, K. et al. Anticancer effects of 6-o-palmitoyl-ascorbate combined with a capacitive-resistive electric transfer hyperthermic apparatus as compared with ascorbate in relation to ascorbyl radical generation. Cytotechnology 63, 425–435 (2011). https://doi.org/10.1007/s10616-011-9363-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-011-9363-8